![]()
1
Growth of Muscle from the Myoblast to Whole Muscle
Terry Brandebourg
Introduction
Better understanding of the growth and development of skeletal muscle, and to a lesser extent, adipose tissue, is an important endeavor in meat science. This goal is driven by the need for the meat industry to consistently satisfy consumer demand for nutritious, high-quality, lean products in as efficient a manner as possible. Importantly, meat products are primarily derived from the skeletal muscle and associated fat of livestock.
Muscle growth, composition, and metabolism are integrally linked to meat quality through effects on yield, tenderness, and color. Typically, meat-producing animals are grown until an optimal balance between muscle mass and fattening is achieved. Upon slaughter, livestock carcasses are dressed leaving only the bones and edible muscles. Dressed carcasses are then aged in a temperature-controlled environment where biochemical processes such as glycolysis and protein degradation contribute to optimal meat quality.
Undoubtedly, advances in our understanding of factors that regulate the growth and development of muscle and the conversion of whole muscle to meat will lead to strategies that enhance meat quality. With these goals in view, this chapter will focus upon the growth of muscle from the myoblast precursor to whole muscle and upon cell culture techniques that allow these processes to be studied.
Overview of Skeletal Muscle Development
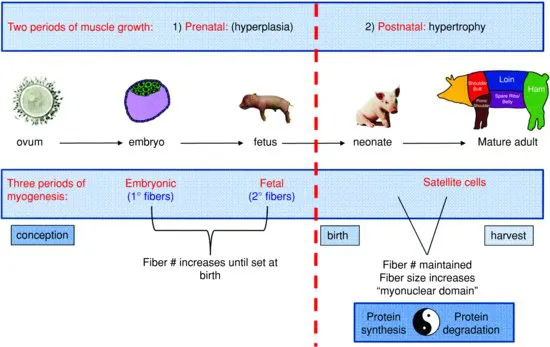
The growth of skeletal muscle can be meaningfully divided into stages by key developmental milestones. Landmarks such as conception, the maturation of the embryo (spanning the eight-cell stage through implantation), parturition, and finally, postnatal growth largely frame periods where specific mechanisms of growth contribute uniquely to muscle development. Thus, such landmarks represent useful points of reference that form a roadmap for better understanding of skeletal muscle development.
Viewed through this paradigm, skeletal muscle development can be divided roughly into two general phases of growth by parturition. Prenatal muscle development occurs primarily through increases in muscle fiber number (hyperplasia). Whereas postnatal growth of muscle is accomplished by increases in the size of preexisting muscle fibers (hypertrophy). In absence of injury, fiber number is essentially maintained during this period as very little new muscle fiber growth occurs after birth.
Alternatively, skeletal muscle development can be broken temporally into three phases of myogenesis. During early gestation, fiber number is increased via embryonic myogenesis (primary fiber formation). A second wave of fetal myogenesis (secondary fiber formation) then occurs during mid- to late-gestation. These two waves of prenatal myogenesis essentially dictate muscle fiber number in the adult. A third wave, satellite cell-related myogenesis mediates the postnatal increase in muscle fiber size that occurs in growing animals. Satellite cell fusion is also responsible for the maintenance of fiber number in the adult by facilitating the regeneration of damaged muscle fibers. Thus, an understanding of the regulation of myogenesis can largely inform all stages of skeletal muscle development (Fig. 1.1).
Types of Muscle
Three types of muscles can be distinguished structurally and physiologically in livestock. Smooth muscle is found in the walls of blood vessels, the lining of the gastrointestinal tract, uterine walls, and walls of respiratory passages. This type of muscle is innervated by the autonomic nervous system, thus its contraction is characterized by slow, but sustained contractile velocity that occurs without conscious thought. A second type of muscle, cardiac muscle, is innervated with an intrinsic nervous system unique to the heart that is specialized for generating highly controlled rhythmic contractions. Finally, skeletal muscle comprises the bulk of muscle in the body and its contraction is controlled by nerves emanating from the spinal cord. Importantly, skeletal muscle represents the primary source of meat from the carcass.
The unique and highly organized structure of skeletal muscle facilitates locomotion, a primary function of this muscle. Skeletal muscle appears striated due to the abundant expression of contractile apparatus proteins and, as discussed later, this muscle can appear reddish or whitish depending upon its fiber composition. Regardless of the anatomical location, skeletal muscles originate on a bone and terminate across the joint of another bone further away from the body's axis in such a way as to allow bones to rotate about the joint and move upon muscle contraction (Engel and Franzini-Armstrong, 2004). Muscles attach either via a tendon or upon a thin sheet of connective tissue (fascia).
Structure of Muscle
It is necessary to first appreciate the structural organization of skeletal muscle in order to understand why its development occurs as it does. The ultra-structure of the muscle cell and components of the contractile apparatus will be discussed in great detail in subsequent chapters. For now, we will focus upon the organization of the myofibril network and how this network interacts with the specialized membrane system of muscle fibers as these interactions are important for both prenatal and postnatal growth.
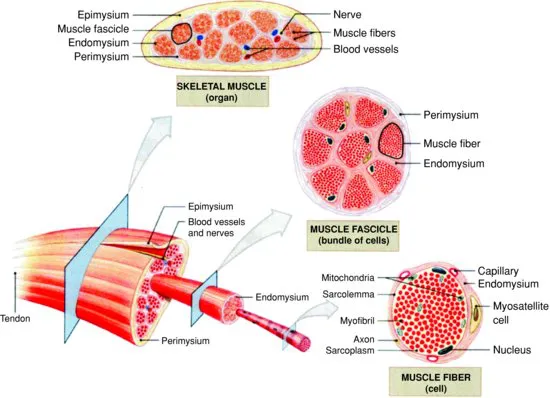
The basic structural unit of skeletal muscle is the muscle fiber (also termed muscle cell, myocyte, or myofiber). Muscle fibers are single, multinucleated cells that have peripherally located nuclei and a highly ordered and densely packed intracellular network of myofibrils, the organelle of contraction. Individual muscles are composed of a variable number of fibers that run parallel to one another. Individual fibers can be 10–100 μM in diameter and often reach many centimeters in length in the adult. Muscle fibers generally stretch from the origin to insertion in whole muscle though some individual muscle fibers terminate intrafascicularly (do not extend this entire length). Thus, intrafascicular fibers can confound efforts to quantify fiber number. Their size, multinucleated nature, specialized contractile apparatus, and unique membrane system distinguish muscle fibers from other animal cells (Fig. 1.2).
Myofibrils are specialized organelles that constitute the contractile apparatus of the muscle fiber. The basic unit of the myofibril is a repeating structure called the sarcomere. Each sarcomere comprises adjacent bundles of actin and myosin filaments which are capped on either end by a dense, proteinasceous Z-disc. Thin actin filaments extending from the Z-discs run in parallel to thick myosin filaments that appear to be centrally located in the sarcomere. Contraction occurs as these filaments interact and slide along one another thus drawing opposing Z-discs closer together.
In keeping with its unique structure, the muscle fiber is defined by a specialized, semi-permeable plasma membrane called the sarcolemma. The large number of channels and pores dispersed throughout the sarcolemma facilitate synaptic transmission and action potential propagation in response to acetylcholine release by motor neurons at neuromuscular junctions (Engel and Franzini-Armstrong, 2004). Thus, the sarcolemma is integral to proper contractile function in addition to performing the normal processes associated with a plasma membrane.
Immediately surrounding the sarcolemma is an extracellular matrix whose components are secreted by the muscle fiber proper. This thin layer of basal lamina and interwoven network of reticular fibers is directly attached to the sarcolemma and functions to maintain muscle integrity. Growth factors that bind to proteoglycans such as heparin sulfates in the basal lamina have been shown to regulate myogenesis and postnatal hypertrophy of muscle cells (Campbell and Stull, 2003). Furthermore, observed links between the basement membrane and sarcomeres of the myofibril suggest that the basal lamina may also function in contraction and myofibrillogenesis though the mechanisms are poorly understood (Street, 1983; Danowski et al., 1992; Ervasti, 2003). Importantly, the basement membrane also facilitates connection of the muscle to tendons.
The endomysium, a final layer of connective tissue primarily composed of reticular fibers, encapsulates the muscle fiber proper and surrounding basement membrane. The endomysium is attached to the sarcolemma by multiple interactions with the basal lamina and functions as a sheath that allows capillaries, nerves, and lymphatics to reside in close proximity to the muscle fiber (Gerrard and Grant, 2003). Importantly, the endomysium is thought to influence meat tenderness (Swatland, 1975a, 1975b, 1975c; Lawrie, 1991).
Organization of Whole Muscle
Just as muscle fibers are multilayered structural units comprising of specialized membranes and connective tissue, whole muscle is organized by an elaborate, ordered system of connective tissue layers that can affect muscle function and meat quality. Among these connective tissue sheaths are the perimysium and the epimysium. The perimysium organizes individual fibers into bundles called muscle fascicles. The number of individual fibers encased within the perimysium can vary. Furthermore, fascicular number and size varies with the function and anatomical location of the muscle. Importantly, intramuscular fat develops both within the perimysium and between bundles so it is possible that local signals emanating from the perimysium may affect the development of marbling through influencing either fat cell number or lipid filling. The epimysium encases multiple muscle fascicles effectively bundling them together to form the whole muscle. The epimysium is continuous around a muscle and is contiguous with the perimysium and endomysium ultimately thickening at junctions with tendons where muscle connects.
These individual layers intercalate to form a rich network that in addition to providing structural integrity also influences muscle growth both through direct and indirect effects. Physical links between the connective tissue layers and muscle fibers are thought to play important roles in regulating myofibrillogenesis and myogenesis (Menko and Boettiger, 1987; Rosen et al., 1992). Indirect effects can also be manifested by the ability of these layers to act as a reservoir for growth factors that are sequestered by proteoglycans in these rich networks (Timpl and Brown, 1996).
Although the muscle fiber is the basic structural unit of skeletal muscle, whole muscle is in fact heterogeneous with regard to cell types found within the tissue. Specialized mononucleated pre-muscle cells called satellite cells can reside between the endomysium and sarcolemma of the muscle fiber. Satellite cells play an important role in muscle hypertrophy serving as a local source of DNA that is essential to support postnatal muscle growth. Additionally, blood vessels, nerves, and lymphatics are interspersed between the perimysium and epimysium throughout the muscle. Motor neurons serve to activate a group of muscle cells which results in contraction of the fibers and thus facilitates movement. This relationship between a motor neuron and the downstream muscle fibers it stimulates is called a motor unit.
Fiber-Type Development
Heterogeneity is also a characteristic intrinsic to skeletal muscle fibers (Swatland, 1975a, 1975b, 1975c). Muscle fibers can be grouped as either slow oxidative, fast oxidative, or fast glycolytic based upon their speed of contraction and the predominant type of metabolism carried out by the fiber. Thus, muscle fibers can be distinguished by their functional properties through measuring the types of myosin heavy chain isoforms and mitochondrial oxidative enzymes expressed by the fiber. Fiber-type distribution within a muscle is related to the functional demands of the muscle, although generally within a given muscle, there will be a mosaic of fiber types with their distribution appearing more or less random (Swatland, 1975a).
Fiber-type distribution within a muscle can also affect the muscle's appearance. A muscle made up of predominantly oxidative muscle fibers will tend to be red in color, while a muscle consisting of predominantly glycolytic fibers will tend to appear white. This is because oxidative fibers have a high demand for oxygen ...