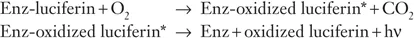![]()
part one
FIVE DIFFERENT BIOLUMINESCENCE SYSTEMS
Sixty years ago, when E. Newton Harvey of Princeton University wrote his masterly book Bioluminescence, which remains today, with its sixteen-page index, the foremost source of information on the countless animals that emit light, little was truly known on how they accomplish such a feat. The critical role of oxygen was perceived, although not as uniquely as we now know it to be, and so was that of enzymes (the special catalytic proteins called luciferases) and the smaller molecules (luciferins) whose reactions with oxygen the luciferases catalyze.
Today, it can be said that in several cases the exact step has been established where the reaction of oxygen with an enzyme-bound luciferin produces light emission. Such success stories are where our book starts. Taken together, these first five chapters illustrate the curiously haphazard impact of evolution on bioluminescence, many different animals discovering their own molecules for the emission of light.
As in most fields of biological research today, biochemists, molecular biologists, chemists, and the physicists who invented powerful new techniques and instrumentation were successful by working together. Bioluminescence is a field of study where progress depends as much on the naturalists exploring remote areas at night as on state-of-the-art instrumentation that ultimately reveals the mechanism of the light emission.
![]()
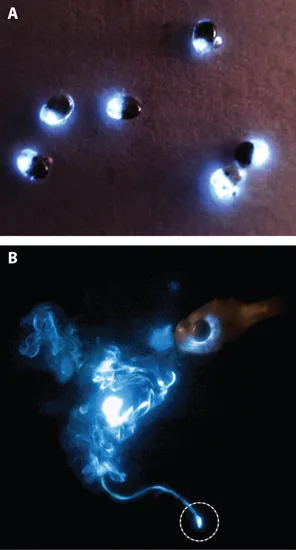
Figure 1.1. A. The bioluminescence of several small bioluminescent crustaceans (Vargula hilgendorfii) at night. Females of all Vargula species are about 50 percent larger than males. B. Massive secretory defensive display response of the luminescent Caribbean cypridinid ostracod Photeros annecohenae, swimming away unharmed at the bottom (circled), to an attack by the cardinal fish (Phaeoptyx pigmentaria) at the upper right. Note the luminescence still glowing within the oral cavity of the fish where the ostracod first responded before being spit out by the fish. The ostracod is rejected even in daylight, which suggests that it is distasteful in addition to being bioluminescent.
chapter one
A MARINE CRUSTACEAN
Bioluminescent Fishes as Plagiarists and Thieves
Vargula hilgendorfii (or Cypridina hilgendorfii, as it used to be called) is a small crustacean in the family Cypridinidae that deserves pride of place in this book for the textbook simplicity of its bioluminescence mechanism. It is about 2 to 3 mm in diameter, protected by a hard shell (Figure 1.1A), and abundant in shallow waters along the southern coast of Japan, as well as in many other areas.
Buried in the sand during the day, it becomes an active scavenger at night, escaping potential predators by squirting a bright blue luminescent puff, which may startle or temporarily blind them, or may serve as a decoy, allowing their escape in darkness. Sometimes the luminescence may attract a predator of the first predator, which eats it or scares it to regurgitate the cypridinid it had captured. There is also strong evidence that cypridinids are distasteful; after being taken in by a predator, such as a fish, they are regurgitated and may swim away unharmed (Figure 1.1B).
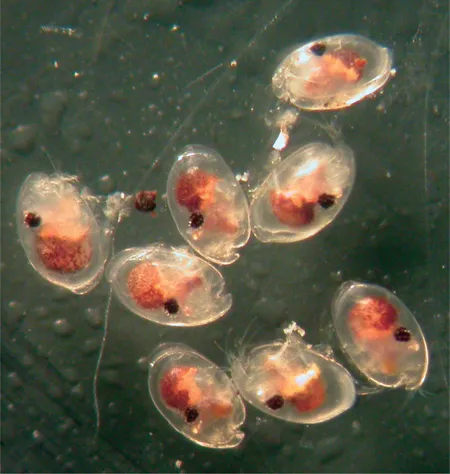
In the Caribbean Sea and, remarkably, apparently not known elsewhere, luminescent males in this family are more inventive. They produce astonishing pulsed displays that leave punctuated tracks of blue light in the water as courtship signals to females (Figure 1.2). Male Photeros annecohenae, for example, start by emitting either two or four pulses at the bottom, at the level of the sea grass. They then swim upward for half a meter in a helical pattern, emitting a light pulse every other turn. If this display does not attract a female, the male goes straight back down and starts spiraling up again. The timing precision of these displays matches that of firefly flashes. Around some reefs in the Caribbean Sea, the local male cypridinids (Figure 1.3) favor horizontal displays, but again with the same timing precision.
Figure 1.2. Courtship displays created by beads of bioluminescence squirted into the water by male cypridinids, swimming at a speed greater than 3 cm per second (or about twenty body lengths a second!); the displays are visible from at least 15 meters. In attempts to attract sexually receptive females, horizontally displaying males competing concurrently leave trails of pulses in complex fan-like patterns (upper left) above the reef. A starlight camera recorded the luminescent displays simultaneously with an infrared camera recording the reef (made visible by means of infrared lights). Both horizontal and vertical display trains are visible, produced by two new but still unnamed species.
Figure 1.3. Horizontally displaying male cypridinid ostracods (a new species in a new genus, name not yet assigned) that produce displays above reefs in Belize, such as shown in Figure 1.2. Each male is 1.6 × 0.95 mm and has a pair of large black compound eyes. The light organ is the yellowish bar below the eye that extends toward the notch at the anterior end of the valves.
Cypridinids secrete their bioluminescence reagents from two separate glands, as a chemist would do from two test tubes. Both secretions are remarkably stable as dry powders. During World War II, the Japanese collected and dried vast amounts of Vargula hilgendorfii; they thought that by moistening the powder on the palm of their hands or spreading it on each other’s backs, soldiers would be able to read maps or keep track of one another at night. Such clever tricks were apparently never employed, but the large store of dry Vargula came to good use later for elucidating the chemistry of the reaction.
How does it work? This is the question that observers of bioluminescences, now as in centuries past, ask first, fascinated as they are by the mystery of a firefly flash or the puff of blue light in the ocean. Like all bioluminescences, emission from Vargula hilgendorfii is the result of a reaction of oxygen (O2), the form of oxygen that we breathe. No exception to the rule that oxygen or another of its active forms is required for bioluminescence has ever been reported. The oxygen requirement had already been demonstrated in the seventeenth century by Robert Boyle, who observed that the luminescence of decomposing fish (now known to be caused by bacteria) or of fungi on rotting wood was extinguished if air was pumped out but restored when air was readmitted. However, the specific molecules with which oxygen reacts differ from one bioluminescent group of organisms to another; this is a key feature of bioluminescence, important to keep in mind.
At the end of the nineteenth century, the French scientist Raphael Dubois established that both a heat-sensitive component (an enzyme that he called luciferase; see Glossary) and a much smaller component (the substrate called luciferin) play a role in firefly (beetle) bioluminescence. In fact, all bioluminescence reactions are catalyzed by enzymes that are generally specific to each taxon. In shorthand, such reactions can be expressed as follows:
These two lines tell that luciferin binds with luciferase (Enz) and then reacts with oxygen; one might say that the role of binding is to put the luciferin in a proper mood to react with oxygen.
The asterisk on “oxidized luciferin*” indicates that this reaction product is in a so-called excited state, a state richer in energy, from which it promptly falls back to the “ground state” with the release of its excess energy, either as light (a photon, designated by hν) and/or as heat, if the conversion is not 100 percent efficient—it never is. The way to grasp how this works is to remind ourselves of fluorescence (Box 1.1).
Box 1.1. What is fluorescence?
We know that some materials “fluoresce” when exposed to light; that is, they take in the energy of “exciting” photons (hνEXC in figure below) that are usually present in ambient light and re-emit some of their energy as fluoresence light (hνFL), whose color is shifted to the red of that of the exciting light. If one exposes tonic water to ultraviolet light, the quinine that it contains emits a beautiful blue fluorescence; in shorthand this is written in two steps:
Q + hνEXC → Q*
Q* → Q + hνFL
Fluorescence is shifted to the red of the exciting light, because some of the energy of the exciting photon ends up inducing motions of vibration and rotation in the newly excited molecule, and therefore hνFL contains less energy than hνEXC. Thus, fluorescence spectra are moved to the red of the absorption spectra. One can think...