eBook - ePub
Medical Biochemistry
Antonio Blanco, Gustavo Blanco
This is a test
- 826 pages
- English
- ePUB (adapté aux mobiles)
- Disponible sur iOS et Android
eBook - ePub
Medical Biochemistry
Antonio Blanco, Gustavo Blanco
Détails du livre
Aperçu du livre
Table des matières
Citations
À propos de ce livre
Medical Biochemistry is supported by over forty years of teaching experience, providing coverage of basic biochemical concepts, including the structure and physical and chemical properties of hydrocarbons, lipids, proteins, and nucleotides in a straightforward and easy to comprehend language. The book develops these concepts into the more complex aspects of biochemistry using a systems approach, dedicating chapters to the integral study of biological phenomena, including particular aspects of metabolism in some organs and tissues, and the biochemical bases of endocrinology, immunity, vitamins, hemostasis, and apoptosis.
- Integrates basic biochemistry principles with molecular biology and molecular physiology
- Provides translational relevance to basic biochemical concepts though medical and physiological examples
- Utilizes a systems approach to understanding biological phenomena
Foire aux questions
Comment puis-je résilier mon abonnement ?
Il vous suffit de vous rendre dans la section compte dans paramètres et de cliquer sur « Résilier l’abonnement ». C’est aussi simple que cela ! Une fois que vous aurez résilié votre abonnement, il restera actif pour le reste de la période pour laquelle vous avez payé. Découvrez-en plus ici.
Puis-je / comment puis-je télécharger des livres ?
Pour le moment, tous nos livres en format ePub adaptés aux mobiles peuvent être téléchargés via l’application. La plupart de nos PDF sont également disponibles en téléchargement et les autres seront téléchargeables très prochainement. Découvrez-en plus ici.
Quelle est la différence entre les formules tarifaires ?
Les deux abonnements vous donnent un accès complet à la bibliothèque et à toutes les fonctionnalités de Perlego. Les seules différences sont les tarifs ainsi que la période d’abonnement : avec l’abonnement annuel, vous économiserez environ 30 % par rapport à 12 mois d’abonnement mensuel.
Qu’est-ce que Perlego ?
Nous sommes un service d’abonnement à des ouvrages universitaires en ligne, où vous pouvez accéder à toute une bibliothèque pour un prix inférieur à celui d’un seul livre par mois. Avec plus d’un million de livres sur plus de 1 000 sujets, nous avons ce qu’il vous faut ! Découvrez-en plus ici.
Prenez-vous en charge la synthèse vocale ?
Recherchez le symbole Écouter sur votre prochain livre pour voir si vous pouvez l’écouter. L’outil Écouter lit le texte à haute voix pour vous, en surlignant le passage qui est en cours de lecture. Vous pouvez le mettre sur pause, l’accélérer ou le ralentir. Découvrez-en plus ici.
Est-ce que Medical Biochemistry est un PDF/ePUB en ligne ?
Oui, vous pouvez accéder à Medical Biochemistry par Antonio Blanco, Gustavo Blanco en format PDF et/ou ePUB ainsi qu’à d’autres livres populaires dans Biological Sciences et Biochemistry. Nous disposons de plus d’un million d’ouvrages à découvrir dans notre catalogue.
Informations
Sujet
Biological SciencesSous-sujet
BiochemistryChapter 1
Chemical Composition of Living Beings
Abstract
Biogenic elements are essential components of living organisms. They include: (1) primary elements (O, C, H, N, Ca, and P), which comprise ∼98% of the total body mass of an adult human and participate in the composition of essential body molecules; (2) secondary elements (Na, K, Cl, S, Mg), which exist as salts and inorganic ions and Fe, which is part of important molecules, as hemoglobin; and (3) trace elements or oligoelements (I, Cu, Zn, Mo, Se, and Co), which are present in very scarce amounts, but are key to body function. The biogenic elements combine to form biological compounds. These include inorganic and organic substances. Among the inorganic compounds is water, the solvent present in body fluids and tissues. It comprises 65% of the total body weight of an adult individual. Other inorganic compounds are nonsoluble, such as calcium phosphate, which is an essential component of bone. The organic biological compounds include proteins, carbohydrates, lipids, and nucleic acids. Others, such as vitamins, hormones, and pigments have carbon as a key component and perform essential roles.
Keywords
biogenic elements
biological compounds
Biogenic elements
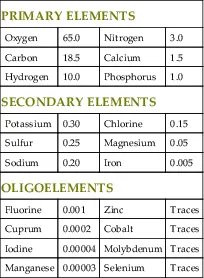
Life emerged on Earth many millions of years after the planet was first formed. Only a small number of elements within the inorganic matter of the Earth’s crust and atmosphere were selected as the building blocks of all living organisms. These basic elements of life are called biogenic elements. Mammals, animals of great complexity, are composed of merely 20 elements, 4 of which (oxygen, carbon, hydrogen, and nitrogen) are the most abundant, comprising approximately 96% of the total body mass (Table 1.1).
Table 1.1
Elements of the Human Body and Their Relative Abundance
| PRIMARY ELEMENTS | |||
| Oxygen | 65.0 | Nitrogen | 3.0 |
| Carbon | 18.5 | Calcium | 1.5 |
| Hydrogen | 10.0 | Phosphorus | 1.0 |
| SECONDARY ELEMENTS | |||
| Potassium | 0.30 | Chlorine | 0.15 |
| Sulfur | 0.25 | Magnesium | 0.05 |
| Sodium | 0.20 | Iron | 0.005 |
| OLIGOELEMENTS | |||
| Fluorine | 0.001 | Zinc | Traces |
| Cuprum | 0.0002 | Cobalt | Traces |
| Iodine | 0.00004 | Molybdenum | Traces |
| Manganese | 0.00003 | Selenium | Traces |
Values are expressed as a percent of total body mass.
All elements of the human body, with the exception of iodine (which has an atomic number of 53), are placed within the first 4 periods of the periodic table and possess atomic numbers lower than 34. Among the four most abundant ones, oxygen has the highest atomic number (8). While oxygen is relatively common on Earth, the other fundamental elements of living organisms are less abundant, suggesting that they have properties, which gave them a selective advantage in becoming the basic units of life. For example, carbon, and not silicon, has been the element around which life developed despite the fact that silicon is widespread and constitutes approximately 21% of the total Earth’s weight.
Carbon belongs to the same group in the periodic table and shares many of the properties of silicon. However, carbon can form more stable chemical bonds, long branched chains, double and triple bonds, covalent bonds with different atoms, and adopts a variety of different spatial conformations. This gives carbon the unique potential to generate a variety of chemical combinations that are essential for the makeup of the molecules of living organisms.
The selection of the other elements that accompany carbon as components of the living matter depends on the size of these atoms and their ability to share electrons in covalent bonds. The smaller atomic size of these elements favors their capacity to establish more stable bonds and stronger molecular interactions.
Taking into consideration their relative amounts, biogenic elements can be classified into three main categories:
1. ...
Table des matières
- Cover
- Title page
- Table of Contents
- Copyright
- Dedication
- About the Authors
- Preface
- Introduction
- Chapter 1: Chemical Composition of Living Beings
- Chapter 2: Water
- Chapter 3: Proteins
- Chapter 4: Carbohydrates
- Chapter 5: Lipids
- Chapter 6: Nucleic Acids
- Chapter 7: Elements of Thermodynamics and Biochemical Kinetics
- Chapter 8: Enzymes
- Chapter 9: Biological Oxidations: Bioenergetics
- Chapter 10: Antioxidants
- Chapter 11: Membranes
- Chapter 12: Digestion - Absorption
- Chapter 13: Metabolism
- Chapter 14: Carbohydrate Metabolism
- Chapter 15: Lipid Metabolism
- Chapter 16: Amino Acid Metabolism
- Chapter 17: Heme Metabolism
- Chapter 18: Purine and Pyrimidine Metabolism
- Chapter 19: Integration and Regulation of Metabolism
- Chapter 20: Metabolism in Some Tissues
- Chapter 21: The Genetic Information (I)
- Chapter 22: The Genetic Information (II)
- Chapter 23: Regulation of Gene Expression
- Chapter 24: Posttranslational Protein Modifications
- Chapter 25: Biochemical Basis of Endocrinology (I) Receptors and Signal Transduction
- Chapter 26: Biochemical Bases of Endocrinology (II) Hormones and Other Chemical Intermediates
- Chapter 27: Vitamins
- Chapter 28: Water and Acid–Base Balance
- Chapter 29: Essential Minerals
- Chapter 30: Molecular Basis of Immunity
- Chapter 31: Hemostasis
- Chapter 32: Apoptosis
- Alphabetic Index
Normes de citation pour Medical Biochemistry
APA 6 Citation
Blanco, A., & Blanco, G. (2017). Medical Biochemistry ([edition unavailable]). Elsevier Science. Retrieved from https://www.perlego.com/book/1810435/medical-biochemistry-pdf (Original work published 2017)
Chicago Citation
Blanco, Antonio, and Gustavo Blanco. (2017) 2017. Medical Biochemistry. [Edition unavailable]. Elsevier Science. https://www.perlego.com/book/1810435/medical-biochemistry-pdf.
Harvard Citation
Blanco, A. and Blanco, G. (2017) Medical Biochemistry. [edition unavailable]. Elsevier Science. Available at: https://www.perlego.com/book/1810435/medical-biochemistry-pdf (Accessed: 15 October 2022).
MLA 7 Citation
Blanco, Antonio, and Gustavo Blanco. Medical Biochemistry. [edition unavailable]. Elsevier Science, 2017. Web. 15 Oct. 2022.