
- 479 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
Imaging in Photodynamic Therapy
About this book
This book covers the broad field of cellular, molecular, preclinical, and clinical imaging either associated with or combined with photodynamic therapy (PDT). It showcases how this approach is used clinically for cancer, infections, and diseases characterized by unwanted tissue such as atherosclerosis or blindness. Because the photosensitizers are also fluorescent, the book also addresses various imaging systems such as confocal microscopy and small animal imaging systems, and highlights how they have been used to follow and optimize treatment, and to answer important mechanistic questions. Chapters also discuss how imaging has made important contributions to clinical outcomes in skin, bladder, and brain cancers, as well as in the development of theranostic agents for detection and treatment of disease. This book provides a resource for physicians and research scientists in cell biology, microscopy, optics, molecular imaging, oncology, and drug discovery.
Tools to learn more effectively

Saving Books

Keyword Search

Annotating Text

Listen to it instead
Information
PART 1
INTRODUCTION
1
Looking out the optical window
1.1 INTRODUCTION
1.2 PHYSICAL PRINCIPLES: LIGHT PROPAGATION AND INTERACTIONS WITH MATTER
1.2.1 LIGHT AS WAVE AND PARTICLE
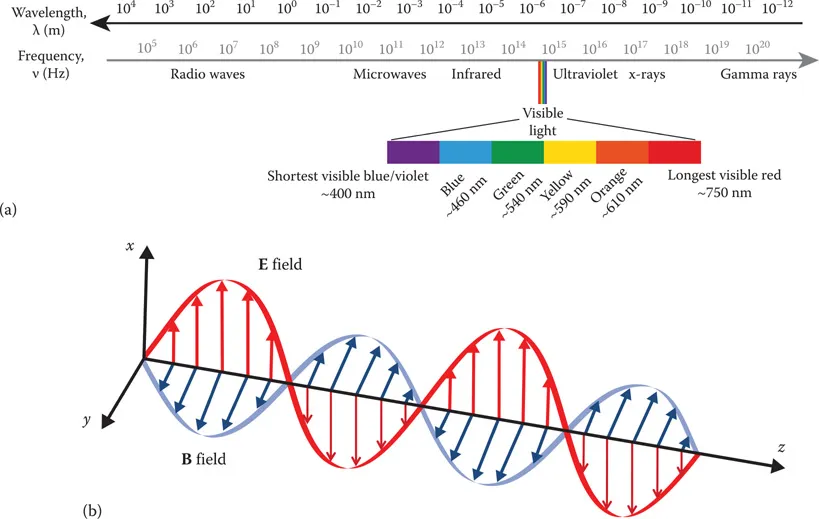
1.2.2 PHASE AND COHERENCE
Table of contents
- Cover
- Half Title
- Title Page
- Copyright Page
- Dedication Page
- Table of Contents
- Series preface
- Preface
- Acknowledgment
- Editors
- Contributors
- PART 1 INTRODUCTION
- PART 2 IN VITRO MICROSCOPY FOR PHOTOSENSITIZER LOCALIZATION IN CELLS
- PART 3 IN VITRO MICROSCOPY OF CELL DAMAGE AND DEATH PROCESSES AFTER PDT
- PART 4 THERANOSTIC AGENTS AND NANOTECHNOLOGY
- PART 5 SMALL ANIMAL IMAGING
- PART 6 CLINICAL IMAGING
- Index
Frequently asked questions
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app