![]()
Chapter
1
FOOD ALLERGY
I like this passage for a couple of reasons. One is that it shows how basic the impulse to use vegetation as medicine is. TCM has thousands of years of recorded history, and clearly it did not begin with the beginnings of civilization, and it did not even start with humanity. The other is that it shows how fundamental the battle is between animals and parasites, nature’s extortionists. Chimps knew these little things were bad for them even if they could not see them or imagine them. They knew they were sick and they tried to do something about it. Readers of my other book about Dr. Li, Food Allergies: Traditional Chinese Medicine, Western Science, and the Search for a Cure know that her explorations for a food allergy treatment began with something called Wu Mei Wan, an old remedy for these worms that feed off people, weakening them without killing them, except for children who may not have time to develop their own defenses. In a body battling parasites, IgE and eosinophils are powerful defenses, but in our more hygienic society, they pick on the wrong targets and become our enemies.
It is a case of mistaken identity. Certain peanut proteins are very similar to those found in helminths, commonly known as parasitic worms. Likewise proteins found in dust mites and cockroaches, which are potent environmental allergens, are similar to some found in shrimp and lobster — major food allergens. The great moment of revelation came when Dr. Li was studying food allergy reactions and it dawned on her that it sounded like parasites so she dipped into the TCM trove Classic Formulas and Strategies and came up with Wu Mei Wan. As readers will also recall, in trying to decide how to deliver this medicine to patients, Dr. Li’s colleagues tasted it in the form of brewed tea, as it would be taken in China. They unanimously agreed that they could not stand the taste.
When I finished writing the other book, the Phase II trial of Food Allergy Herbal Formula-2 (FAHF-2), the successor to Wu Mei Wan, was still in a state of equipoise. That is, the work was done but the data had not been compiled or interpreted or published.
To recap, this double-blind, randomized, placebo-controlled study enrolled 68 subjects, 12–45 years of age, with allergies to peanut, tree nut, sesame, fish, and/or shellfish. Food allergies for all study participants were confirmed at the outset by a stringent double-blind, placebo controlled food challenge (DBPCFC). Forty-six received FAHF-2 and 22 placebo. After six months of therapy, subjects underwent another DBPCFC. For those who demonstrated increases in eliciting dose, a repeat challenge was performed three months after stopping therapy.1
The results were disappointing to hopeful food allergy parents. There was no clinical breakthrough from this study. However, the laboratory results do suggest positive immune modulation is possible with FAHF-2. Immunological studies of the subjects’ PBMCs (peripheral blood mononu-clear cells) drawn at baseline and incubated with FAHF-2 and food allergen produced significantly less IL-5, greater IL-10 and increased numbers of Tregs than untreated cells — a shift away from the allergic Th2-dominant cytokines.
While the subjects’ cells tested in the lab outside of their bodies showed benefit with FAHF-2 treatment, what happened with the subjects themselves? It turns out that 44% had trouble sticking with the protocol. The real numbers of non-compliant subjects may have been higher, but the count was taken according to how many pills were returned at the conclusion of the study period. Even more pills may still be sitting in medicine cabinets in subjects’ homes.
As with most studies, in retrospect, there seem to have been holes in the design. This do not mean that FAHF-2 cannot work in humans. It was asking a great deal for nearly 70 individuals ranging in age from 12, where they would have at least been subject to some element of maternal supervision, through college age and into middle age to swallow 10 pills at each of three meals during the day, i.e., 30 pills per day. They had to remember to pack their noon dose to be taken at lunch. Strict medication regimens are best practiced under the watchful eye of mothers. Moreover, six months is a very short period of time to effect a wholesale renovation of the immune system. In her clinical practice, Dr. Li projects 2–4 years duration for treatment, so six months is a drop in the bucket. According to Dr. Li,
In the earlier book, I described the creation of a refined form of FAHF-2, which employs butanol (B-FAHF-2) to separate the active ingredients from the inert ones. The FDA has approved use of B-FAHF-2 for further experiments without having to go back to square one with murine models and early-phase human trials.
The test tube indicators are encouraging. Dr. Li says,
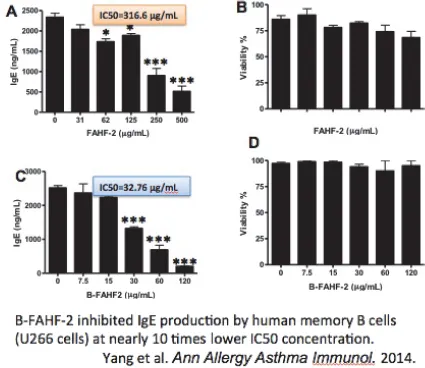
B-FAHF-2 More Potently Inhibited IgE
Production than FAHF-2 in vitro
One of the things that make this avenue of research so exciting is that it holds out the possibility of making long-term changes in the underlying immune mechanisms. Allergic responses are “amnestic” — remembered. A patient can go years with no exposure to an allergen and then respond violently. It is this tendency that makes “avoidance” a dirty word in the food allergy community.
As Dr. Li says,
This is one of the few subjects that lead her to employ military metaphors. She likens it to field commanders who are getting bad orders — good soldiers implementing wrong tactics.
Lack of cytotoxicity is a crucial element. In addition, there is no suppression of innate immunity, as there is with steroids, which have the side effect of leaving patients more vulnerable to bacterial or viral infection.
This is unique finding for a natural product. They are not killing the cells. The cells can still be there, but just not produce the IgE. Also, sometimes with immunomodulation, the IgE levels are still there, only they are dormant. Then when the body needs them, they can reactivate, when you encounter an intestinal parasite, for example.
The parasite connection is fundamental.
Dr. Li listens to her patients, no matter how young.
A new study is planned for B-FAHF-2 to be administered over 26 months using six pills per day instead of the 30 pills over six months. This one will be evenly divided, 17 each of medicine and placebo. While smaller, the study should produce robust data with easier compliance. This is just one of several studies using the refined food allergy formula planned over the next several years in support of the eventual aim of having a medicine that can be prescribed without accompanying knowledge of TCM. B-FAHF-2 is an FDA-registered investigational new drug (IND).
Many mothers ask if the goal of Dr. Li’s food allergy treatment is to “bite proof” patients, i.e., give them protection against small inadvertent ingestion of their allergens, desensitize them to their allergens, or to cure their allergies. The goal has always been to “reeducate” the immune system to fight the things that need fighting and ignore the ones that do not with enough capacity to fight natural enemies like parasites — a cure.
Of course those mice were all fed through a tube.
What will the biochemistry show?