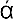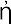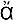![]()
CHAPTER I
The Dawn of Physics
IT IS VERY difficult to trace the origin of the science of physics, just as it is difficult to trace the origin of many great rivers. A few tiny springs bubbling from under the green foliage of tropical vegetation, or trickling out from beneath the moss-covered rocks in the barren northern country; a few small brooks and creeks running gaily down the mountain slopes and uniting to form rivulets which in turn unite to form streams big enough to deserve the name “river.” The rivers grow broader and broader, being strengthened by numerous tributaries, and finally develop into mighty flows—be it the Mississippi or the Volga, the Nile or the Amazon—carrying their waters into the ocean.
The springs which gave birth to the great river of physical science were scattered all over the surface of the earth inhabited by
Homo sapiens, i.e., thinking man. It seems, however, that most of them were concentrated on the southern tip of the Balkan peninsula inhabited by the people now known as “ancient Greeks”; or, at least, it seems so to us who inherited the culture of these early “intellectuals.” It is interesting to notice that whereas other ancient nations, such as Babylonia and Egypt, contributed a great deal to the early development of mathematics and astronomy, they were completely sterile in the development of physics. The possible explanation of that deficiency as compared with Greek science is that Babylonian and Egyptian gods lived high up among the stars, while the gods of the ancient Greeks lived at the elevation of only about 10,000 feet, at the top of Mount Olympus, and so were much closer to down-to-earth problems. According to a legend the term “magnetism” originated from the name of a Greek shepherd, M
γνης, who was surprised to notice that the tip of his iron-shod staff was attracted by a stone (magnetic iron ore) lying along the roadside. Similarly the term “electricity” comes from the Greek word
λεκτρον, for amber, maybe because some other Hellenic shepherd, trying to polish a piece of amber by rubbing it against the coat of one of his sheep, noticed that it acquired a mysterious property to attract loose pieces of wood.
PYTHAGOREAN LAW OF STRINGS
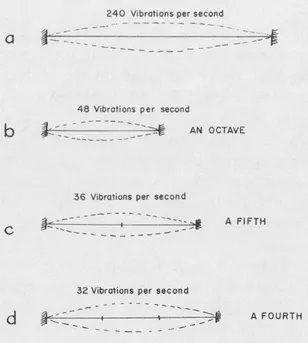
Whereas these legendary discoveries would hardly stand their ground in any legal priority suit, the discovery of the Greek philosopher Pythagoras, who lived in the middle of the 6th century B.C., is well documented. Persuaded that the world is governed by number, he was investigating the relation between the lengths of the strings in the musical instruments which produce harmonic combinations of sound. For this purpose he used the so-called monochord, i.e., a single string which can be varied in length and subjected to different tensions caused by a suspended weight. Using the same weight, and varying the length of the string, he found that the pairs of harmonic tones were obtained when the string’s lengths stood in simple numerical relations. The length ratio 2 : 1 corresponded to what was known as “octave,” the ratio 3 : 2 to a “fifth,” and the ratio 4 : 3 to a “fourth.” This discovery was probably the first mathematical formulation of a physical law, and can be well considered as the first step in the development of what is now known as theoretical physics. In modern physical terminology we may reformulate Pythagoras’ discovery by saying that the frequency, i.e., the number of vibrations per second, of a given string subjected to a given tension is inversely proportional to its length. Thus, if the second string (Fig. I-1b) is half as long as the first one (Fig. I-1a), its vibration frequency will be twice as high. If the two string lengths stand in the ratio of 3 : 2 or 4 : 3, their vibration frequencies will stand in the ratios 2 : 3 or 3 : 4 (Fig. I-1c,d). Since the part of the human brain which receives the nerve signals from the ear is built in such a way that simple frequency ratios, as 3 : 4, give “pleasure” whereas the complex ones, as 137 : 171, “displeasure” (it is for the brain physiologists of the future to explain that fact!), the length of strings giving perfect accord must stand in simple numerical ratios.
FIG. I—1.
Pythagorean law of strings.
Pythagoras tried to go one step further by suggesting that, since the motion of planets “must be harmonious,” their distances from the earth must stand in the same ratios as the lengths of the strings (under equal tension) which produce the seven basic tones produced by the lyre, the national Greek musical instrument. This proposal was probably the first example of what is now often called “pathological physical theory.”
DEMOCRITUS, THE ATOMIST
Another important physical theory, which in modern terminology could be called “a theory without any experimental foundation” but which turned out to be a “dream that came true,” was proposed by another ancient Greek philosopher, Democritus, who lived, thought, and taught around the year 400 B.C. Democritus conceived the idea that all material bodies are aggregates of innumerable particles so small as to be unnoticeable by the human eye. He called these particles
atoms, or indivisibles (
τομος) in Greek, because he believed that they represented the ultimate stage of the division of material bodies into smaller and smaller parts. He believed that there are four different kinds of atoms: the atoms of stone, dry and heavy; the atoms of water, heavy and wet; the atoms of air, cold and light; and the atoms of fire, slippery and hot. By a combination of these four different kinds of atoms all known materials were supposed to be made. The soil was a combination of stone and water atoms. A plant growing from the soil under the influence of sun rays consisted of the stone and water atoms from the soil and the fire atoms from the sun. That is why dry wood logs which have lost all their water atoms would burn, liberating fire atoms (flame) and leaving behind stone atoms (ashes). When certain kinds of stones (metallic ores) were put in the flame, stone atoms united with the fire atoms producing the substances known as metals. Cheap metals like iron contained very small amounts of fire atoms and therefore looked rather dull. Gold had the maximum amount of fire atoms and thus was brilliant and valuable. Consequently, if one could add more fire atoms to the plain iron, one should be able to make precious gold!
A student who would tell all that in his introductory chemistry examination would certainly get an F grade. But, although these particular examples of the nature of chemical transformation were certainly wrong, the fundamental idea of obtaining almost unlimited numbers of different substances by a combination of only a few basic chemical elements was undoubtedly correct and now represents the foundation of chemistry today. It took, however, twenty-two centuries from the time of Democritus to the time of Dalton to make things right.
ARISTOTELIAN PHILOSOPHY
One of the giants of the ancient Greek world was a man named Aristotle, who became famous on two counts: first, because he was a real genius; second, because he was a tutor and later a protégé of Alexander the Great of Macedonia. He was born in 384 B.C. in the Greek colonial town Stagira on the Aegean Sea to a former court physician of the Macedonian royal family. At the age of 17 h...