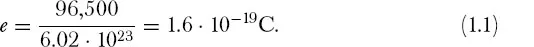![]()
Part One The Birth of a New Physics
Classical physics has its origins in the 17th century, with Galileo’s1 experiments on falling bodies, Kepler’s2 calculations of the planetary orbits and Newton’s3 postulates and mathematical laws of motion. During the following two centuries, the Newtonian system of mechanics, together with its theory of gravitation, dominated scientific thinking; its achievements were unprecedented. Perhaps its most impressive feat was predicting the existence of the planet Neptune. Calculations based on Newtonian mechanics indicated that the slight perturbations that had been observed in the orbit of the planet Uranus could be accounted for by the presence of an additional planet in the solar system, one previously unknown. In 1846, this new planet, Neptune, was found at the exact position in space indicated by the calculations.4
By the middle of the 19th century, the kinetic theory of matter and the science of thermodynamics had between them solved the ancient mystery of the nature of heat and had provided an understanding for the ‘arrow of time’ inherent in the workings of nature. Classical physics reached its zenith in the second half of the 19th century with the publication of Maxwell’s5 theory of electromagnetism and the discovery of the electromagnetic waves whose existence it had predicted. This theory summarised and unified everything that was known at the time about electrical and magnetic phenomena and provided the first comprehensive conceptual basis for the science of optics.
However, towards the end of the 19th century, it became clear that there were important physical phenomena for which classical physics had no satisfactory explanation. The electron, X-rays and radioactivity, all of which were discovered within a few years of each other in the last decade of the century, were beyond the competence of classical physics to explain. Moreover, there were even instances where the hypotheses and laws of classical physics were found to be totally incompatible with the results of experiments in more conventional fields, such as studies of the speed of light relative to different observers and the emission and absorption of heat radiation. It became apparent that despite its great achievements, some of the most fundamental principles underlying classical physics were incorrect. Clearly, fresh ideas and theories were needed.
At the beginning of the 20th century, three new hypotheses were put forward that changed the face of physics. They were:
1. The theory of relativity.
2. Quantum theory.
3. The nuclear model of the atom.
This triad laid the foundations of modern physics. They, and the effects derived from them, are the subject of this book.
The new theories of modern physics not only resolved the problems left unanswered by classical physics but extended the reach of the physical sciences into previously unknown fields. In general, the familiar and well-established laws of classical physics remain valid for dealing with phenomena that occur on a ‘normal’ scale, in the human sense of the term. However, when dealing with phenomena occurring on the cosmic scale on the one hand, and on the atomic scale on the other, only the more comprehensive laws of modern physics can be employed.
Part One of our study of 20th century physics opens with the discovery of the electron, the first elementary particle to be identified; no other single discovery has ever led to the understanding of so many physical phenomena previously thought to be unrelated. This is followed by a short review of the Maxwellian theory of electromagnetism and of the empirical and theoretical advances that led to the general acceptance of the reality of atoms. These three developments provided the background for the scientific revolution ushered in by Einstein’s theory of special relativity and Planck’s quantum hypothesis during the first decade of the new century. Part One concludes with a brief account of the theory of general relativity.
![]()
Chapter 1.1
The Electron
When first investigated in the 18th and 19th centuries, electrical and magnetic phenomena were generally construed in terms of æthereal fluids, as were those associated with heat and light. These fluids were thought to comprise minute mutually repelling particles. Thus, heat was either thought to be vibrations in the fluid caloric or an accumulation of this fluid in the interstices of materials. Light was either a flux of particles emitted at high speed from luminous bodies or the vibrations of a ubiquitous fluid æther. Electric fluids — electricity — flowed readily through metals and other conductors but did not penetrate insulators such as paper and glass. Opinion was divided as to whether there was just one electric fluid or two — a positive fluid and a negative fluid.
The possibility that electricity might not be a continuous fluid was first raised in the middle of the 19th century following Faraday’s6 quantitative researches on electrolysis. These showed the existence of a systematic relationship between the amount of electricity passed through an electrolytic cell and the quantity of material that undergoes chemical reaction (electrolysis) in the cell. Thus, the passage of a certain amount of electricity — 96,500 coulombs in modern terms — always liberates a gram-equivalent of substance from the electrolyte, whether it is the metal released at the negative cathode or the non-metal at the positive anode. Putting aside his misgivings about atomism, Faraday recognised that this suggested electricity might be atomic in nature and that a natural indivisible unit of electricity exists. In 1891 Stoney7 suggested that this natural unit of electricity be called an electron.
On this view, every ion carries an integer multiple of this natural unit. For example, a silver ion, Ag+, carries a single natural unit of positive charge; a typical copper ion, Cu++, carries two such units. Given that a gram-equivalent of monovalent ions comprises a mole, the magnitude of the natural unit of electricity, e, can be calculated by dividing the 96,500 coulombs by Avogadro’s number, 6.02 · 1023:
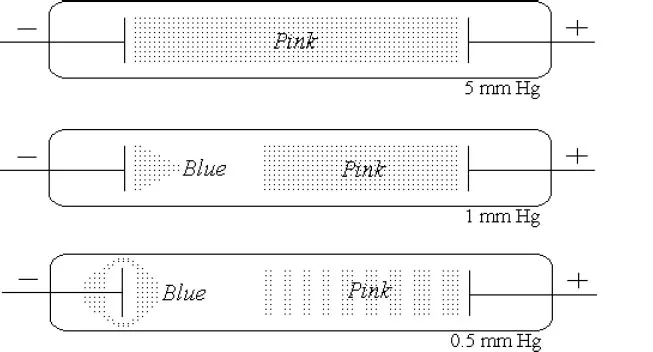
The term electron is now used to designate the elementary particle that carries the natural unit of negative charge and which was first identified towards the end of the 19th century in experiments on the conduction of electricity through gases at very low pressures. At atmospheric pressure, gases do not usually conduct electricity. However, at reduced pressures of 0.5mmHg to 10mmHg and with applied potentials of several thousand volts, they can be made to pass a current. These greatly reduced gas pressures were first achieved at the end of the 19th century following the advances made at that time in vacuum pump technology. The gases were contained in narrow glass tubes, called discharge tubes, into which suitable electrodes had been inserted. The passage of the current is accompanied by the appearance of striking colours in the tubes (Fig. 1.1).
Fig. 1.1. The conduction of electricity through air at low pressures in a discharge tube. The colours result from the excitation and ionisation of the atoms of the gas in the discharge tube. The bands and the coloured and dark regions arise from the variations in the electric field strength throughout the tube.
At still lower pressures, ∼0...