![]()
1
Biochar for environmental management: an introduction
Johannes Lehmann and Stephen Joseph
What is biochar?
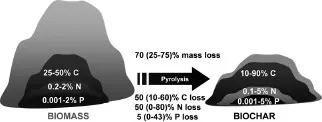
Biochar is the product of heating biomass in the absence of or with limited air to above 250°C, a process called charring or pyrolysis also used for making charcoal (Chapter 3). The material distinguishes itself from charcoal or other carbon (C) products in that it is intended for use as a soil application or broader for environmental management. In some instances, the material properties of biochar may overlap with those of charcoal as an energy carrier, but many types of biochar do not easily burn and charcoals are typically not made to address soil issues (Nomenclature in Box 1.1). An important defining feature of biochars, similar to charcoal, is a certain level of organic C forms, called fused aromatic ring structures (Chapter 6). These structures are formed during pyrolysis and are key to biochar properties with respect to mineralization (Chapter 10) or adsorption (Chapter 9). Therefore, biochar is typically enriched in C (Figure 1.1), and even more in phosphorus (P) or other metals such as calcium (Ca) or magnesium (Mg) and sometimes even nitrogen (N). The chemical properties of the organic C structure of biochars are fundamentally different from those of the material that the biochar was produced from and depleted in oxygen (O) and hydrogen (H). In contrast, the macro-morphological characteristics of biochars typically resemble those of the starting material, which means that it typically looks the same, apart from its black color. The intended use as a soil amendment also requires that biochars do not contain harmful levels of heavy metals or organic contaminants (IBI, 2013), in keeping with related efforts to make composts and other soil amendments safe for soil. Despite these common criteria, it would be wrong to conclude that biochar is a narrowly defined material. In fact, biochars can have very different properties, which have to be recognized, as discussed throughout this book.
Figure 1.1 Conversion efficiency of biomass, C, N and P during pyrolysis (data from Enders et al (2012); typical losses followed by range in brackets)
Box 1.1 Nomenclature of biochar and related materials in comparison to pyrogenic C structures
Johannes Lehmann, Joseph J. Pignatello, Michael Bird, Stephen Joseph
The following nomenclature for biochar and related terms has been adopted in this book and may provide guidance for achieving greater clarity. In some instances, clarity in conversation may also improve conceptualization and scientific advances, which is intended to promote understanding of biochar properties and its behavior in the environment.
Biochar: Biochar is the solid product of pyrolysis, designed to be used for environmental management. IBI (2013) defines biochar as: ‘A solid material obtained from thermochemical conversion of biomass in an oxygen-limited environment. Biochar can be used as a product itself or as an ingredient within a blended product, with a range of applications as an agent for soil improvement, improved resource use efficiency, remediation and/or protection against particular environmental pollution and as an avenue for greenhouse gas (GHG) mitigation.’ In addition, to be recognized as biochar according to IBI (2013) or Delinat (2012), the material has to pass a number of material property definitions that relate both to its value (e.g., H/Corg ratios relate to the degree of charring and therefore mineralization in soil) and its safety (e.g., heavy metal content). This publication uses the term biochar even when citing publications that use other terms but clearly refer to the use of such materials in the context defined for biochar.
Hydrochar: Hydrochar is the solid product of hydrothermal carbonization (HTC) or liquefaction (sometimes referred to as HTC material), and is distinct from biochar due to its production process and properties (Libra et al, 2011). It typically has higher H/C ratios (Schimmelpfennig and Glaser, 2012) and lower aromaticity than biochar as well as little or no fused aromatic ring structures. Hydrochar is not covered in this publication and only occasionally discussed in comparison to biochar.
Pyrogenic Carbonaceous Material (PCM): PCM is introduced here as the umbrella term for all materials that were produced by thermochemical conversion and contain some organic C, such as charcoal, biochar, char, black carbon, soot, activated carbon. The term refers to the material and not the C atom.
Char: Char is defined for the purpose of this publication as the material generated by incomplete combustion processes that occur in natural and man-made fires.
Charcoal: Charcoal is produced by thermochemical conversion from biomass (mainly but not exclusively wood) for energy generation. The term is sometimes used in the context of other uses, e.g., medicine, filtration, separation etc. If processed further by any form of activation, use of the term ‘activated carbon’ is proposed.
Activated carbon: Activated carbon denotes a PCM that has undergone activation, for example by using steam or additions of chemicals. It is used in filtration or separation processes, sometimes in restoration and for specialized experiments in soil (competition, inoculation, etc). ‘Carbon’ in this context should not be abbreviated to ‘C’, since it does not refer to the C atom in activated carbon, but to the material (which also contains other atoms than C). The acronym ‘AC’ for activated carbon will be used in this publication only if needed repeatedly, but the preferred spelling is ‘activated carbon’. Clarification is needed in those instances where biochars were modified after production for which some sources use the term ‘activation’. Such treatment of biochars is typically ill defined and it should be explained in detail what ‘activation’ of biochars means in a particular study. The use of the term ‘activated biochar’ is discouraged.
Black carbon: The term black carbon (carbon spelled out) is extensively used in the atmospheric, geologic, soil science and environmental literature to refer to PCMs dispersed in the environment from wildfires and fossil fuel combustion. The term should be taken to refer to the entire material, not just the fused ring fraction or the C atom. The use of this term is discouraged (or be used only if absolutely necessary and in the context described here), to avoid confusion with ‘black C’, which is defined below.
Soot: Soot is a secondary PCM and a condensation product (Chapter 3). Chars, charcoal, biochars, black carbons (and, to a limited extent, also activated carbon) may contain soot, but soot can also be identified as a separate component resulting from gas condensation processes.
Ash: Ash is the operationally defined fraction of biomass or PCM (according to ASTM D1762-84) and typically includes inorganic oxides and carbonates (Enders et al, 2012). For the purposes of this publication, the term does not describe the solid residue of combustion which commonly contains some residual organic C.
When referring to the C atoms of the PCM, the letter C should be used as in ‘pyrogenic C’ or ‘black C’. A selection of terms referring to C forms in PCM relevant to this publication includes:
• ‘Black C’ spelled with ‘C’ and not ‘carbon’ refers to the C atom, and not to the material that also contains H, O, N and ash minerals (Figure 1.2). ‘Black C’ should not be abbreviated to BC as this can be confused with biochar (which is in some publications abbreviated to BC; the acronym BC is therefore not used here).
• ‘Pyrogenic C’ (abbreviated to PyC after first use) is synonymous with black C. It should be used preferentially to ‘black C’.
• PyC (or black C) should refer to the (non-inorganic) C atoms that have undergone pyrogenic or thermal transformation, and by this definition only include C present in fused rings, including C on surfaces of fused aromatic C that may also bind to other atoms than C such as C-O/N, non-protonated C and protonated C. In this publication, the term does not include non-transformed C present in residual carbohydrates or lignin structures, or in tars, or in functional groups bound to fused aromatic C such as carboxyl groups. Different methods to quantify PyC (or black C) typically attempt to capture this C fraction (Chapter 24), but do so with varying success or intentionally capture a portion of it (e.g., only the fused aromatic C without the surface C). When referring to a certain analytically defined fraction, the method should be stated in conjunction with the term PyC (e.g., PyC quantified by CTO-375).
• ‘Total Organic Carbon’ (abbreviated TOC) refers to the entire organic C component of any material, and is similarly defined for PCMs (Chapter 8), including all thermally altered organic C as well as remaining untransformed organic C. ‘Total inorganic carbon’ (abbreviated TIC) mainly includes carbonate and possibly other compounds such as oxalates (Figure 1.2).
• In some cases ‘soot C’ is appropriate to indicate the C atom properties of soot (which is a secondary PCM as defined above).
Figure 1.2 Illustrative sketch of a possible distribution of different C forms and other atoms in biochar (component and acronyms explained in text)
A brief history of biochar research and application
Valuing biochar-rich soils and the concept of adding biochar to soil and in potting mixes reaches back several centuries (Chapters 2 and 12) and has found entry into some traditional management concepts in many regions worldwide (Chapter 2). Even though some notable research was done, the historic reports and scientific studies started as mostly observational and were initially in large part gathered from plant growth responses on former charcoal storage sites (Chapter 12). Biochar application was discussed in major agricultural textbooks (Allen, 1846) and in scientific journals (Anonymous, 1851) and developed into commercial products as a form of ‘manure’ in the mid 1800s, but with varying success as seen for biochar made from peat (Durden, 1849). By the second half of the nineteenth century, scientific studies on biochar had increased substantially, not the least due to Justus von Liebig’s publications (Liebig, 1852) providing quantitative proof combined with theoretical underpinning of why biochar may improve nutrient availability. This interest in biochar continued into the twentieth century (e.g., Retan, 1915; Morley, 1927), but most of the research and development subsequently ceased by the middle of the twentieth century, possibly trailing the development and marketing of inorganic fertilizers. Notable research and development on biochar started again in the 1980s in Japan (Ogawa and Okimori, 2010). The present interest in biochar research and development was mainly motivated by research on Amazonian Dark Earths (also called Terra Preta de Indio; Mann, 2002; Marris, 2006). These soils found in the Amazon Basin were created by Amerindian populations several hundred to a few thousand years before present, but maintained their fertility largely due to the high proportion of biochar-type organic matter (Glaser and Birk, 2012). Even though Terra Preta soils do not provide a direct analogue to biochar management (Lehmann, 2009) and are by far not the only soils containing biochar (Chapter 2), they can be credited for spurring recent investigation into whether biochar can provide broader soil benefits in its own right (Glaser et al, 2002). In parallel, naturally produced chars from vegetation fires are becoming re-appreciated as the reason for the high fertility attributes of some soils such as those in the U.S. midwest (Mao et al, 2012).
The term ‘biochar’ was introduced only recently, first as a term to distinguish activated carbon made from fossil fuel and activated carbon made from biomass (Bapat et al, 1999), and shortly thereafter to replace the term ‘charcoal’ as a fuel (Karaosmanoglu et al, 2000) and to distinguish it from coal. Biochar, as the term used in this book and by now more widely accepted globally in the context of a soil amendment, was introduced in 2006 (Lehmann et al, 2006) based on conversations with Peter Read.
Research on Terra Preta and on naturally occurring chars (often under the term black C) dominated the scientific literature on biocharrelevant topics ten years ago, but in 2008 the number of articles in academic journals on purposeful application of biochar to soil started to increase (Figure 1.3). The term charcoal continues to be used in the context of a soil amendment, but with a decreasing proportion. The publication activity of biochar in the scientific literature now exceeds that in the more established subject of compost science (Figure 1.3). Similarly, citations of scientific articles on biochar have risen and are also higher than those on compost for the ten most-cited journal ...