
eBook - ePub
The Behavioral, Molecular, Pharmacological, and Clinical Basis of the Sleep-Wake Cycle
- 220 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
The Behavioral, Molecular, Pharmacological, and Clinical Basis of the Sleep-Wake Cycle
About this book
The Behavioral, Molecular, Pharmacological, and Clinical Basis of the Sleep-Wake Cycle provides the first comprehensive overview on the molecular methodologies used to evaluate sleep while also examining the cellular, biochemical, genetic, and therapeutic aspects of the sleep-wake cycle. There have been profound changes in the landscape of approaches to the study of sleep – mainly in the areas of molecular biology and molecular techniques. With this great focus on using multidisciplinary molecular methods, chapters address significant advances in the molecular mechanisms underlying sleep and the techniques researchers use to study this phenomenon.
Written by world-leading experts in the area, this book is of great interest to researchers working in the sleep field and to anyone interested in one of the most mysterious phenomena in science – why we sleep and why we cannot survive without it.
- Reviews the neurobiological and cellular mechanisms of the sleep-wake cycle
- Provides the implications of sleep in health and disease
- Contrasts different techniques to study molecular mechanisms
- Contains case studies to better illustrate points
- Covers sleep disturbance and health problems involved in sleep
- Includes chapters on the ontogeny of sleep, along with multiple mechanisms for sleep generation
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
PhysiologyChapter 1
The Sleep–Wake Cycle: An Overview
Timothy Roehrs1,2 and Thomas Roth1,2, 1Sleep Disorders and Research Center, Henry Ford Health System, Detroit, MI, United States, 2Department of Psychiatry and Behavioral Neurosciences, School of Medicine, Wayne State University, Detroit, MI, United States
Abstract
This chapter provides an overview of the sleep–wake cycle. We describe the nature of the electrophysiology of normal sleep and the two distinct sleep states, rapid eye movement (REM) and nonrapid eye movement (NREM). The differential endocrine, autonomic, respiratory, thermal physiology and regulation, and cognitive processing associated with REM and NREM are outlined. The interactive, but independent, control of sleep and wakefulness by homeostatic and circadian processes and the ultradian regulation of REM and NREM states within sleep is also outlined. We provide an overview of the specific neurobiological mechanisms that control sleep and wakefulness and the circadian mechanisms that control the daily timing of sleep and wakefulness. Finally, we briefly discuss some factors that alter the nature and characteristics of sleep and wakefulness.
Keywords
NREM; REM; homeostatic; circadian; ultradian; sleep–wake neurobiology
1.1 Introduction
The sleep–wake cycle in healthy humans is a 24-hour cycle composed of approximately one-third sleep and two-thirds wake. The sleep–wake cycle is under complex, interacting circadian and homeostatic processes. Within the 24-hour sleep–wake cycle is a 90–120 minutes ultradian cycle (basic rest activity cycle), most clearly evident during sleep, but also hypothesized as being present during wakefulness. Sleep and circadian bioscientists continue to amass information regarding the genetic and neurobiological mechanisms underlying the sleep–wake cycle with information about some of the basic features and mechanisms emerging, but there is much yet to be discovered.
Sleep is a vital behavior with the appetitive and essential nature of sleep clearly evident in a human’s inability to maintain wakefulness for more than 2 or 3 days. As the state of sleep need progressively increases during periods of sustained wakefulness (i.e., sleep deprivation), brief microsleeps begin to intrude into wakefulness during ongoing behavior and during periods of inactivity. As sleep drive further increases it is expressed as longer episodes of unintended sleep (i.e., naps).1 This vital, compulsory nature of sleep is in contrast to one’s ability to food or fluid deprive oneself to death.
Sleep is characterized by a stereotypic posture, minimal movement, reduced responsivity to stimuli, reversibility, and species-specific diurnal timing and duration. In humans, sleep is recognized behaviorally by recumbence and eye closure, but some mammals sleep with eyes open (e.g., cattle) or while standing (e.g., horse, elephant).2 The immobility of human sleep is relative in that sleep walking and talking occur in some human sleep disorders and among animals some fish swim in place and mammals move periodically. The sleep state can be differentiated from death, coma, and hibernation by the characteristics of arousability and rapid reversibility. Sensory (nonvisual) monitoring of both exogenous and endogenous stimulation continues during sleep such that, for example, the vital stimulus of hypoxemia arouses even a severely sleep deprived individual and parents arouse to the cry of their baby. Further, sensory discrimination occurs as the parent does not arouse to the cry of another baby whose cry is of a similar stimulus intensity. Among mammals the daily duration of sleep varies from 2 to 20 hours with that of humans being approximately 8 hours.3 Larger animals have less daily sleep; for example, elephants sleep about 3 hours per day, while the chipmunk sleeps about 16 hours. Sleep is very light or absent during migration or postpartum in some birds and fish and northern fur seals sleep with one half of the brain at a time. Sleep in adult humans, in many, but not all cultures, occurs as a single bout during the dark hours, while for various other mammals sleep occurs in multiple bouts and for some mammals sleep is linked to the light period.
Sleep scientists measure sleep electrophysiologically, as behavioral assessment of sleep and its intensity by testing arousability or reversibility is obtrusive and disruptive of the very state being assessed.4 Electrophysiological measures correlate well with behavioral observations, but they further reveal subtleties that are not apparent behaviorally and subjectively. For example, some sleep disorders are associated with brief (3–15 seconds) electroencephalographic (EEG) arousals of which the sleeping individual is unaware. The simultaneous recording of the EEG, the electrooculogram (EOG), and the electromyogram (EMG) are the accepted standard measures of sleep and waking and together these measures are termed polysomnography (PSG).4
1.2 Electrophysiology of Sleep: Polysomnography
Behind the closed eyes and relative behavioral quiescence of sleep is an active, complex, and highly organized process composed of two distinct brain states: nonrapid eye movement (NREM) sleep and rapid eye movement (REM) sleep. As will be seen below this distinction goes much beyond the presence or absence of eye movements for which these two states are named. Fig. 1.1 depicts PSGs of wake NREM and REM sleep.


We describe the characteristics of a PSG (i.e., EEG, EOG, and EMG) of sleep–wake in detail. In contrast to the low voltage (10–30 µV) and fast frequency (16–35 Hz) of activated wakefulness, the cortical EEG (C3/4-A1/2) of relaxed, eyes-closed wakefulness is characterized by increased voltage (20–40 µV) and an 8–12 Hz frequency. During the transition to sleep, sometimes called drowsy sleep or transitional sleep, the EEG frequency becomes mixed while the voltage remains at the level of relaxed wakefulness. In NREM sleep EEG voltage is further increased and frequency is further slowed. When arousal threshold is highest, the EEG of NREM sleep has a 0.5–2.5 Hz frequency with voltages of 75 µV and higher, which is termed slow wave sleep (SWS). The EMG, highest in wakefulness, is gradually reduced during NREM sleep, although limb and body movements occur periodically during NREM and there still is voluntary control of musculature.
The EOGs of wakefulness reveal REMs, which during the transition to NREM sleep (i.e., drowsy sleep) become slow and rolling. Importantly, the rolling eye movements mark the onset of the functional blindness that all humans experience during sleep. This contributes to the dangers of drowsy driving. The EOG becomes quiescent during NREM SWS. After the first 90–120 minutes of NREM sleep the healthy normal person enters REM sleep.
The EOG of REM sleep, for which this sleep state is named, is characterized by rapid conjugate eye movements. The cortical EEG of REM reverts to the low voltage, mixed frequency pattern of drowsy sleep. The second defining characteristic of REM sleep is its skeletal muscle atonia, which is reflected in the EMG achieving its lowest level of the 24-hour period. The skeletal muscle atonia of REM sleep occurs through a process of postsynaptic inhibition of motor neurons at the dorsal horn of the spinal cord. Another important feature of REM sleep is its tonic and phasic components. The tonic components of REM sleep are the persistent muscle atonia and the desynchronized EEG. The phasic components are intermittent and include bursts of eye movements occurring against a background of EOG quiescence. Coupled with the eye movement bursts are muscle twitches, typically involving peripheral muscles. These twitches are superimposed on the tonic muscle atonia of REM and probably reflect sympathetic drive breaking through the postsynaptic motor inhibition (see Section 1.3.1).
Fig. 1.2 illustrates the progression of sleep stages in a healthy young adult across an 8-hour sleep period. NREM and REM sleep alternate in 90- to 120-minute cycles with the predominance of NREM SWS occurring in the first 4 hours of the night and REM sleep occurring in the last 4 hours.5 Of note across the night the duration of NREM SWS episodes diminish, while the duration of REM sleep episodes increase. This differential distribution of sleep stages across the night is associated with the unique sleep state–specific physiological and cognitive function changes we will now describe.
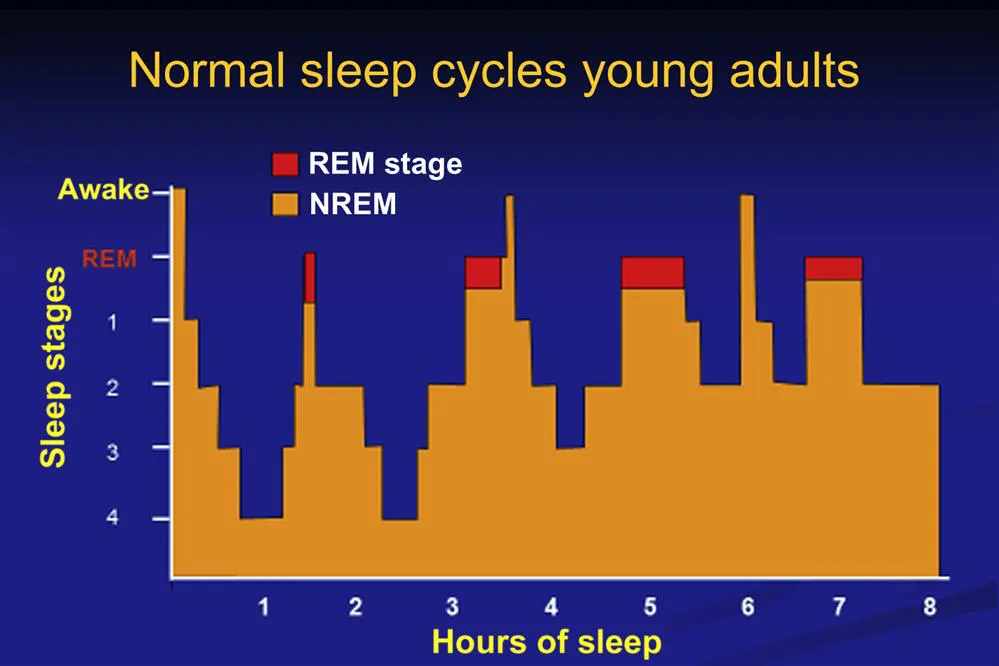
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- List of Contributors
- Introduction
- Chapter 1. The Sleep–Wake Cycle: An Overview
- Chapter 2. Electrophysiological Correlates of the Sleep–Wake Cycle
- Chapter 3. Physiological Mechanisms for the Control of Waking
- Chapter 4. Neurochemistry and Pharmacology of Sleep
- Chapter 5. The Role of Neuroglobin in Brain Function and Sleep–Wake Cycle
- Chapter 6. Overview of Sleep Disorders
- Chapter 7. Obesity and Sleep Disturbances
- Chapter 8. Sleep Disorders and Genes
- Chapter 9. Alcoholism and Sleep
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access The Behavioral, Molecular, Pharmacological, and Clinical Basis of the Sleep-Wake Cycle by Eric Murillo-Rodriguez in PDF and/or ePUB format, as well as other popular books in Medicine & Physiology. We have over 1.5 million books available in our catalogue for you to explore.