For chemists, attempting to mimic nature by synthesizing complex natural products from raw material is a challenge that is fraught with pitfalls. To tackle this unique but potentially rewarding task, researchers can rely on well-established reactions and methods of practice, or apply their own synthesis methods to verify their potential. Whatever the goal and its complexity, there are multiple ways of achieving it. We must now establish a strategic and effective plan that requires the minimum number of steps, but lends itself to widespread use.
This book is structured around the study of a dozen target products (butyrolactone, macrolide, indole compound, cyclobutanic terpene, spiro- and polycyclic derivatives, etc.). For each product, the different disconnections are presented and the associated syntheses are analyzed step by step. The key reactions are described explicitly, followed by diagrams showing the range of impact of certain transformations. This set of data alone is conducive to understanding syntheses and indulging in this difficult, but worthwhile activity.

eBook - ePub
Retrosynthetic Analysis and Synthesis of Natural Products 1
Synthetic Methods and Applications
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Retrosynthetic Analysis and Synthesis of Natural Products 1
Synthetic Methods and Applications
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
1
Total Synthesis: Some Elements to Contemplate
1.1. Total synthesis – why and for what purpose?
Nature is an immeasurable source of rather complex molecules, which have always attracted the curiosity of chemists. Since Friedrich Wöhler prepared urea from ammonium cyanate in 1828, vital force theory has been challenged, allowing the synthesis of organic compounds from the simplest to the most complex, such as spongistatin 1 [HUD 07, HUA 18], and thereby the emergence of organic synthesis.
In less than two centuries, this branch of chemistry has reached a certain degree of maturity that has made it possible to prepare a considerable number of the most complex compounds, whose exact structures have been revealed as a result of the concomitant development of purification, analysis or characterization techniques (HPLC and UPLC, 1D and 2D NMR, mass spectrometry, X-ray diffraction, etc.).
The isolation and structural determination of natural products is most often guided by the existence of biological properties of interest to human health. In order to confirm or invalidate the proposed structures [NIC 05, MAR 17a] and also to identify the functional groups responsible for, or even essential to, these activities, it is necessary to carry out structure-activity studies. This assumes there are significant quantities of these natural compounds as well as different analogues with regard to their structure or even the spatial arrangement of these same functional groups; it is also one of the roles assigned to chemical synthesis [WIL 07]. Does this mean that organic synthesis becomes a simple tool available to researchers in the life sciences? No, of course not [HOF 13, NIC 18].
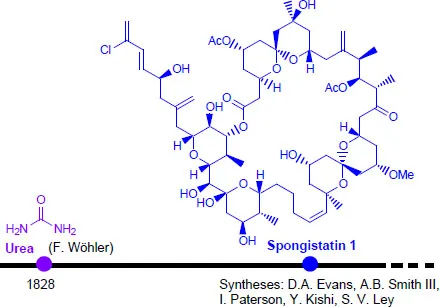
Figure 1.1. Evolution of target complexity in just over 150 years. For a color version of the figures in this chapter, see www.iste.co.uk/piva/analysis1.zip
With increasing ecological awareness, organic synthesis can also indirectly dispose of the required molecules, in order to preserve certain endemic species and their biotopes.
To minimize the impact of chemical synthesis on the environment, it is also important to develop new methods and concepts that produce less waste; this is all the more true since those generated using so-called fine chemistry are generally very numerous. This trend is commendable but remains difficult to achieve; the E factor, defined as the ratio of kg of waste per kg of product formed, is between 25 and 100 for the most advanced products (pharmaceuticals), while for base chemicals, this number is closer to 1 [SHE 07, ROS 15].
Table 1.1. Environmental impact factors across chemical industries
| Industry | Annual tonnage as products | Factor E | Annual tonnage as waste | Number of people of steps |
| Petrochemical | 106–108 t | 0.1 | 107 t | separation |
| Heavy chemistry | 105–106 t | <1–5 | 5.106 t | 1–2 |
| Fine chemistry | 100–104 t | 5–50 | 5.105 t | 3–4 |
| Pharmaceutical | 10–1000 t | 25–> 100 | 105 t | > 6 |
1.2. The different approaches
There are two distinct conflicting approaches: the approach developed in the academic world and that recognized in industry.
Of course, the effectiveness criterion, which can be correlated to the number of steps and overall performance, remains common to both.
For academic syntheses, in addition to these efficiency criteria, there is the primacy of experiment, which is often reflected in publications by the words “first total synthesis of...”. In many cases, to reach the target, teams use methodologies that they have developed themselves. The choice of target may be the result of:
- – direct application of this method;
- – interest in the compound’s properties, or its unique and even fascinating structure.
Last but not least, training young researchers through research is obviously a factor. A thesis in total synthesis undoubtedly makes it possible to “see” more chemistry and probably to test one’s character by being faced with a challenge at each stage [NIC 11].
The success and effectiveness criteria for the approaches implemented in industry are: cost reduction (in reagents and human resources), a minimum number of operations (for processing and purification), the reduction in associated waste and risks.
For complex targets, particularly those with several stereocenters, stereochemistry control can be performed:
- – By hemisynthesis: one of the most well-known cases is that of taxol, a diterpene initially isolated from the bark of Taxus baccata and known for its highly cytotoxic properties. To collect sufficient quantities, Françoise Guéritte and Pierre Potier’s team at the ICSN in Gif-sur-Yvette succeeded in isolating 10-deacetylbaccatin III from yew tree needles, a precursor with the same taxane skeleton; it was possible to regioselectively graft the appropriate side chain onto the hydroxy group at the C-13 position of the taxane skeleton [DEN 88, DAN 96].
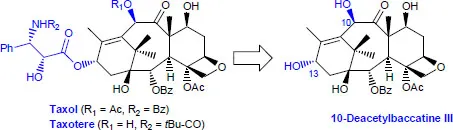 Figure 1.2. Taxol and Taxotere® from 10-deacetylbaccatin III
Figure 1.2. Taxol and Taxotere® from 10-deacetylbaccatin III - – According to a chiron approach: it is a question of benefiting from molecular platforms and carrying out various selective transformations. Stephen Hanessian is one of the researchers who has made the most out of this concept [HAN 12, BRI 17]. In the synthesis of dihydromevinolin, L-glutamic acid was used as a molecular block to prepare the lactone synthon with two stereogenic centers, which after several steps, gives rise to the key precursor of cycloaddition according to Diels–Alder [HAN 87, HAN 90].
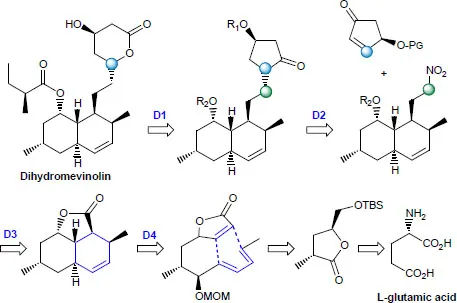 Figure 1.3. Dihydromevinolin from L-glutamic acid
Figure 1.3. Dihydromevinolin from L-glutamic acid
LEGEND OF FIGURE 1.3.–
- D1: ring enlargement (Baeyer–Villiger reaction).
- D2: introd...
Table of contents
- Cover
- Table of Contents
- Preface
- 1 Total Synthesis: Some Elements to Contemplate
- 2 Squamostolide
- 3 Rubrenolide
- 4 Bipinnatin J
- 5 Tubingensin B
- 6 Polygonatine A
- 7 (+)-Intricatetraol
- 8 Enigmazole A
- 9 Biyouyanagin A
- 10 Elatol
- 11 Thiomarinol H
- 12 Oblongolides A and C
- List of Abbreviations
- Index
- End User License Agreement
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Retrosynthetic Analysis and Synthesis of Natural Products 1 by Olivier Piva in PDF and/or ePUB format, as well as other popular books in Sciences physiques & Chimie analytique. We have over 1.5 million books available in our catalogue for you to explore.