
eBook - ePub
Aging and Health - A Systems Biology Perspective
- 206 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Aging and Health - A Systems Biology Perspective
About this book
Aging is a major risk factor for chronic diseases, which in turn can provide information about the aging of a biological system. This publication serves as an introduction to systems biology and its application to biological aging. Key pathways and processes that impinge on aging are reviewed, and how they contribute to health and disease during aging is discussed. The evolution of this situation is analyzed, and the consequences for the study of genetic effects on aging are presented. Epigenetic programming of aging, as a continuation of development, creates an interface between the genome and the environment. New research into the gut microbiome describes how this interface may operate in practice with marked consequences for a variety of disorders. This analysis is bolstered by a view of the aging organism as a whole, with conclusions about the mechanisms underlying resilience of the organism to change, and is expanded with a discussion of circadian rhythms in aging. Finally, the book presents an outlook for the development of interventions to delay or to reverse the features of aging. The publication is recommended to students, researchers as well as professionals dealing with public health and public policy related to an aging society.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
Genetics in MedicineYashin AI, Jazwinski SM (eds): Aging and Health - A Systems Biology Perspective.
Interdiscipl Top Gerontol. Basel, Karger, 2015, vol 40, pp 177-188 (DOI: 10.1159/000364982)
Interdiscipl Top Gerontol. Basel, Karger, 2015, vol 40, pp 177-188 (DOI: 10.1159/000364982)
______________________
Conservative Growth Hormone/IGF-1 and mTOR Signaling Pathways as a Target for Aging and Cancer Prevention: Do We Really Have an Antiaging Drug?
Vladimir N. Anisimov
Department of Carcinogenesis and Oncogerontology, N.N. Petrov Research Institute of Oncology, St. Petersburg, Russia
______________________
Abstract
Inactivation of the GH/insulin/IGF-1 signaling molecules corresponding genes as well as the inactivation of serine/threonine protein kinase mTOR increases life span in nematodes, fruit flies and mice. Evidence has emerged that antidiabetic biguanides and rapamycin are promising candidates for pharmacological interventions leading to both life span extension and prevention of cancer. The available data on the relationship of two fundamental processes-aging and carcinogenesis-have been suggested to be a basis for understanding these two-side effects of biguanides and rapamycin.
© 2015 S. Karger AG, Basel
There are nine tentative hallmarks of aging in mammals, which may represent common denominators of aging in different organisms: genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered cell-to-cell communication [1]. At the same time, there is also sufficient similarity in the patterns of changes observed during normal aging and the process of carcinogenesis (table 1) [2]. As can be seen in figure 1, DNA damage induced by environmental and endogenous carcinogenic factors [reactive oxygen species, ionizing radiation, ultraviolet, constant illumination (light at night), some diets, oncogenes, etc.] may lead to cellular senescence or cellular lesions which could be deleted by apoptosis. The same agents can induce damage which is followed by neoplastic transformation, thus leading to cancer [2, 3]. During the last decade, the intensive search for antiaging remedies has led to the conclusion that both insulin/IGF-1 signaling (IIS) and nutrient response pathways defined by the mTOR protein kinase pathways control aging and age-asso-ciated pathology in worms, insects and mammals [4]. In each of these organisms, genetic downregulation or interruption of this signaling pathway can lead to major extension of longevity. There are two functionally distinct mTOR complexes called mTORC1 and mTORC2. mTORC1 is activated by insulin and related growth factors through phosphatidylinositol-3-OH kinase and AKT kinase signaling and repressed by AMP-activated protein kinase, a key sensor of cellular energy status [4]. The mTORC1 is involved in promoting messenger RNA translation and protein synthesis through ribosomal protein S6 kinases (S6Ks) and 4E-BP protein, which in the hypophosphorylated form acts as a negative regulator of the cap-binding protein eIF4E. mTORC1 also stimulates lipid biosynthesis, inhibits autophagy, and through hypoxic response transcription factor HIF-1α regulates mitochondrial function and glucose metabolism. Rapamycin suppresses mTORC1 and indirectly mTORC2, which leads to metabolic lesions like glucose intolerance and abnormal lipid profile [4]. The phosphorylation of S6K1 at T389 by TORC1 is susceptible to rapamycin. The life span of S6K1 -deficient female mice increased by 19% in comparison to the wild-type controls without any effect on the incidence of tumor development [5]. It is worth noting that there was no significant effect of the protein knockout on the life span of male mice. These data suggest that S6K1 is involved in mammalian life span regulation downstream of TORC1. Taking into consideration the negative effect of rapamycin on glucose tolerance and liver insulin sensitivity [5], Lamming et al. [6] studied the effect of mTORC1 and mTORC2 regulator gene modification on the life span of mice. There was no increase in life span in either female or male mtor+/-, Raptor+/-, mlst8+/- or mtor+/- Raptor+/- mice. However, female mtor+/- mlst8+/- mice lived longer by 14.4% in comparison to wild-type mice. The longevity of male mtor+/- mlst8+/- mice was unaffected. Female mtor+/- mlst8+/- mice were not calorie restricted through reduced food intake or increased energy expenditure, and had normal body weights and levels of activity consistent with the phenotypic effects. mtor+/- mlst8+/- mice exhibited an approximately 30-60% reduction in the abundance of hepatic mTOR, Raptor, mLST8, and Rictor, whereas the expression of mTOR complex subunits was less affected in Raptor+/- and mtor+/- Raptor+/- heterozygotes. The authors believe that suppression mTORC1 signaling is sufficient for life span prolongation independent of changes in glucose homeostasis.
Table 1. Changes developing in organism during natural aging and carcinogenesis: effects of geroprotectors

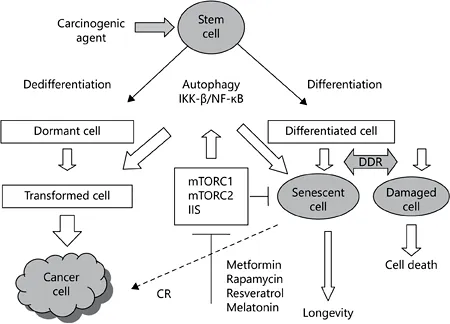
Fig. 1. Relationship between aging and carcinogenesis: the key role of IIS and mTOR signaling. DNA damage induced by environmental and endogenous factors (reactive oxygen species, ionizing radiation, ultraviolet, constant illumination, some diets, oncogenes, etc.) may lead to cellular senescence or cellular lesions which could be deleted by apoptosis. The same agents can induce damage followed by neoplastic transformation, thus leading to cancer. Metformin, rapamycin, and some other compounds with mTOR and US-inhibitory potential (resveratrol, melatonin) are able to modify both aging and carcinogenesis. DDR = DNA damage response.
Calorie restriction (CR) is the only known intervention in mammals that has been consistently shown to increase life span, reduce incidence and retard the onset of age-related diseases, including cancer and diabetes. CR has also been shown to increase resistance to stress and toxicity, and maintain youthful levels of function and vitality in laboratory mammals at advanced chronological age [7]. CR in rhesus monkeys have produced physiological responses strikingly similar to those observed in rodents and delayed the onset of age-related diseases, but effects on longevity were not consistent [8]. Data from these studies indicate that long-term CR reduces morbidity and mortality in primates, and thus may exert beneficial ‘antiaging’ effects in humans. Although understanding the role of GH and IIS in the control of human aging is incomplete and somewhat controversial, available data indicate that dietary prevention of excessive IGF-1 and insulin secretion and using diet and exercise to enhance insulin sensitivity may represent the most hopeful approaches to cancer prevention and to extending human health span and life span [2-4]. Metformin, rapamycin, and some other compounds with mTOR and IIS-inhibitory potential (resveratrol, melatonin) are able to modify both aging and carcinogenesis. This chapter focuses on the effects of biguanides and rapamycin, whereas data on resveratrol and melatonin will be discussed elsewhere.
Antidiabetic Biguanides as Geroprotectors
The concept of CR mimetics is now being intensively explored [3]. The antidiabetic biguanides, phenformin, buformin and metformin were observed to reduce hyperglycemia, improve glucose utilization, reduce free fatty acid utilization, gluconeogenesis, serum lipids, insulin, and IGF-1, reduce body weight and decrease metabolic immunodepression both in humans and rodents [3]. The results of studies on the effect of antidiabetic biguanides on the life span in mice and rats are summarized in table 2.
Table 2. Summary on effects of biguanides and rapamycin on life span and spontaneous carcinogenesis in rodents
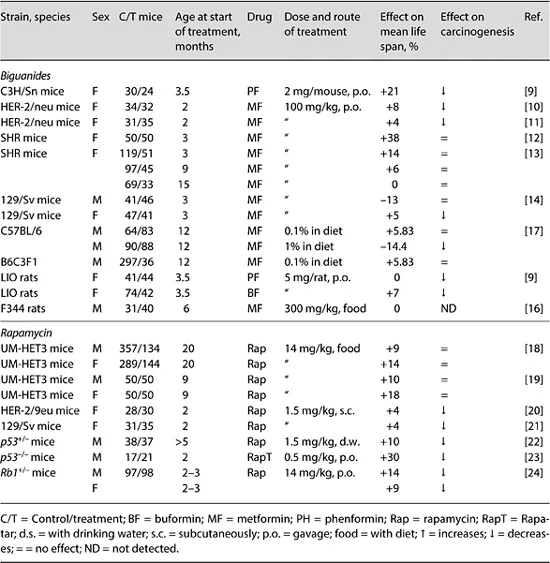
The treatment with phenformin prolonged the mean life span of female C3H/Sn mice by 21% (p < 0.05) and the maximum life span by 26% in comparison with the controls [9]. At the time of death of the last mice in the control group, 42% of phenformin-treated mice were alive. The treatment with phenformin failed to influence the mean life span of female LIO rats; however, it increased the maximum life span by 3 months (10%) in comparison with the controls [9]. The treatment with phenformin slightly decreased the body weight of rats and delayed age-related switching off of estrous function. Similar findings were observed in female rats exposed to another bi-guanide, buformin [9]. Administration of metformin to female transgenic HER-2/ neu mice did not change the body weight or temperature; it slowed down the agerelated rise in blood glucose and triglyceride levels, decreased the serum level of cholesterol and β-lipoproteins, delayed the age-related irregularity in estrous cycle, extended the mean life span by 4-8% and the maximum life span by 1 month in comparison with the control animals [10, 11]. It is well known that excess of body weight and obesity leads to development of metabolic syndrome, type 2 diabetes, premature switching off of reproductive function and risk of cancer [2, 9]. The mechanism behind the geroprotective effect of metformin could reside in its ability to lower body weight.
Metformin increased the mean life span of the last 10% of survivors by 20.8% and the maximum life span by 2.8 months (10.3%) in female SHR mice in comparison with control mice [12]. The decreased body temperature and postponed age-related switching off of estrous function were observed in the group of metformin-treated mice. In another set of experiments, female SHR mice were given metformin from the age of 3, 9 or 15 months [13]. Metformin started at the age of 3 months increased the mean life span by 14% and maximum life span by 1 month, whereas the treatment started at the age of 9 months by 6%; metformin started at the age of 15 months did not affect life span.
The treatment with metformin slightly modified the food consumption but failed to influence the dynamics of body weight. Metformin decreased by 13.4% the mean life span of male 129/Sv mice and slightly increased the mean life span of females (by 4.4%). Metformin failed to influence spontaneous tumor incidence in male 129/Sv mice, decreased 3.5-fold the incidence of malignant neoplasms in female mice, while somewhat stimulated formation of benign vascular tumors [14].
Significant prolongation (by 20.1%) of the survival time was observed in male (but not female) transgenic mice with Huntington's disease without affecting fasting blood glucose levels. Increasing the dose of the drug did not improve the survival of mice [15]. In the NIA study [16], 6-month-old male F344 rats were randomized to one of four diets: control, CR, diet supplemented with metformin and standard diet pair fed to metformin. There were no significant differences in the mean life...
Table of contents
- Cover Page
- Front Matter
- Introduction to the Theory of Aging Networks
- Applications to Aging Networks
- Computational Systems Biology for Aging Research
- How Does the Body Know How Old It Is? Introducing the Epigenetic Clock Hypothesis
- The Great Evolutionary Divide: Two Genomic Systems Biologies of Aging
- Development and Aging: Two Opposite but Complementary Phenomena
- Aging as a Process of Deficit Accumulation: Its Utility and Origin
- Low-Grade Systemic Inflammation Connects Aging, Metabolic Syndrome and Cardiovascular Disease
- Modulating mTOR in Aging and Health
- Melatonin and Circadian Oscillators in Aging - A Dynamic Approach to the Multiply Connected Players
- Diet-Microbiota-Health Interactions in Older Subjects: Implications for Healthy Aging
- Systems Biology Approaches in Aging Research
- Conservative Growth Hormone/IGF-1 and mTOR Signaling Pathways as a Target for Aging and Cancer Prevention: Do We Really Have an Antiaging Drug?
- Author Index
- Subject Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Aging and Health - A Systems Biology Perspective by A. I. Yashin,S. M. Jazwinski,A.I., Yashin,S.M., Jazwinski, Tamàs Fülöp,Tamàs, Fülöp in PDF and/or ePUB format, as well as other popular books in Medicine & Genetics in Medicine. We have over 1.5 million books available in our catalogue for you to explore.