![]()
1
Organic Light-Emitting Devices and Their Applications for Flat-Panel Displays
Qing Wang, Gang Yu, and Jian Wang
CONTENTS
1.1 Introduction
1.2 Conjugated Polymers in PLEDs
1.3 PLED Structures, Processes, and Performance
1.4 Novel Devices and Novel Functions in Thin-Film Polymer Devices
1.4.1 Dual-Function Polymer Device and Display Matrices
1.4.2 Polymer Light-Emitting Electrochemical Cells
1.4.3 PLED with Stable Cathode Electrode
1.4.4 Highly Efficient White PLEDs
1.4.5 PLED and PLEC in Surface Cell Configuration
1.4.6 Optocouplers Made with Semiconducting Polymers
1.5 Flat-Panel Displays Made with Solution-Processable Organic Semiconductors
1.5.1 SMOLEDs/PLEDs as Emitter Elements in Flat-Panel Displays
1.5.2 PMOLED Displays vs. AMOLED Displays
1.5.3 Monochrome AMPLEDs Made with Solution-Processable Polymers
1.5.4 Full-Color AMPLED Modules
1.5.5 Performance Simulation for FC AMOLEDs
1.5.6 AMOLED for Graphic and Motion Picture Applications
1.6 Summary and Remarks
Acknowledgment
References
1.1 Introduction
The electroluminescence (EL) phenomenon was first discovered in a piece of carborundum (SiC) crystal, by H.J. Round in 1907.1 Commercial research into light-emitting diode (LED) technology started in the early 1960s, when Nick Holonyak, Jr., created the first inorganic LED in 1962.2,3 Work on gallium arsenide phosphide (GaAsP) led to the introduction of the first mass-produced commercial 655-nm red LEDs in 1968, by Hewlett-Packard and Monsanto. In the 1950s, Bernanose first observed EL in organic material by applying a high-voltage alternating-current field to crystalline thin films of acridine orange and quinacrine.4,5 The direct current–driven EL cell using single crystals of anthracene was first demonstrated by Pope and his coworkers after the discovery of LEDs made with III–V compound semiconductors.6 In 1975, the first organic EL devices made with a polymer, polyvinyl carbazole (PVK), were demonstrated.7
In early attempts to develop organic EL devices, the driving voltage of such devices was on the order of 100 V or above to achieve a significant light output.8,9,10 Vincett et al. achieved an operation voltage of <30 V by using a thermally deposited thin film of anthracene.11 The research had been mainly in the academic field until Dr. C.W. Tang and his coworkers at Kodak Chemical showed, for the first time, efficient organic light-emitting devices (OLEDs) in multilayer configuration with significant performance improvement.12 Nowadays, small-molecule OLEDs (SMOLEDs) made by means of a thermal deposition process have been used for commercial display products. Pioneer Corporation has commercialized organic EL (OEL) display panels since 1999 for consumer electronics use, such as in car audio, CD/MP3 player, A/V receiver, etc. Kodak and Sanyo Electric Co. Ltd. demonstrated the first full-color (FC) 2.4-inch (2.4″) active matrix (AM) SMOLED display in 1999. Their joint manufacturing venture, SK Display Corp., produced the world’s first AM SMOLED displays for Kodak’s EasyShare LS633 camera.13 The world’s first commercially available OLED TV is SONLY XEL-1 with an 11″ size, released by SONY Corporation in 2007.14 Nowadays, AM SMOLED displays have become ubiquitous in smartphones. In early 2013, LG started selling 55″ OLED TVs worldwide.15
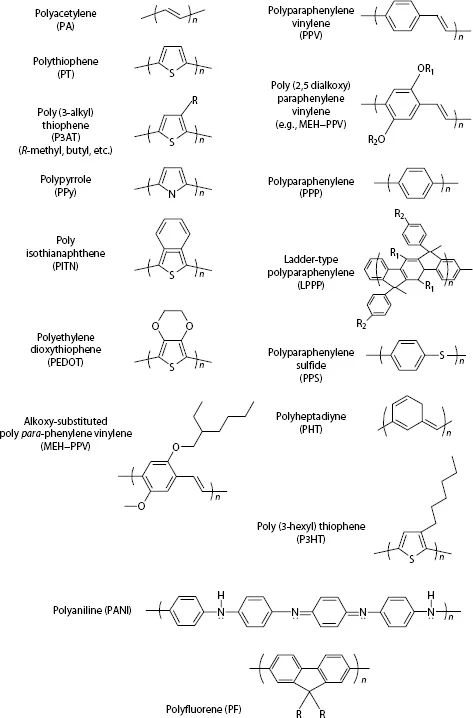
Another type of organic semiconductor, conjugated polymer, was discovered in 1977 by Alan J. Heeger, Alan G. MacDiarmid, and Hideki Shirakawa.16,17 In addition to the focus on its novel physical and chemical properties in heavily doped states, great attention was paid to its intrinsic properties in undoped semiconducting phase, its nonlinear optical properties under photoexcitation,18,19 and its interfacial behaviors with metal contacts. Schottky diodes made with polyacetylene film were demonstrated in a metal–semiconductor polymer–metal configuration.20,21 Their optoelectric and electro-optical properties were studied. Although significant photosensitivity was demonstrated, the EL property of this system was intrinsically weak because of its electronic structure. Considerable works in the early and middle 1980s in the field of conjugated polymer were done on searching and developing new materials with solution processability. A popular, well-studied system consisted of polythiophene derivatives, one of which was poly(3-alkyl) thiophene (P3AT) (Figure 1.1). Solution-processed metal/P3AT/metal thin-film devices were demonstrated at the University of California at Santa Barbara in 1987.22 Following the first demonstration of a light-emitting device with unsubstituted poly(para-phenylene vinylene) (PPV) (Figure 1.1) by Burroughes et al. at Cambridge University,23 a highly efficient polymer LED (PLED) device was made with a solution-processable polymer, poly(2-methoxy-5-(2′-ethyl-hexyloxy)–1,4-phenylene vinylene), MEH–PPV (Figure 1.1), by Alan J. Heeger’s group in Santa Barbara, California.24 As will be discussed in later chapters, the commercialized soluble PPV derivatives nowadays are based on a synthesis approach originally developed by Fred Wudl’s group in Santa Barbara in 1988,25,26 and later modified by UNIAX Corporation (now DuPont Displays) in the middle 1990s and Aventis Research & Technologies GmbH (which later became Covion Organic Semiconductors GmbH, and now is part of Merck) in the late 1990s. Soluble PPV derivatives synthesized following this approach not only have high molecular weight but also show excellent solubility in common organic solvents. Most importantly, these materials have intrinsically low charged impurity, typically <1014 cm−3, and high photoluminescent efficiency (typically in the range of 20–60%).25,26,27,28 PLEDs made with such PPV films show high EL efficiency, low operation voltage, and long device lifetime.29,30,31 Displays made with PPV emitters were commercialized in 2002 by Philips (Norelco electric razor: Spectra 8894XL).
FIGURE 1.1
Chemical structures of popular conjugated polymers.
Although the energy gap in a PPV derivative can be enlarged by reducing the conjugation and planarization between the phenyl group and the vinyl group (as observed in PPVs with phenyl groups attaching at 2- or 5- or both sites),27,32,33 it is not large enough to produce the saturated blue color needed for FC displays. Conjugated polymers with an optical energy gap of >2.9 eV are needed for PLEDs with blue emission. Significant effort has been made on searching and developing wide-energy-gap polymers (such as poly (p-phenyl) and its functional derivatives).34,35,36,37,38,39,40,41,42,43,44,45,46 In addition to being used for making blue emitters, the same building blocks can also be used for making red and green emitters (as the host) by copolymerizing them with a proper emitter group (as the guest).47,48,49 The red, green, and blue material sets developed by several companies (including Covion and Dow Chemical) are all soluble in common organic solvents with high optoelectric performance and good film-forming properties.49,50
PLED-based displays are attractive owing to their processing advantages in device manufacture. The organic materials used are soluble in common organic solvents or in water. Large-size, uniform, and pinhole-free thin films can be cast from solutions at room temperature by means of spin coating or other coating techniques commonly seen in the printing and painting industries. Because of the characteristic large elongation at rupture of polymers, they are flexible and easily fabricated onto rigid or flexible ...