![]()
Chapter 1
Introduction
Abstracts
Chapter 1 briefly reviews the historical development of furnaces and notes their position at the heart of our industrial society. The concept of a furnace and furnace efficiency is defined together with a classification of the types of furnace based on energy source and product type. The designer’s basic objectives are outlined and the importance of achieving satisfactory product quality as the over riding key objective is explained.
The remainder of the chapter examines the industries in which furnaces are used and the types of furnaces used in those industries. This includes ceramics, cement & lime, glass, metal ore smelting and refining, metal processing, furnaces with reducing atmospheres, oil refining and petrochemical applications as well as incineration and resource recovery furnaces. The chapter concludes with a discussion on the drivers for improved efficiency.
Key words
furnace definition; classification; furnace efficiency; furnace applications; reducing atmospheres; drivers for improved efficiency
Chapter contents
1.1 What is a Furnace?
1.2 Where are Furnaces Used? Brief Review of Current Furnace Applications and Technology
1.3 Drivers for Improved Efficiency
1.4 Concluding Remarks
References
Combustion and furnaces are the very heart of our society. Furnaces and kilns are used to produce virtually everything we use and many of our food and drink items, either directly such as the metal products used in everyday life, the packaging for our food and drink or indirectly such as the tools that are used to grow that food. Even a natural product, such as timber, requires drying to make it suitable for most uses, and that drying occurs in a kiln.
From the earliest days of human existence food was cooked over open fires and sticks were charred to harden them. However, open fires provided little control over the heating process, and the birth of the Bronze Age some 5000-6000 years ago would have required the construction of a forced draft furnace to achieve the temperature required to smelt the ore and produce liquid metal for casting. Metal production remained small scale for centuries owing to the scarcity of suitable fuel (charcoal) and the high cost of its production. The breakthrough came as a result of the determination and tenacity of Abraham Darby who worked much of his life to reduce the cost of cast iron by using coke in place of charcoal. He finally succeeded and a lasting testament to his work is the world’s first iron bridge, completed in 1779 crossing the River Seven at Coalbrookdale in North West England, Figure 1.1.
Figure 1.1 The iron bridge at Coalbrookdale showing the detail adjacent to an abutment and “Coalbrookdale by Night” - Philip James de Loutherbourg
Darby’s pioneering work laid the foundations for the industrial revolution because iron production was no longer constrained by fuel supply and blast furnaces proliferated in the Severn Valley and even changed the way artists perceived the landscape, as exemplified in Philip James de Loutherbourg’s famous landscape “Coalbrookdale by Night” now exhibited in the Science Museum, London. In addition to pioneering the use of coke, Darby also utilised steam engines for powering his furnace bellows and hence make the operation far less dependent on water power and less reliant on local rainfall, which was problematic and on many occasions drought had interrupted production for long periods. By establishing his furnaces independence of the vagaries of the charcoal burners and the weather, Darby triggered an industrial revolution that is continuing to this day.
The vast majority of the materials we use, and the food and drink we consume, have been heated at some stage during the production process. Combustion processes are conveniently divided into high and low temperature process. Although there is no strict dividing line between the two, processes with wall temperatures below 400-500°C are often considered low temperature, while those above 500-800°C are generally considered high temperature. High temperature processes include, cement and lime manufacture, brick and ceramic manufacture, most metal processing, glass making, etc., while low temperature processes include drying processes, food processing and sterilisation, steam raising, oil refining, etc..
It is much more difficult to ensure efficient use of the fuel’s energy in high temperature processes than their low temperature counterparts. For low temperature processes, such as steam raising, efficiencies of over 80% are commonly obtained, whereas for high temperature processes, efficiencies exceeding 50% are rare. Furnace design engineers of the future will be required to maximise overall process efficiency in a carbon constrained world. This requirement will be driven both, by the community need to achieve greenhouse gas reduction, as well as the process economics owing to high fossil fuel costs, especially for processes requiring premium fuels, such as oil and gas. The aim of this book is to assist engineers to achieve higher performance both from existing, and from new furnace designs, by providing the readers access to the tools that assist with gaining an in depth understanding of the fundamentals of the individual processes. The book does not attempt to provide an in-depth understanding of any individual process because that would require a book in itself, for each industry, as can be seen from the wide variety of furnaces outlined in this introduction. In any case, we consider that the weakness of that approach is that the knowledge remains confined to that particular industry. For example, the excellent kiln efficiencies achieved by the lime industry were almost unknown in the pulp and paper industry in the early 1980s hence their lime kilns used some 30–50% more fuel than those in the mainstream industry at that time.
1.1 What is a Furnace?
The Oxford English Dictionary defines a furnace as “an enclosed structure for intense heating by fire, esp. of metals, or water”, whereas a kiln is described as “as furnace or oven for burning, baking or drying, esp. for calcining lime or firing pottery”. In reality, there is little difference between the two, kilns and furnaces both operating within a similar temperature range. The two names owe more to tradition than to functional differences.
Furnaces are the basic building block of our industrial society, indeed they are the foundation of our entire civilisation, as shown above. The principle objective of a furnace is to attain a higher processing temperature than can be achieved in the open air. Although some processes could be carried out in the open air, to do so would be far less efficient, the fuel consumption would be much higher and control of the process would be much more difficult.
Furnaces can be used to facilitate a wide range of chemical reactions, or in some cases to simply undertake physical processes, such as annealing or drying. Design of the former is normally more complex than the latter but not exclusively so. One of the challenges of furnace design is to determine the critical rate determining step(s) and to ensure that these are effectively addressed in the design. By achieving this, smaller more efficient furnaces and more cost effective designs can often be developed.
In this book we shall be dealing with furnaces for processing materials at high temperatures, that is, above 400°C, especially those where the product is directly exposed to the flame. We shall not discuss steam boilers, the design and manufacture of which is a highly specialised subject. In any case steam boilers have reached such high efficiencies that only marginal gains are possible. Future gains in the efficiency of electricity generation, for example, depend on improving the cycle efficiency; while this will impact on boiler design that is not where the gains will originate. Readers should consult Babcock and Wilcox (2005) for further information on boiler design.
1.1.1 Furnace Outline
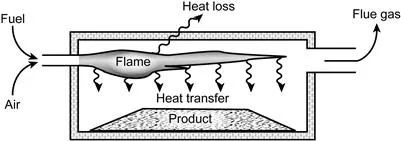
The basic concept of a furnace is shown in Figure 1.2, below.
Figure 1.2 The Basic Elements of a Furnace
Heat is liberated by burning fuel with air (or oxygen), or from electrical energy, and some of ...