While many clinicians currently recognize that bisphosphonate usage is associated with osteonecrosis of the jaws, this book establishes the causal relationship between the two, explaining the biologic modes of action that enable bisphosphonates taken both orally and intravenously to induce osteonecrosis of the jaws. Like the first edition, it presents definitive treatment protocols for patients who present at each stage in the progression of osteonecrosis, but this new edition also includes a simplified staging system and information about the serum CTX test for oral bisphosphonate cases. In addition, several management principles and surgical techniques are provided, along with new data on osteopenia and osteoporosis, the conditions for which bisphosphonates are primarily prescribed. The book offers a simple method for predicting risk as well as crucial recommendations for preventing the disease from developing when bisphosphonate therapy is indicated. Comprehensive case histories related to both oral and intravenous bisphosphonates provide direct guidance in managing patients spanning the full presentation spectrum. A must-have for the student and practicing clinician alike.

eBook - ePub
Oral and Intravenous Bisphosphonate–Induced Osteonecrosis of the Jaws
History, Etiology, Prevention, and Treatment, Second Edition
- 160 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Oral and Intravenous Bisphosphonate–Induced Osteonecrosis of the Jaws
History, Etiology, Prevention, and Treatment, Second Edition
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
1
Description and History of Bisphosphonate-Induced Osteonecrosis of the Jaws
What Is Osteonecrosis of the Jaws?
Bisphosphonate-induced osteonecrosis of the jaws refers to a condition characterized by exposure of bone in the mandible or maxilla persisting for more than 8 weeks in a patient who has taken or currently is taking a bisphosphonate and who has no history of radiation therapy to the jaws.1 However, while the exposed bone is indeed dead (osteonecrosis), bone death is actually a secondary result of bisphosphonate bone toxicity, which is similar to the genetic disease osteopetrosis, as explained in chapter 4. Clinically, the disease presents as exposed alveolar bone that occurs spontaneously (Fig 1-1a) or becomes evident following an invasive surgical procedure such as tooth removal (Fig 1-1b), periodontal surgery, apicoectomy, or dental implant placement. The disease manifests mostly in the jaws. To date, it has not been reported in other skeletal sites as exposed bone; however, recent reports have identified femur fractures caused by long-term use (6 years or more) of alendronate (Fosamax, Merck)2,3 (Fig 1-2). Osteonecrosis of the jaws always originates in the alveolar bone and may then extend to the basilar bone or ramus (Figs 1-3 and 1-4). Occasionally, early subclinical radiographic signs—including sclerosis of the lamina dura (Fig 1-5), loss of the lamina dura (Fig 1-6), and/or widening of the periodontal ligament space (Fig 1-7), particularly in association with molars—have been observed.
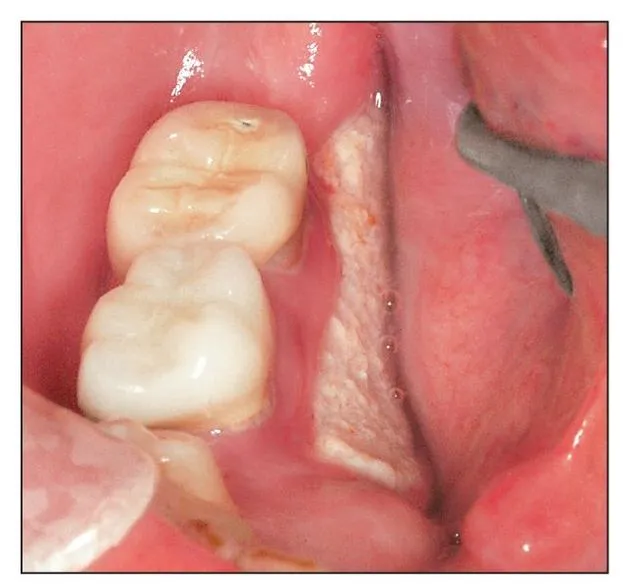
Fig 1-1a Spontaneous bone exposure of the lingual cortex in the mandibular molar region is a common presentation of bisphosphonate-induced osteonecrosis of the jaws.
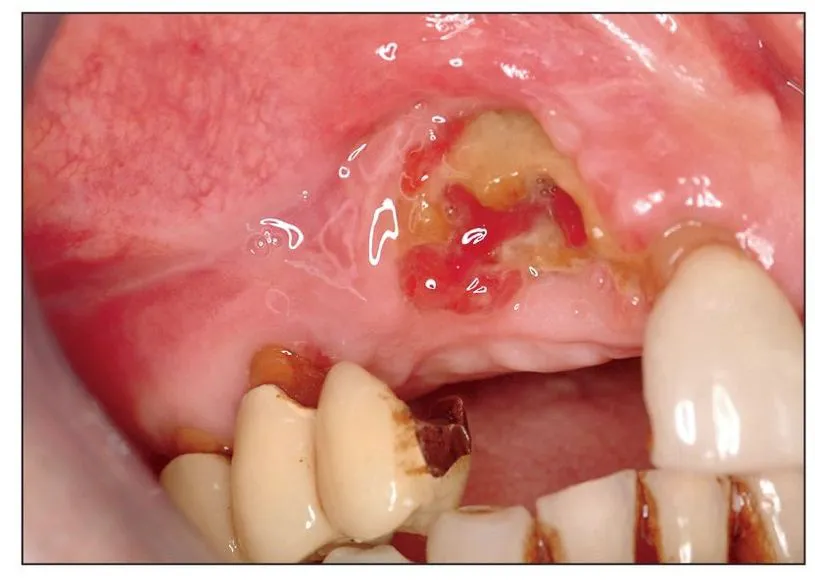
Fig 1-1b Nonhealing exposed bone developed after the removal of a maxillary central incisor and the adjacent lateral incisor.
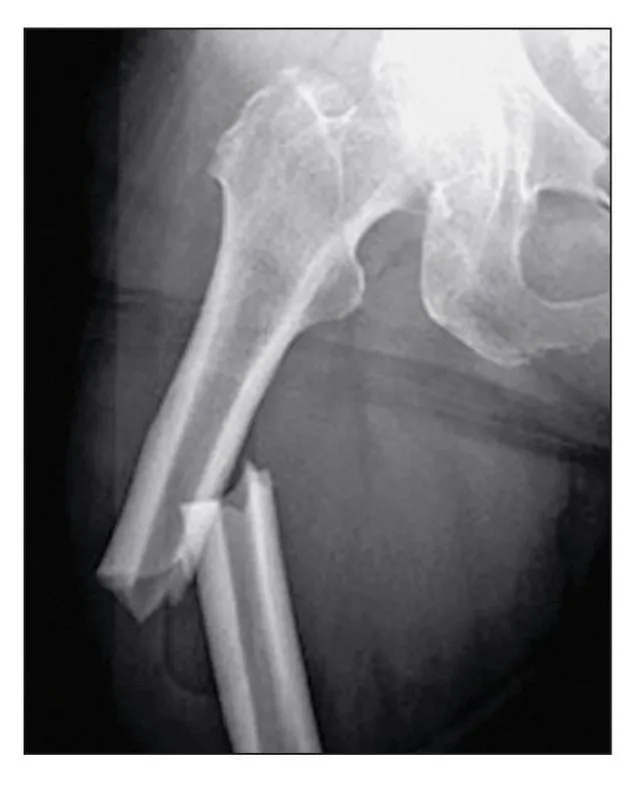
Fig 1-2 Atypical fracture of the femur caused by extended use of alendronate (Fosamax).
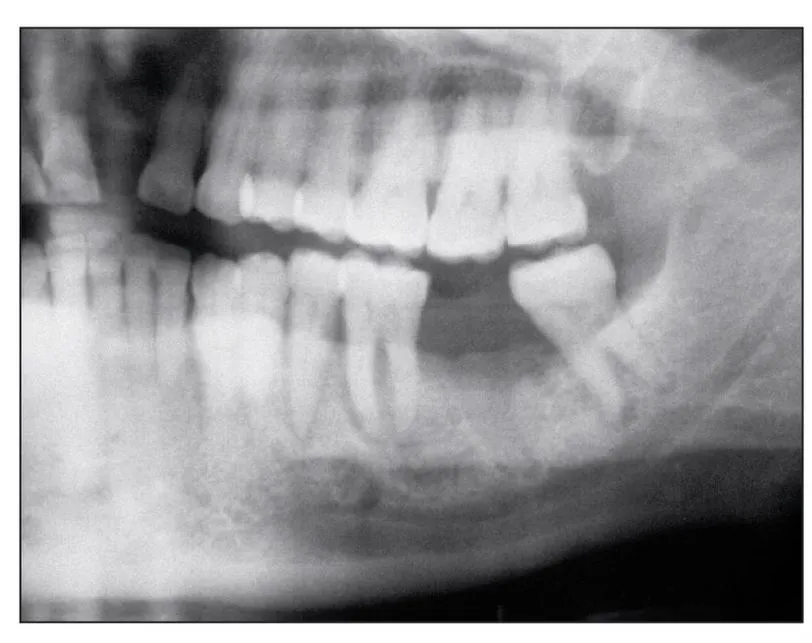
Fig 1-3a Radiographic osteosclerosis in the alveolar bone of the mandibular second molar socket.
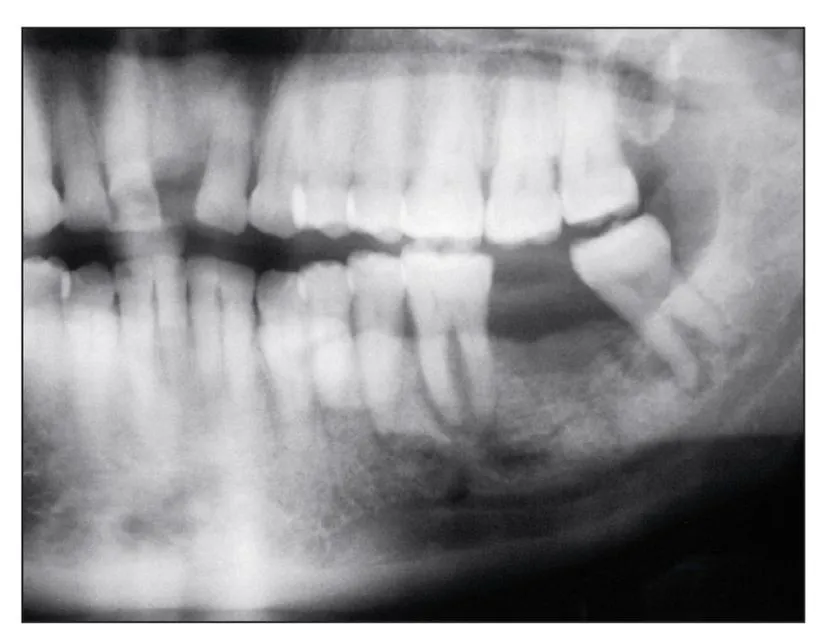
Fig 1-3b As bisphosphonate-induced osteonecrosis persists, it usually exhibits osteolysis and extends toward the inferior border.
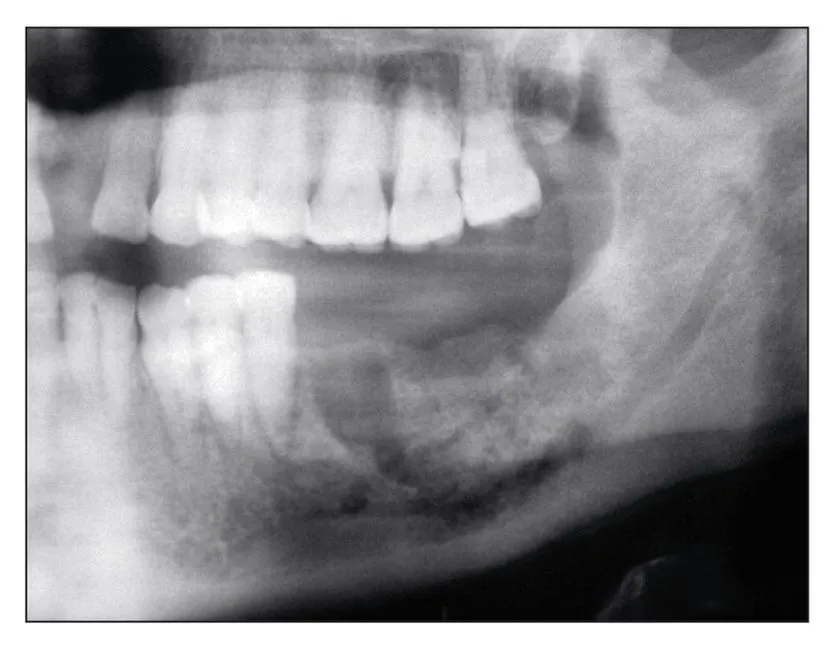
Fig 1-3c Persistent bisphosphonate-induced osteonecrosis. Osteolysis and osteosclerosis become more evident.
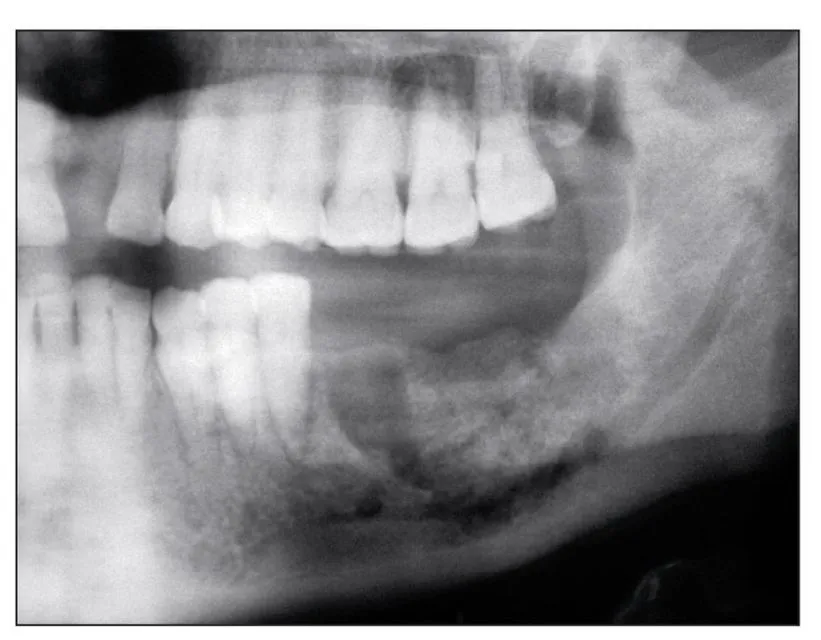
Fig 1-3d Further continuation of the bisphosphonate-induced osteonecrosis accompanied by secondary infection can extend into the inferior border, posing a risk of pathologic fracture.
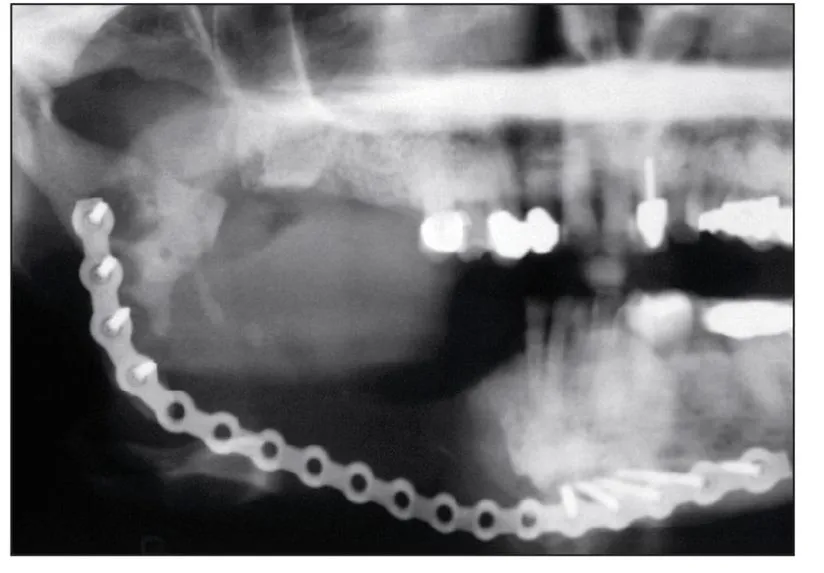
Fig 1-4 Even after a hemimandibular resection, evidence of bisphosphonate-induced osteonecrosis remains in the ramus and condylar neck.
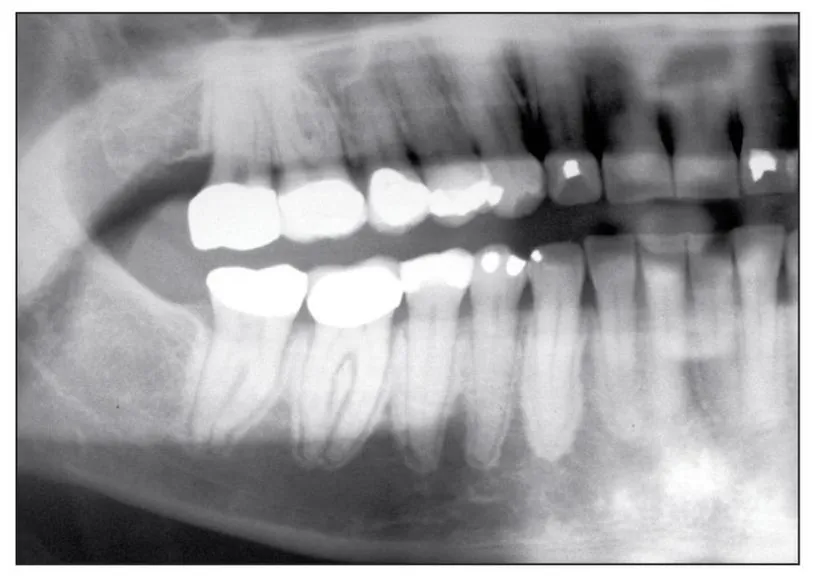
Fig 1-5 Sclerosis of the lamina dura may be seen before exposed bone develops; this is an early sign of bisphosphonate toxicity to the alveolar bone.

Fig 1-6 In some cases, loss of the lamina dura can be an early sign of bisphosphonate toxicity to the alveolar bone.
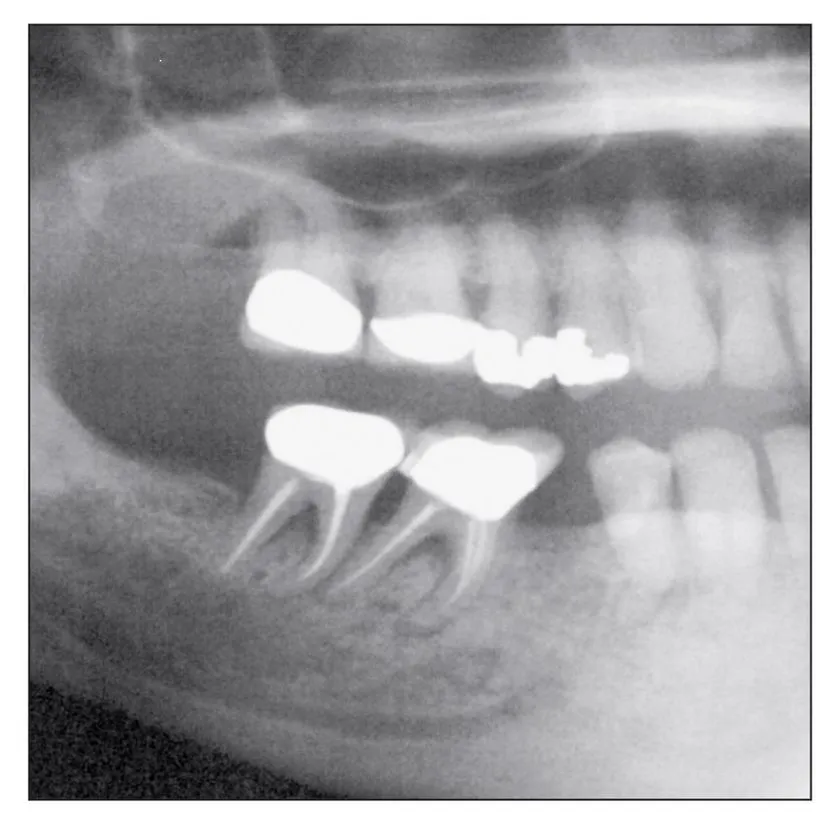
Fig 1-7 Widening of the periodontal ligament space may be an early or overt sign of bisphosphonate toxicity to the alveolar bone and may also be associated with significant osteolysis.
Early Reports of Bisphosphonate-Induced Osteonecrosis of the Jaws
Bisphosphonate-induced osteonecrosis of the jaws was first described by Marx and Stern in 2002.4 At that time, it was only a curious finding of exposed, nonhealing bone; when debridement was performed, the condition worsened and led to increased amounts of exposed bone. All of the patients described in that report were receiving pamidronate (Aredia, Novartis) for control of malignant tumor deposits in bone. The profession was first formally notified of this drug complication in a medical alert published by the author in 2003 in the Journal of Oral and Maxillofacial Surgery (JOMS), which described 36 cases associated with intravenous bisphosphonates (pamidronate or zoledronate [Zometa, Novartis]).5
Prior to the publication of this medical alert, representatives of Novartis, which produces the two intravenous bisphosphonates (pamidronate and zoledronate) that cause this form of osteonecrosis, were invited to examine 2 of the patients and to discuss the other 34 who were being seen. Although they expressed concern about the plight of these patients, the Novartis representatives denied the possibility that the osteonecrosis was in any way connected with their drugs, because no evidence of bone necrosis was found in their preclinical animal studies or in the more than 3,600 patients enrolled in their human clinical trials. They attributed the bone exposures to the chemotherapy these patients had received and possibly to the dexamethasone that had been given to about 55% of their patients. During that visit, the medical director of Novartis, Dr Peter Tarassoff, was given an advance copy of the medical alert. When it appeared in JOMS, Dr Tarassoff and his Novartis coauthors had already issued a retort strongly denying any causal relationship between the intravenous bisphosphonates produced by Novartis and the osteonecrosis observed in the jaws of these patients.6
The toxicity of chemotherapy was a natural and convenient culprit to blame. Indeed, the very issue of JOMS that carried the medical alert also carried a report by Wang et al of three cases of bone necrosis in the mandible that they too attributed to chemotherapy.7 Yet, it was reported that all three patients had been taking Aredia. Realizing their oversight, these authors later published a retraction article identifying Aredia as the cause of the exposed bone.8 Shortly afterward, compelling findings were reported in the November 2005 issue of JOMS, and three additional reports appeared in the Journal of the American Dental Association in December 2005.9–12 Since the original 2003 publication, more than 1,100 additional reports by over 4,500 authors and at least 14 position papers have been written about what is now accepted as bisphosphonate-induced osteonecrosis of the jaws (BIONJ).
Ironically, the strongest evidence to date of a cause-and-effect relationship between bisphosphonates and osteonecrosis of the jaws is provided by the results of a study Novartis conducted to gain marketing approval by the Food and Drug Administration (FDA). In this study, patients with bone metastasis received either their normal chemotherapy alone or chemotherapy plus a steroid regimen and an intravenous bisphosphonate (either Aredia or Zometa). Patients with the same malignancies but without bone metastasis also received their normal chemotherapy and an identical steroid regimen, but they were not given intravenous bisphosphonates. Interestingly, only those patients who received an intravenous bisphosphonate ever developed exposed bone. This amounted to one of the best randomized, controlled, and truly double-blinded studies in the history of medicine or dentistry.
Flaws in the original study by Novartis are largely the cause of so much disbelief. Their claim that their animal studies failed to show osteonecrosis is likely due to the fact that animal bone physiology is extremely resistant to chemical and physical injury. The profession of oral and maxillofacial surgery has sought to produce a reliable animal model for osteoradionecrosis—its historical nemesis—for four decades. Yet with each attempt, either exposed bone could not be produced or the radiation dose was increased to the point of killing the study animal. In their human clinical trials, Novartis claimed that none of the 3,600+ patients developed exposed bone. This is false; the truth is, they never thought to look for exposed bone in the mouth. Oral examinations before or after intravenous bisphosphonate therapy were never conducted, nor was a dentist or an oral and maxillofacial surgeon...
Table of contents
- Cover
- Title Page
- Copyright Page
- Table of Contents
- Dedication
- Preface to the Second Edition
- 1 - Description and History of Bisphosphonate-Induced Osteonecrosis of the Jaws
- 2 - Modes of Action and Pharmacokinetics of the Bisphosphonate Family
- 3 - Medical Indications for Bisphosphonates
- 4 - Osteopetrosis: A Genetic Analogue to Bisphosphonate-Induced Osteonecrosis of the Jaws
- 5 - Risks, Prevention, and Management of Intravenous Bisphosphonate–Induced Osteonecrosis
- 6 - Risks, Prevention, and Management of Oral Bisphosphonate–Induced Osteonecrosis
- 7 - Clinical Pathologic Case Samples
- Glossary
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Oral and Intravenous Bisphosphonate–Induced Osteonecrosis of the Jaws by Robert E. Marx in PDF and/or ePUB format, as well as other popular books in Medicine & Dentistry. We have over 1.5 million books available in our catalogue for you to explore.