1.1 Introduction
The human race has been interested in colouring materials since prehistoric times typified by the application of natural dyes for furs and textiles. These early dyes were mainly of vegetable origin, with some also of animal origin. Developments extending over many thousands of years led to rather complicated dyeing processes being produced. Among these were woad (natural indigo) which was obtained from the plant Indigofera tinctoria, Tyrian purple which was extracted from the gland of a purple snail and developed by the Phoenicians and Alizarine which was obtained from madder Campeachi wood extract.
The use of picric acid, obtained by Wolfe in 1771 by treating indigo with nitric acid, was subsequently used for dyeing silk yellow, but did not gain any significant attention. In 1856, William H. Perkin succeeded in obtaining a dye he called Mauvine. This was achieved by oxidation of a mixture of aniline bases to produce a violet cationic dye. The brilliant violet hue on silk attracted immediate attention and stimulated other chemists to carry out similar experiments. In this way similar discoveries were achieved; in 1859 Verguin discovered fuchsine, while Griess discovered diazo compounds which led to the development of the currently large class of synthetic dyes, namely the azo compounds. The first true azo dye, Bismark Brown, was developed by Martius in 1863.1
This chapter discusses the principles of dyeing in a general manner, the classification of dyes highlighting specific examples of dye classes. The following chapters will present considerably more detailed discussions regarding textile and industrial dyeing with reference to principles, processes and types of dyes.
1.2 Principles of dyeing
The objective of dyeing is to produce uniform colouration of a substrate usually to match a pre-selected colour. The colour should be uniform throughout the substrate and be of a solid shade with no unlevelness or change in shade over the whole substrate. There are many factors that will influence the appearance of the final shade, including: texture of the substrate, construction of the substrate (both chemical and physical), pre-treatments applied to the substrate prior to dyeing and post-treatments applied after the dyeing process. The application of colour can be achieved by a number of methods, but the most common three methods are exhaust dyeing (batch), continuous (padding) and printing.
1.3 Exhaust dyeing
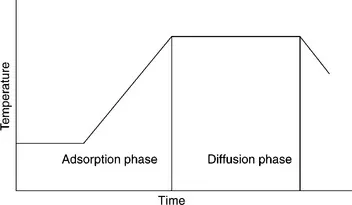
In exhaust dyeing the dye, which is wholly or partially soluble in the dyebath, is transported to the fibre surface by the motion of the dye liquor or by motion of the substrate being dyed. The dye is adsorbed onto the fibre surface and ideally diffuses into the whole of the fibre. Depending upon the dye being used, the interactions between the dye and the fibre can be either chemical or physical. Exhaust dyeing is usually conducted using dilute solutions of dyes, normally termed long liquor dyeing, and can involve liquor to substrate ratios from 8:1 up to 30:1. As described above there are two main phases to exhaust dyeing, the adsorption phase and the diffusion phase. Most exhaust dyeing involves a temperature gradient whereby the dyeing is commenced at a fairly ambient temperature (30–40°C) with the temperature being increased slowly up to a final temperature which is dependent upon the dyes being used (Fig. 1.1).
1.1 Typical dyeing profile.
Depending upon the dyes being used, during the diffusion phase, changes to the dyebath pH may be made to facilitate covalent fixation of the dye which has diffused into the substrate.
Exhaust dyeing recipes, including auxiliaries together with the dyes, are traditionally made up by percent weight relative to the weight of substrate being dyed. The auxiliaries are introduced first into the dyebath and allowed to circulate to enable uniform concentration throughout the dyebath and on the substrate surface. The dyes are then introduced into the dyebath and again allowed to circulate before the temperature is raised in order to obtain a uniform concentration throughout the dyebath. Gaining uniform concentrations of both auxiliaries and dyes is paramount since non-uniform concentrations on the substrate surface can lead to unlevel dye uptake. The speed of dye uptake (exhaustion) of individual dyes can vary and will depend upon their chemical and physical properties together with the type and construction of substrate being dyed. The dyeing rate also depends upon the dye concentration, the liquor ratio, temperature of the dyebath and the influence of the dyeing auxiliaries. Rapid exhaustion rates lead to unlevelness of dye distribution over the substrate surface, so dyes have to be carefully selected when used in multi-dye recipes; many dye manufacturers produce information stating which dyes from their ranges are compatible to achieve level build-up of dye during dyeing. Dyers wish to achieve the highest exhaustion possible to minimise dye remaining in the effluent and increase batch to batch reproducibility, whilst still obtaining the shade required by the customer. The dyeing process will eventually end in equilibrium, whereby the dye concentration in the fibre and the dyebath do not change significantly. It is envisaged that dye adsorbed onto the substrate surface has diffused into the whole of the substrate resulting in a uniform shade required by the customer and that there is only a small concentration of dye left in the dyebath. This is where the final shade of the substrate is checked against the standard. If there is any deviation from the required shade, small additions of dye may be made to the dyebath to achieve the required shade.
Dyers wish to achieve the correct shade the first time of dyeing in order to minimise further processing and reduce costs. In order to do this uniform dyeing rates and high exhaustion rates of dyes are required. To achieve short dyeing cycles, thereby maximising production, most modern dyeing equipment is enclosed ensuring that the dyebath is maintained at the required temperature and that there are no temperature variations within the dyebath. Some dyeing machines can be pressurised enabling the dye liquor to be heated to 130°C allowing substrates, such as polyester...