
eBook - ePub
Therapeutic Antibody Engineering
Current and Future Advances Driving the Strongest Growth Area in the Pharmaceutical Industry
- 696 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Therapeutic Antibody Engineering
Current and Future Advances Driving the Strongest Growth Area in the Pharmaceutical Industry
About this book
The field of antibody engineering has become a vital and integral part of making new, improved next generation therapeutic monoclonal antibodies, of which there are currently more than 300 in clinical trials across several therapeutic areas. Therapeutic antibody engineering examines all aspects of engineering monoclonal antibodies and analyses the effect that various genetic engineering approaches will have on future candidates. Chapters in the first part of the book provide an introduction to monoclonal antibodies, their discovery and development and the fundamental technologies used in their production. Following chapters cover a number of specific issues relating to different aspects of antibody engineering, including variable chain engineering, targets and mechanisms of action, classes of antibody and the use of antibody fragments, among many other topics. The last part of the book examines development issues, the interaction of human IgGs with non-human systems, and cell line development, before a conclusion looking at future issues affecting the field of therapeutic antibody engineering.
- Goes beyond the standard engineering issues covered by most books and delves into structure-function relationships
- Integration of knowledge across all areas of antibody engineering, development, and marketing
- Discusses how current and future genetic engineering of cell lines will pave the way for much higher productivity
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
Immunology1
Introduction to biologics and monoclonal antibodies
Abstract:
The first recombinant protein therapeutic was Humulin®, recombinant human insulin, which was licensed in 1982. Today, there are four different types of biologics, including replacement protein therapeutics with enzymatic or regulatory activity, protein therapeutics with binding specific targets, protein-based prophylactic vaccines, and protein diagnostics. Monoclonal antibodies and Fc fusion proteins are the major forms of therapeutic proteins possessing binding activities. Monoclonal antibodies have two major functional domains, the FAb arms, which are responsible for binding the antigens, and the Fc domain, which interacts with the immune system. The potency of a monoclonal antibody depends on the strength of binding, the epitopes to which the antibodies bind, and the Fc activity. Monoclonal antibodies are named according to the sources from which they were obtained as well as the human component of the antibody.
Keywords
biologics protein therapeutics vaccines
insulin small molecules monoclonal antibodies Fc fusion proteins naming of therapeutic antibodies
1.1 Introduction
Emil von Behring, considered to be the father of immunology and winner of the first Nobel Prize in Medicine and Physiology in 1901, and Shibasaburo Kitasato, eventual founder of Japan’s famed Kitasato Institute, were the first to discover that a substance in blood was capable of neutralizing diphtheria toxin (Behring and Kitasato, 1890). This substance was subsequently named “Antikörper”, or “antibodies”, and was determined to have specificity for one toxin over another. In the following years, the terms “Antisomatogen” and “Immunkörperbildner” also were coined to describe the material that induced the formation of the Antikörper. The term “antigen” eventually arose from the combination of those names (Schroeder and Cavacini, 2010), and thus the foundation of immunology consisting of an antigen and its cognate antibody was laid for the next century of antibody research.
The field of genetically engineered therapeutic monoclonal antibodies (MAbs), the topic of this book, has been built on the shoulders of many inventions over decades of research, but two key discoveries in the mid-1970s stand out as seminal events that laid the groundwork for this field to exist as it does today. When Stanley Cohen, Herbert Boyer, and their colleagues made the first recombinant DNA molecules in 1973 by isolating a bacterial plasmid, cutting it site-specifically with the restriction endonuclease EcoRI, inserting foreign DNA into it, and then reforming the modified plasmid with DNA ligase (Cohen et al., 1973), they started a revolution that has changed the medical world in ways likely envisioned by only the wildest imaginations at that point in time. Virtually every new drug developed by the pharmaceutical industry today has been derived by a process that includes cloning and expression of the drug target, and in many cases, X-ray crystallographic analysis of the drug and recombinant target together. Likewise, the entire field of monoclonal antibodies derives from a key event, i.e. the discovery by Köhler and Milstein (1975) that murine B cells could be fused with murine myeloma cells to produce single fusion cell lines (hybridomas), which produce antibodies with a single, unique specificity (i.e. monoclonal antibodies). The field of genetically engineered MAbs, which marries standard molecular biology with antibody technologies, can be traced directly back to those two seminal papers.
Now, more than 35 years later, the field of genetically engineered MAbs, using technologies descended from both Cohen et al. (1973) and Köhler and Milstein (1975), is a rapidly maturing field in which hundreds of researchers in academia and in dozens of biotechnology and biopharmaceutical companies are engaged. Genetically engineered therapeutic antibodies have now been marketed for over a quarter century, starting with the United States Food and Drug Administration (FDA) approval of ReoPro® in December of 1984. Since that approval, an additional 39 recombinant therapeutic antibodies and related Fc fusion proteins (FcFPs) have been approved for marketing, with hundreds more now in clinical trials to follow them. This book describes the current status of therapeutic antibody engineering as well as the path to get to where we are today.
It also attempts to address the major issues, challenges, and opportunities we will face as we push forward into the second decade of the twenty-first century.
1.2 Definitions of biologies
Biologics have been defined by the FDA as “a virus, therapeutic serum, toxin, antitoxin, vaccine, blood, blood component or derivative, allergenic product, or analogous product, or arsphenanaine, or derivative of arsphenamine (or any other trivalent organic arsenic compound), applicable to the prevention, treatment or cure of a disease or condition of human beings” (Public Health Services Act 42 USC § 262(i)). While this definition by statute is now antiquated, it does provide a broad guidance as to what may be included in the term “biologic.” Clearly, chemical compounds like arsphenines that were historically considered as biologics no longer fit the description. Leader et al. (2008) recently summarized the field of biologics, placing biopharmaceutical proteins into four major categories:
1. protein therapeutics with enzymatic or regulatory activity (e.g. replacement therapies such as insulin, growth hormone, Factor IX, β-glucocerebrosidase);
2. protein therapeutics with special targeting activity (e.g. MAbs or other binding proteins, including FcFPs, that bind specific therapeutic targets. Two examples are the anti-tumor necrosis factor (TNF)-α MAb, Remicade®, and the anti-TNF-α/β FcFP, Enbrel®);
3. protein-based prophylactic vaccines (e.g. human papilloma virus (HPV) vaccine made using virus-like particles containing HPV major capsid protein L1); and
4. protein diagnostics (e.g. biomarkers such as glucagon, and imaging agents such as technetium- or indium-conjugated antibodies) (Leader et al., 2008).
Figure 1.1 shows the fundamental structure of an immunoglobulin G (IgG), and a typical FcFP, etanercept, as compared with the non-antibody biologic, human growth hormone (HGH), the peptide hormone insulin, and the small molecule penicillin G. This book will focus largely on therapeutic MAbs and FcFPs, with some reference to the therapeutic protein history and markets as they relate to the development and acceptance of therapeutic MAbs by the industry. Table 1.1 defines some of the most significant similarities and differences between non-MAb biologics, MAbs, small molecules, and traditional vaccines. The key differences between biologics and small molecules lie in the types of targets addressed, molecular size and complexity, inherent toxicities, off-target activities, drug metabolism, pharmacokinetics, route of administration, method of manufacturing, and product homogeneity (Table 1.1). Biologics can be subdivided into three major categories: monoclonal antibody (MAb) products, non-MAb products, and vaccines. Since vaccines are used to stimulate an immune response and, at least historically, have been typically developed for prophylactic use rather than therapeutic use (Table 1.1), they will not be addressed further here.
Table 1.1
General properties of non-MAb biologics, MAbs, small molecules, and traditional vaccines
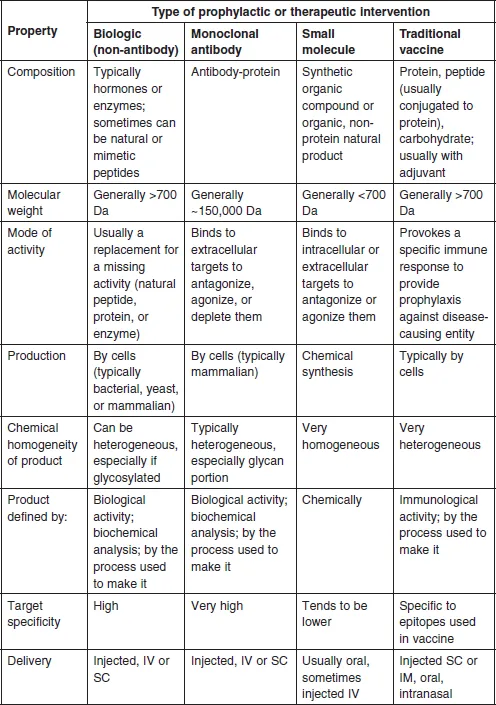
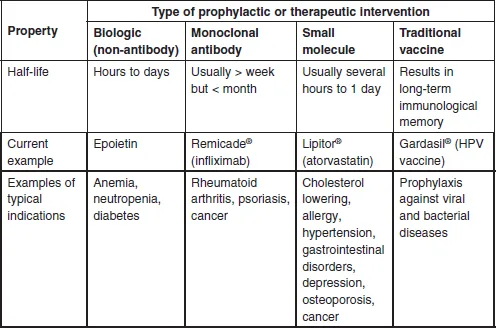
Abbreviations: Da: Dalto...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- List of figures
- List of tables
- List of acronyms, abbreviations, and definitions
- Foreword
- Preface
- About the authors
- Chapter 1: Introduction to biologics and monoclonal antibodies
- Chapter 2: Value proposition for therapeutic monoclonal antibodies and Fc fusion proteins
- Chapter 3: Antibody structure–function relationships
- Chapter 4: Fundamental technologies for antibody engineering
- Chapter 5: Sources of antibody variable chains
- Chapter 6: Variable chain engineering – humanization and optimization approaches
- Chapter 7: Antibody interactions with the immune system
- Chapter 8: Monoclonal antibody targets and mechanisms of action
- Chapter 9: Therapeutic antibody classes
- Chapter 10: Antibody Fc engineering for optimal antibody performance
- Chapter 11: IgG glycans and glyco-engineering
- Chapter 12: Antibody fragments as therapeutics
- Chapter 13: Multiple antibody and multi-specificity approaches
- Chapter 14: FcFPs and similar constructs using Fc
- Chapter 15: Antibody-drug conjugates
- Chapter 16: Development issues: antibody stability, developability, immunogenicity, and comparability
- Chapter 17: Interactions of human IgGs with non-human systems
- Chapter 18: Cell line development
- Chapter 19: Issues facing therapeutic monoclonal antibodiesfor the future
- Useful public websites related to antibody engineering
- References
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Therapeutic Antibody Engineering by William R Strohl,Lila M Strohl in PDF and/or ePUB format, as well as other popular books in Medicine & Immunology. We have over 1.5 million books available in our catalogue for you to explore.