Contents
6.1 Overview
6.2 Photocurrent Conversion Processes
6.3 Photogenerated Carrier Dynamics in the Vicinity of Donor/Acceptor Interfaces
6.3.1 Introduction
6.3.2 Impedance Spectroscopic Study of Heterojunction OPV Cells
6.4 Structural Effects of Organic Films on Exciton and Carrier Dynamics
6.4.1 Introduction
6.4.2 Effects of Intermolecular Charge-Transfer Excitons on the External Quantum Efficiency for Heterojunction OPV Cells
6.5 The Correlation between Open-Circuit Voltage and Photogenerated Carrier Dynamics
6.5.1 Introduction
6.5.2 Influence of Charge Accumulation of Photogenerated Carriers in the Vicinity of Donor–Acceptor Interface on the Open-Circuit Voltage of Heterojunction OPV Cells
6.6 Concluding Remarks and Perspectives
References
6.1 Overview
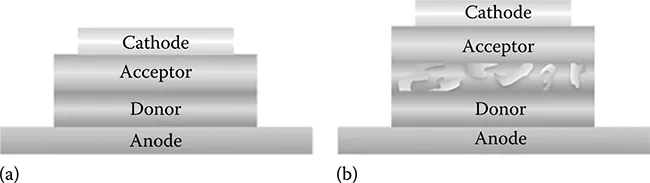
Understanding the photoexcited carrier dynamics in organic photovoltaic (OPV) cells helps us to improve their power conversion efficiency (η) for practical use because some breakthroughs in the recent decade have been based on bulk-heterojunctions (BHJs) between donor (D) and acceptor (A) materials. The BHJ plays a role of increasing the D-A interface area involving photocarrier generation regions, and thus the number of photocarriers increases at the interfaces [1,2]. Although the BHJ can improve the η of OPV cells by up to ca. 8%, the improving rate of η does not significantly increase [3–5]. This is presumably because there are still major unclear points for BHJ as follows: (1) the nanostructure of the BHJ (although its schematic illustration has been shown) and (2) the reproducibility of BHJ fabrication. Figure 6.1 schematically illustrates typical structures of (a) a D-A hetero double-layered OPV cell and (b) a BHJ-OPV cell. As shown in Figure 6.1b, because the BHJ is fabricated by coevaporation of donor and acceptor materials or by spin-coating conductive polymers, the D-A interface thus formed is too complex to analyze and reproduce the nanostructure compared to that of a double-layered heterojunction [2,6]. These facts make it difficult to discuss the photoexcited carrier dynamics at the BHJ D-A interface. Accordingly, the reason why the BHJ improves the η has been unclear so far.
FIGURE 6.1 Schematic illustration of typical structures used for organic photovoltaic cells: (a) heterojunction type and (b) BHJ type.
In addition, both the electronic states (corresponding to a built-in potential, etc.) at the D-A interface under illumination conditions and the origins of open circuit voltage (Voc), which are related to the carrier dynamics, have also remained unclear [7–11]. For these reasons, we have not obtained any decisive breakthroughs to develop the OPV cells commercially available.
In the present chapter, we will discuss photogenerated carrier dynamics, electronic states at the D-A interface under illumination conditions, and the origins of Voc, using an OPV cell [ITO/Zn(OEP)/C60/Al] with a D-A hetero double-layered structure. Here, ITO, Zn(OEP), and Al express indium-tin-oxide, zinc octaethylporphyrin, and aluminum, respectively.
Contrary to the BHJ-OPV cells with a complex D-A interface, an OPV cell with a simple D-A hetero double-layered structure is more preferable to discuss the photo-excited carrier dynamics and to clarify what factors significantly contribute to the η because experimental results thus obtained are easy to be analyzed on the basis of the simple structured OPV cells.
6.2 Photocurrent Conversion Processes
As shown in Figure 6.1, the device structure of the present OPV cell has only one D-A interface. The Fermi level (EF), the vacuum level (VL), and the energy levels of the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) for both donor and acceptor films play an important role for the generation rate of excitons, carriers (electrons and holes), and current in OPV cells under photoirradiation [12].
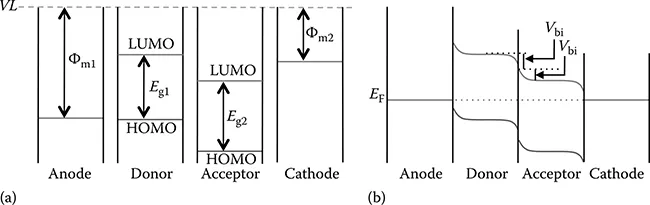
Figure 6.2 illustrates an energy-level alignment based on the Mott-Schottky model before and after the anode, donor, acceptor, and cathode films contact each other [12–14]. Here, Φm and Eg denote a work function of individual electrodes and an energy gap of the D and A materials, respectively.
FIGURE 6.2 Schematic representation of the energy-level alignment based on the Mott-Schottky model: (a) before and (b) after the contact of individual films.
As shown in Figure 6.2, when neutral organic and metal films are separated from each other, the energy diagram of each film is aligned using the common VL. After the contact of individual films, their EF levels equilibrate to be the same due to charge transfer (either electrons or holes) at each interface in association with a band bending [15]. This band bending induces an electrical field (in other words, generates a built-in potential: Vbi) in the vicinity of the individual interfaces for the OPV cell. However, these interfacial electronic structures after the contact have not yet been understood precisely. Indeed, another alignment model considering the VL shift at a metal/organic interface has been reported recently [16].
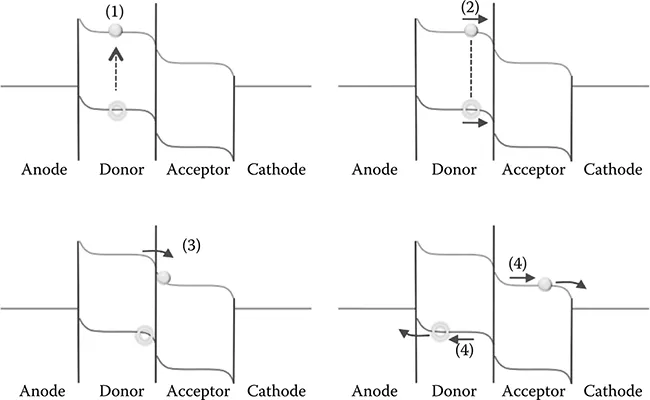
As shown in Figure 6.3, the fundamental mechanism of the photogenerated current in OPV cells has been considered as the following four elementary processes: (1) excitons are generated by photoabsorption of a donor film, (2) excitons thus formed diffuse to the D-A interface, (3) excitons are dissociated to electrons and holes at the D-A interface, and (4) electrons and holes are, respectively, moved to and collected at each electrode.
FIGURE 6.3 Four elementary processes of photogenerated current in OPV cells.
For the first process (1), excitons are generated by photoabsorption of donor films. Here, the exciton is defined to be an electron-hole pair via Coulombic attractive force, which can be classified into the Mott-Wannier and Frenkel types. For the former type, the electron-hole interaction is weak, which results in the exciton radius being much larger than the lattice constant [17–19]. In this case, the binding energy of this type of exciton is typically in the order of 0.1 eV. On the other hand, for the latter type, the Coulombic interaction is very strong, and the exciton tends to have a radius on the same order as the unit cell. Thus, the electron and hole are localized in the same unit cell. In this case, the binding energy of the Frenkel-type exciton is in the order of 1.0 eV [20]. In addition, if a donor film consists of small organic molecules, excitons are further categorized into inter- and intramolecular type excitons [21].
For the second process (2), the excitons thus generated diffuse in the organic donor film to the D-A interface. The exciton diffusion length corresponding to the moving distance for its lifetime has been reported to be 5–50 nm [20] for organic donor films, which depends on the crystal structure, molecular orientation of organic films, and the exciton types [6]. Accordingly, the exciton diffusion length plays an important role of determining a thickness of organic donor film because excitons generated in a region with a distance to the D-A interface longer than the exciton diffusion length disappear by electron-hole recombination prior to reaching to the interface. Because the exciton diffusion length is typically shorter than the film thickness of 50–100 nm for OPV cells, this physical quantity significantly affects the photocurrent conversion efficiency [12].
For the third process (3), excitons are separated to electrons and holes at the D-A ...