![]()
Chapter 1
Overview of Membrane Science and Technology
1.1 Introduction
Membranes have gained an important place in chemical technology and are used in a broad range of applications. The key property that is exploited is the ability of a membrane to control the permeation rate of a chemical species through the membrane. In controlled drug delivery, the goal is to moderate the permeation rate of a drug from a reservoir to the body. In separation applications, the goal is to allow one component of a mixture to permeate the membrane freely, while hindering permeation of other components.
This book provides a general introduction to membrane science and technology. Chapters 2–4 cover membrane science, that is, topics that are basic to all membrane processes, such as transport mechanisms, membrane preparation, and boundary layer effects. The next six chapters cover the industrial membrane separation processes that represent the heart of current membrane technology. Carrier facilitated transport is covered next, followed by a chapter reviewing the medical applications of membranes. The book closes with a chapter that describes various minor or yet-to-be-developed membrane processes, including membrane reactors, membrane contactors, and piezodialysis.
1.2 Historical Development of Membranes
Systematic studies of membrane phenomena can be traced to the eighteenth century philosopher scientists. For example, Abbé Nolet coined the word “osmosis” to describe permeation of water through a diaphragm in 1748. Through the nineteenth and early twentieth centuries, membranes had no industrial or commercial uses, but were used as laboratory tools to develop physical/chemical theories. For example, the measurements of solution osmotic pressure made with membranes by Traube and Pfeffer were used by van't Hoff in 1887 to develop his limit law, which explains the behavior of ideal dilute solutions; this work led directly to the van't Hoff equation. At about the same time, the concept of a perfectly selective semipermeable membrane was used by Maxwell and others in developing the kinetic theory of gases.
Early membrane investigators experimented with every type of diaphragm available to them, such as bladders of pigs, cattle or fish, and sausage casings made of animal gut. Later, collodion (nitrocellulose) membranes were preferred, because they could be made reproducibly. In 1907, Bechhold devised a technique to prepare nitrocellulose membranes of graded pore size, which he determined by a bubble test [1]. Other early workers, particularly Elford [2], Zsigmondy and Bachmann [3], and Ferry [4] improved on Bechhold's technique, and by the early 1930s microporous collodion membranes were commercially available. During the next 20 years, this early microfiltration membrane technology was expanded to other polymers, notably cellulose acetate.
Membranes found their first significant application in the testing of drinking water at the end of World War II. Drinking water supplies serving large communities in Germany and elsewhere in Europe had broken down, and filters to test for water safety were needed urgently. The research effort to develop these filters, sponsored by the US Army, was later exploited by the Millipore Corporation, the first and still the largest US microfiltration membrane producer.
By 1960, the elements of modern membrane science had been developed, but membranes were used in only a few laboratory and small, specialized industrial applications. No significant membrane industry existed, and total annual sales of membranes for all industrial applications probably did not exceed US$20 million in 2012 dollars. Membranes suffered from four problems that prohibited their widespread use as a separation process: they were too unreliable, too slow, too unselective, and too expensive. Solutions to each of these problems have been developed during the last 40 years, and membrane-based separation processes are now commonplace.
The seminal discovery that transformed membrane separation from a laboratory to an industrial process was the development, in the early 1960s, of the Loeb–Sourirajan process for making defect-free, high-flux, anisotropic reverse osmosis membranes [5]. These membranes consist of an ultrathin, selective surface film on a much thicker but much more permeable microporous support, which provides the mechanical strength. The flux of the first Loeb–Sourirajan reverse osmosis membrane was 10 times higher than that of any membrane then available and made reverse osmosis a potentially practical method of desalting water. The work of Loeb and Sourirajan, and the timely infusion of large sums of research and development dollars from the US Department of Interior, Office of Saline Water (OSW), resulted in the commercialization of reverse osmosis and was a major factor in the development of ultrafiltration and microfiltration. The development of electrodialysis was also aided by OSW funding.
Concurrent with the development of these industrial applications of membranes was the independent development of membranes for medical separation processes, in particular, the artificial kidney. Kolf and Berk [6] had demonstrated the first successful artificial kidney in The Netherlands in 1945. It took almost 20 years to refine the technology for use on a large scale, but these developments were complete by the early 1960s. Since then, the use of membranes in artificial organs has become a major life-saving procedure. More than 800 000 people are now sustained by artificial kidneys and a further million people undergo open-heart surgery each year, a procedure made possible by development of the membrane blood oxygenator. The sales of these devices comfortably exceed the total industrial membrane separation market. Another important medical application of membranes is for controlled drug delivery systems. A key figure in this area was Alex Zaffaroni, who founded Alza, a company dedicated to developing these products, in 1966. The membrane techniques developed by Alza and its competitors are widely used in the pharmaceutical industry to improve the efficiency and safety of drug delivery.
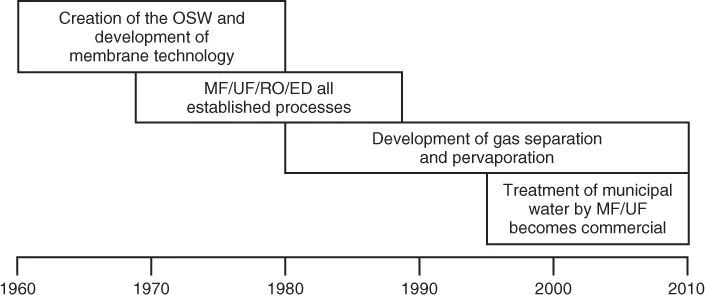
The modern membrane separation industry began in the 1960s. The creation of this industry can be divided into the four phases shown in Figure 1.1. In the first phase, building on the original Loeb–Sourirajan technique, other membrane formation processes, including interfacial polymerization and multilayer composite casting and coating, were developed for making high-performance membranes. Using these processes, membranes with selective layers as thin as 0.1 µm or less are now being produced by a number of companies. Methods of packaging membranes into large-membrane-area spiral-wound, hollow-fine-fiber, capillary, and plate-and-frame modules were also developed, and advances were made in improving membrane stability. The support of the OSW was key to these developments.
In the second phase, beginning in the early 1970s, the developments that came out of the OSW program began to appear in commercial membrane units; by the 1980s, microfiltration, ultrafiltration, reverse osmosis, and electrodialysis were all established processes. The third phase, which began in the 1980s, was the emergence of industrial membrane gas separation processes. The first major product was the Monsanto Prism® membrane for hydrogen separation, introduced in 1980 [7]. Within a few years, Dow was producing systems to separate nitrogen from air, and Cynara and Separex were producing systems to separate carbon dioxide from natural gas. Gas separation technology is continuing to evolve and expand; further growth will be seen in the coming years. Another development of the 1980s was the introduction by GFT, a small German engineering company, of the first commercial pervaporation systems for dehydration of alcohol and other solvents. Pervaporation has been slow to take off and 2010 sales were still not more than $20 million. However, the development of bioethanol from biomass has produced a new, very large potential market for the technology, which may lead to future growth.
The final development phase, which began in the mid-1990s, was the development of reliable, economical microfiltration/ultrafiltration systems for the treatment of municipal water sources and for use in membrane bioreactors in sewage treatment plants. These applications were targets for membrane developers as early as the 1960s, but membrane fouling leading to low fluxes could not be overcome. In the late 1980s, Dr. Kazuo Yamamoto began to develop low-pressure, submerged air-sparged membranes [8]. It took another 10 years for companies like Kubota, Mitsubishi Rayon, and Zenon to scale up and bring these ideas to the commercial stage, and by the late 1990s, commercial systems began to be installed. Since then, treatment of municipal water has become one of the most rapidly growing areas of membrane technology. Membrane systems are competitive with conventional biological treatment in terms of price and cost, and produce a far superior treated water product.
1.3 Types of Membranes
This book is limited to synthetic membranes, excluding all biological structures, but the topic is still large enough to include a wide variety of membranes that differ in chemical and physical composition and in the way they operate. In essence, a membrane is nothing more than a discrete, thin interface that moderates the permeation of chemical species in contact with it. This interface may be molecularly homogeneous—that is, completely uniform in composition and structure—or it may be chemically or physically heterogeneous—for example, containing holes or pores of finite dimensions or consisting of some form of layered structure. A normal filter meets this definition of a membrane, but, by convention, the term filter is usually limited to structures that separate particulate suspensions larger than 1–10 µm. The principal types of membrane are shown schematically in Figure 1.2 and are described briefly below.