![]()
PART IV
MATERIALS PROPERTIES AND TESTING
![]()
CHAPTER 31
VISCOSITY MEASUREMENT
ANN M. ANDERSON, BRADFORD A. BRUNO, AND LILLA SAFFORD SMITH
31.1 Viscosity background
31.2 Common units of viscosity
31.2.1 Absolute viscosity, μ
31.2.2 Kinematic viscosity, v
31.2.3 Nonstandard units
31.2.4 Distinction between rheology and viscometry
31.2.5 Mathematical formalism
31.2.6 Relation of viscosity to molecular theory
31.2.7 Effect of pressure and temperature on viscosity
31.2.8 Correlations of viscosity with temperature for gases
31.2.9 Correlations of viscosity with temperature for liquids 31.2.10 Effect of pressure on viscosity
31.3 Major viscosity measurement methods
31.3.1 Drag-type viscometers
31.3.2 Bubble (tube) viscometers
31.3.3 Rotational viscometers
31.3.4 Flow-type viscometers
31.3.5 Orifice-type (cup) viscometers
31.3.6 Vibrational (resonant) viscometers
31.4 ASTM standards for measuring viscosity
31.5 Questions to ask when selecting a viscosity measurement technique
References
31.1 VISCOSITY BACKGROUND
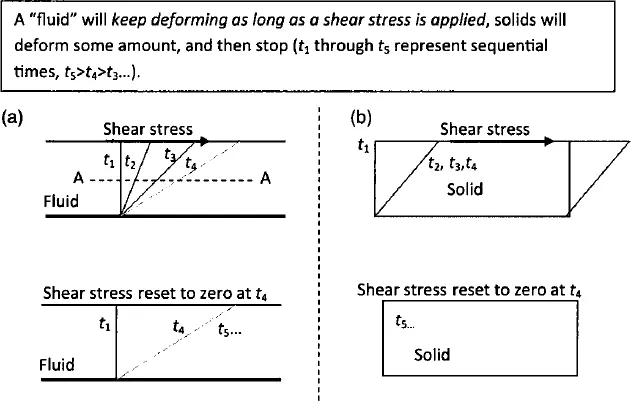
The most significant mechanical difference between materials classified as “fluids” and those classified as solids is in their reaction to shear stresses. (Recall that a shear stress is a distributed force, or force per unit area, whose direction of action is within the plane of application. If you slide your open hand over a desk top the friction between your hand and the desk will create a shear stress on your hand.) As illustrated in Figure 31.1, a solid (within its elastic limit) will deform through some limited angle while a shear stress is applied, and will then return to its original configuration when the stress is removed. A fluid on the other hand will deform continuously as long as a shear stress is applied, and will not return to its original shape when the stress is removed. For a fluid the rate of shear deformation is related to the magnitude of the shear stress applied, while for a solid the amount of shear deformation is related to the magnitude of the shear stress applied. This seemingly innocuous mechanical difference in reaction to shear stresses gives rise to the large difference in character between solids and fluids. In fact it gives rise to all of the behaviors that one thinks of as inherently “fluid”: the ability to flow, the ability to fill volumes of arbitrary shape, the ability to spread out and “wet” certain surfaces, etc. It also gives rise to the complex nature of fluid mechanics because it allows for the very large material deformations that in turn give rise to phenomena like turbulence.
Viscosity (or more precisely the shear viscosity, defined below) is the material property that defines the quantitative relation between the applied shear stress and the shear deformation rate in a fluid. Qualitatively the viscosity indicates the “thickness” or resistance to flow of a fluid. Since viscosity is the property that controls and quantifies the shear stress/shear rate behavior that is definitional to fluids, it is in many regards the most important physical property of a fluid.
Unfortunately, as alluded to above, the term “viscosity” is actually used to denote several related, but different, physical properties. It is important to understand these distinctions in terms from the outset. First, the term “viscosity” is most commonly used in conjunction with effects arising from shear forces and shear deformations in fluids. When used in this context, the most common one, the property is more precisely called the “shear viscosity” or the “first coefficient of viscosity.” However, when used in this sense, it is almost always simply referred to as “viscosity.” This is contrasted with the “bulk viscosity,” associated with volume dilatation. Bulk viscosity is rarely an important parameter and hence is not as well known or understood as the more common shear viscosity. Bulk viscosity is discussed briefly in Section 31.1.3. Second, it should be noted that even the “shear viscosity” described above is often stated in two different forms, the absolute or dynamic viscosity, μ, and the kinematic viscosity or momentum diffusivity, ν, where ν = μ/ρ and ρ is the fluid’s density. Although the dynamic and kinematic viscosities are clearly related properties, they are dimensionally dissimilar and it is critically important to always distinguish between them. More is said on the distinction between dynamic and kinematic viscosity in the following section on common viscosity units.
The remainder of this chapter begins by discussing the units in which viscosity is measured. Then the distinction between the larger field of rheology and its subfield viscometry is made in the context of differentiating between the so-called Newtonian and non-Newtonian fluids. After that the chapter provides a brief theoretical and mathematical overview of viscosity. Finally, the majority of the chapter provides detailed and practical information on methods for measuring viscosity.
31.2 COMMON UNITS OF VISCOSITY
There are several systems of units used with viscosity; many of them are archaic and/or closely tied to one specific viscosity measuring technique (e.g., the Saybolt cup and the “Saybolt Universal Second,” and the Krebs unit) or one particular industry (e.g., SAE oil grade and the automotive industry). It is impossible to capture all of these systems in one document, but an attempt is made below to define and relate the most common and standard units associated with viscosity measurement.
31.2.1 Absolute Viscosity, μ
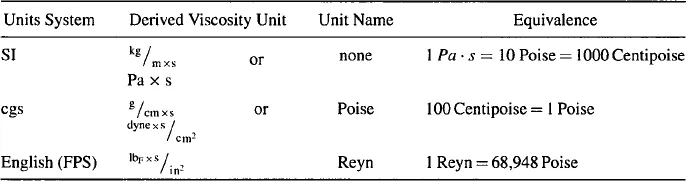
In terms of the SI (Le Systeme Internationale d’Unites) system of fundamental units the derived units for absolute viscosity, μ, are kg/m × s which is equivalent to Pa · s (Pascal-seconds). This grouping of units has not received a name of its own. In the closely related cgs (centimeter, gram, and second) system of units, the derived unit of g/cm × s or dyne × s/cm2 is called a “Poise” (after Poiseuille). More commonly a centipoise, cP = 1/100th of a Poise is used. In the FPS (foot, pound, and second) system of units, the units of absolute viscosity are IbF · s/in2, which is called the Reyn (after Osbourne Reynolds). Refer to Table 31.1 for a collection of units of absolute viscosity.
TABLE 31.1 Units for Absolute/Dynamic Viscosity, μ
31.2.2 Kinematic Viscosity, ν
Recall that the dynamic (kinematic) viscosity, v, is defined as the absolute viscosity divided by the fluid density, ρ. In the SI system of fundamental units the units for kinematic viscosity are meter square per second, which is not a named grouping. It should be noted that the units of kinematic viscosity (m2/s) are identical to the units of thermal diffusivity used in heat transfer, and mass (species) diffusivity used in diffusion. This leads to the kinematic viscosity being referred to as the coefficient of momentum diffusivity by analogy. In the cgs system the unit of kinematic viscosity is the centimeter square per second called the “Stokes” (after G.G. Stokes). More commonly the kinematic viscosity is given in centistokes (cSt) where 100 cSt = 1 Stokes. In the FPS system kinematic viscosity would be foot square per second or inch square per second, neither of which is a named unit.
31.2.3 Nonstandard Units
Kinematic viscosity is also often given in “Saybolt Universal Seconds” or SUS (also sometimes SSU “Saybolt Seconds Universal” or SUV “Saybolt Universal Viscosity), which is directly related to the Saybolt viscosity cup measuring system (see Section 31.2.2). Of course, the unit of “seconds” is not a dimensionally correct unit for the physical quantity of kinematic viscosity, so this system is problematic. The Saybolt measurement system is based on ASTM method D88 and measurements in SUS can be converted into more standard (dimensionally correct) viscosity units using procedures provided in ASTM 2161. There are countless other such “legacy” scales of viscosity associated with different industries, and unfortunately there is often no standard method for converting these legacy measures into dimensionally correct viscosity units. A number of online viscosity converters exist (see www.coleparmer.com, www.gardco.com, or www.cannon.com, for example) (Table 31.2).
TABLE 31.2 Units for Kinematic Viscosity, v
31.2.4 Distinction Between Rheology and Viscometry
A simple linear relationship between shear stress and shear strain rate is observed in a wide variety of fluids (Figure 31.2a). The constant slope of the line labeled Newtonian is the (shear) viscosity of the fluid. Fluids demonstrating such a relationship are known as Newtonian fluids. Many common fluids like air, gases in general, water, or simple oils demonstrate Newtonian beha...