Aviation ground instructor Peter J. Swatton, well reputed for his previous works in the field of pilot ground training, presents the subject in seven parts including basic aerodynamics; level flight aerodynamics; stability; manoeuvre aerodynamics; and other aerodynamic considerations. Each chapter includes self-assessed questions, 848 in total spread over eighteen chapters, with solutions provided at the end of the book containing full calculations and explanations.

eBook - ePub
Principles of Flight for Pilots
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Principles of Flight for Pilots
About this book
Organised and written as an accessible study guide for student pilots wishing to take commercial ground examinations to obtain ATPL or CPL licenses, Principles of Flight for Pilots also provides a reliable up-to-date reference for qualified and experienced personnel wishing to further improve their understanding of the Principles of Flight and related subjects. Providing a unique aerodynamics reference tool, unlike any book previously Principles of Flight for Pilots explains in significant depth all the topics necessary to pass the Principles of Flight examination as required by the EASA syllabus.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Part 1
The Preliminaries
1
Basic Principles
1.1 The Atmosphere
The Earth’s atmosphere is the layer of air that surrounds the planet and extends five hundred miles upwards from the surface. It consists of four concentric gaseous layers, the lowest of which is the troposphere in which all normal aviation activities take place. The upper boundary of the troposphere is the tropopause, which separates it from the next gaseous layer, the stratosphere. The next layer above that is the mesosphere and above that is the thermosphere.
The height of the tropopause above the surface of the earth varies with latitude and with the season of the year. It is lowest at the poles being approximately 25 000 feet above the surface of the Earth and 54 000 feet at the Equator. These heights are modified by the season, being higher in the summer hemisphere and lower in the winter hemisphere.
Above the tropopause the stratosphere extends to a height of approximately one hundred thousand feet. Although these layers of the atmosphere are important for radio-communication purposes, because of the ionised layers present, they are of no importance to the theory of flight.
Since air is compressible the troposphere contains the major part of the mass of the atmosphere. The weight of a column of air causes the atmospheric pressure and density of the column to be greatest at the surface of the Earth. Thus, air density and air pressure decrease with increasing height above the surface. Air temperature also decreases with increased height above the surface until the tropopause is reached above which the temperature remains constant through the stratosphere.
1.2 The Composition of Air
Air is a mixture of gases the main components of which are shown in Table 1.1.
Water vapour in varying quantities is found in the atmosphere up to a height of approximately 30 000 ft. The amount in any given air mass is dependent on the air temperature and the passage of the air mass in relationship to large areas of water. The higher the air temperature the greater the amount of water vapour it can hold.
1.2.1 The Measurement of Temperature
Centigrade Scale. The Centigrade scale is normally used for measuring the air temperature and for the temperature of aero-engines and their associated equipment. On this temperature scale water freezes at 0° and boils at 100° at mean sea level.
Table 1.1 Gas Components of the Air.
| Element | Volume | Weight |
| Nitrogen | 78.09% | 75.5% |
| Oxygen | 20.95% | 23.1% |
| Argon | 0.93% | 1.3% |
| Carbon Dioxide | 0.03% | 0.05% |
Note: For all practical purposes the atmosphere is considered to contain 21% oxygen and 79% nitrogen.
Kelvin Scale. Often for scientific purposes temperatures relative to absolute zero are used in formulae regarding atmospheric density and pressure. Temperatures relative to absolute zero are measured in Kelvin. A body is said to have no heat at absolute zero and this occurs at a temperature of –273.15 °C.
1.2.2 Air Density
Air density is mass per unit volume. The unit of air density is either kg per m3 or gm−3 and the symbol used is ‘ρ’. The relationship of air density to air temperature and air pressure is given by the formula:
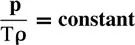
where ρ is the density, p is pressure in hPa and T is the absolute temperature.
The Effect of Air Pressure on Air Density. If air is compressed the amount of air that can occupy a given volume increases. Therefore, both the mass and the density are increased. For the same volume if the pressure is decreased then the reverse is true. From the formula above if the air temperature remains constant then the air density is directly proportional to the air pressure. If the air pressure is doubled so is the air density.
The Effect of Air Temperature on Air Density. When air is heated it expands so that a smaller mass will occupy a given volume and provided that the air pressure remains constant then the air density will decrease. Thus, the density of the air is inversely proportional to the absolute temperature. The rapid decrease of air pressure with increased altitude has a far greater effect on the air density than does the increase of density caused by the decrease in temperature for the same increased altitude. Thus, the overall effect is for the air density to diminish with increased altitude.
The Effect of Humidity on Air Density. Until now it has been assumed that the air is perfectly dry; such is not the case. In the atmosphere there is always some water vapour present, albeit under certain conditions a miniscule amount. However, in some conditions the amount of water vapour present is an important factor when determining the performance of an aeroplane. For a given volume the amount of air occupying that volume decreases as the amount of water vapour contained in the air increases. In other words, air density decreases with increased water-vapour content. It is most dense in perfectly dry air.
1.3 The International Standard Atmosphere
The basis for all performance calculations is the International Standard Atmosphere (ISA) which is defined as a perfect dry gas, having a mean sea level temperature of +15 °C, which decreases at the rate of 1.98 °C for every 1000 ft increase of altitude up the tropopause which is at an altitude of 36 090 ft above which the temperature is assumed to remain constant at –56.5 °C. The mean sea level (MSL) atmospheric pressure is assumed 1013.2 hPa (29.92 in. Hg). See Table 1.2.
Table 1.2 International ...
Table of contents
- Cover
- Aerospace Series List
- Title Page
- Copyright
- Series Preface
- Preface
- Acknowledgements
- List of Abbreviations
- Weight and Mass
- Part 1: THE PRELIMINARIES
- Part 2:: BASIC AERODYNAMICS
- Part 3: LEVEL-FLIGHT AERODYNAMICS
- Part 4: STABILITY
- Part 5: MANOEUVRE AERODYNAMICS
- Part 6: OTHER AERODYNAMIC CONSIDERATIONS
- Part 7: CONCLUSION
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Principles of Flight for Pilots by Peter J. Swatton, Peter Belobaba, Jonathan Cooper, Roy Langton, Allan Seabridge, Peter Belobaba,Jonathan Cooper,Roy Langton,Allan Seabridge in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Aeronautic & Astronautic Engineering. We have over 1.5 million books available in our catalogue for you to explore.