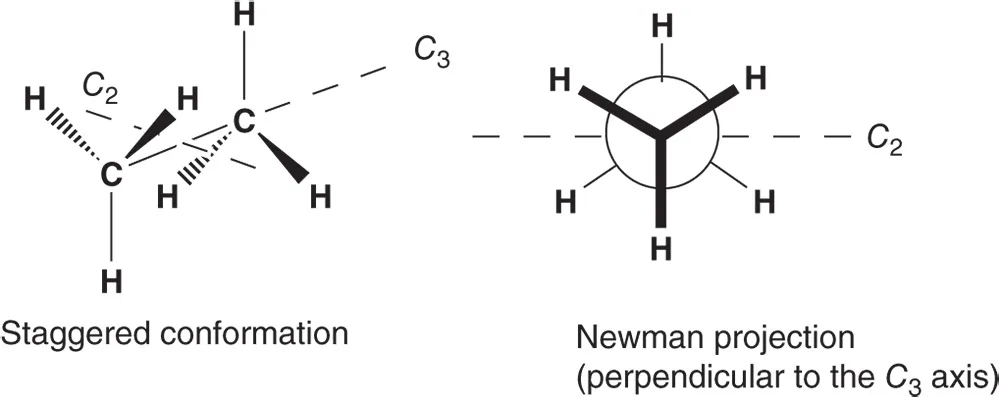
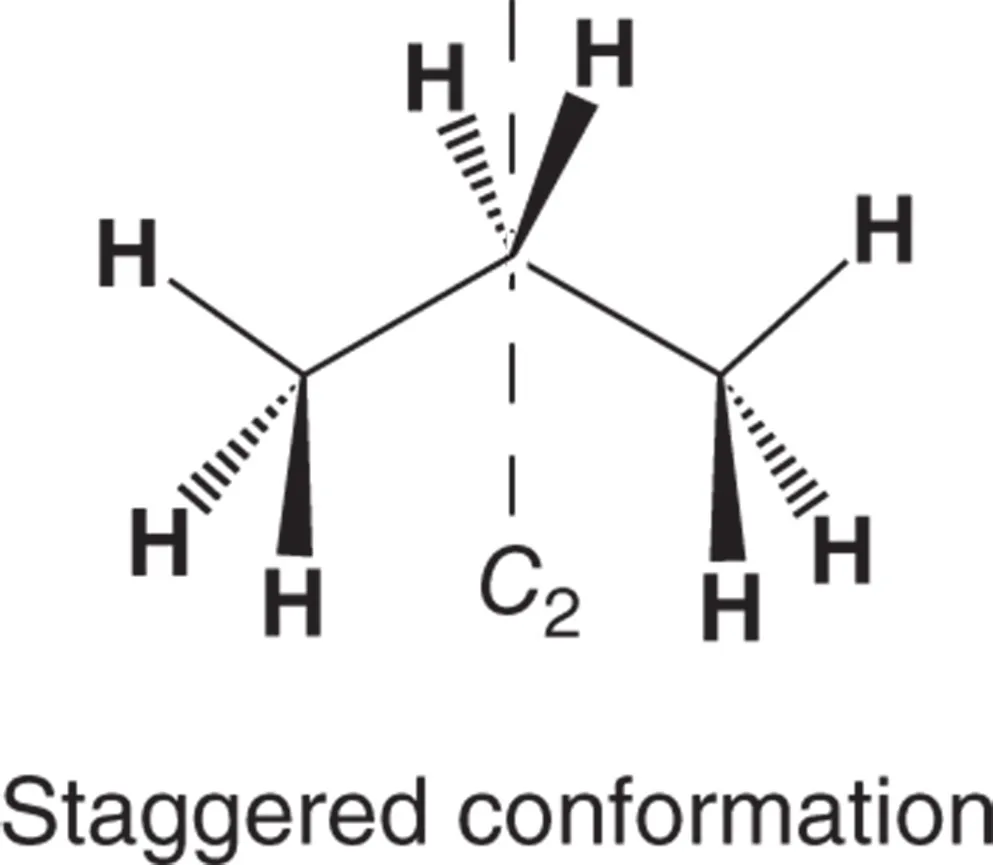
Provides references and answers to every question presented in the primary Organic Chemistry textbook
Successfully achieving chemical reactions in organic chemistry requires a solid background in physical chemistry. Knowledge of chemical equilibria, thermodynamics, reaction rates, reaction mechanisms, and molecular orbital theory is essential for students, chemists, and chemical engineers. The Organic Chemistry presents the tools and models required to understand organic synthesis and enables the efficient planning of chemical reactions.
This volume, Organic Chemistry: Theory, Reactivity, and Mechanisms in Modern Synthesis Workbook, complements the primary textbook—supplying the complete, calculated solutions to more than 800 questions on topics such as thermochemistry, pericyclic reactions, organic photochemistry, catalytic reactions, and more. This companion workbook is indispensable for those seeking clear, in-depth instruction on this challenging subject.
Written by prominent experts in the field of organic chemistry, this book:
- Works side-by-side with the primary Organic Chemistry textbook
- Includes chapter introductions and re-stated questions to enhance efficiency
- Features clear illustrations, tables, and figures
- Strengthens reader?s comprehension of key areas of knowledge
Organic Chemistry: Theory, Reactivity, and Mechanisms in Modern Synthesis Workbook is a must-have resource for anyone using the primary textbook.