![]()
CHAPTER 1
Antibiotic resistance: The unfolding crisis
Ramanan Laxminarayan
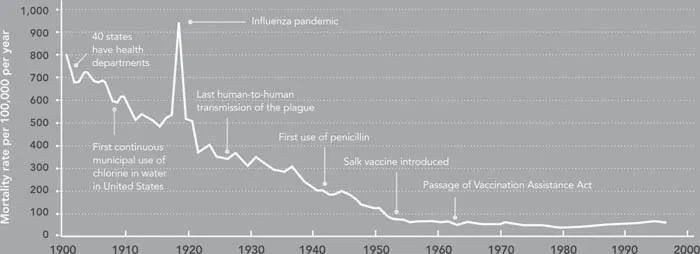
The introduction of penicillin in 1941 is among the most significant technological advances in modern medicine. Although many improvements in public health and medicine and a decline in infectious disease mortality preceded the introduction of penicillin, antibiotics have made possible further reductions in deaths and disability from infectious disease (Figure 1.1). Perhaps equally important, they have facilitated the vast expansion of other medical interventions, such as kidney and heart transplants, by allowing clinicians to prevent surgical site infections and infections in immuno-suppressed patients, such as organ recipients. Now, growing levels of bacterial resistance to antibiotics threaten our ability not just to treat infectious diseases but also to perform other procedures and treatments that fundamentally depend on affordable and effective antibiotics.
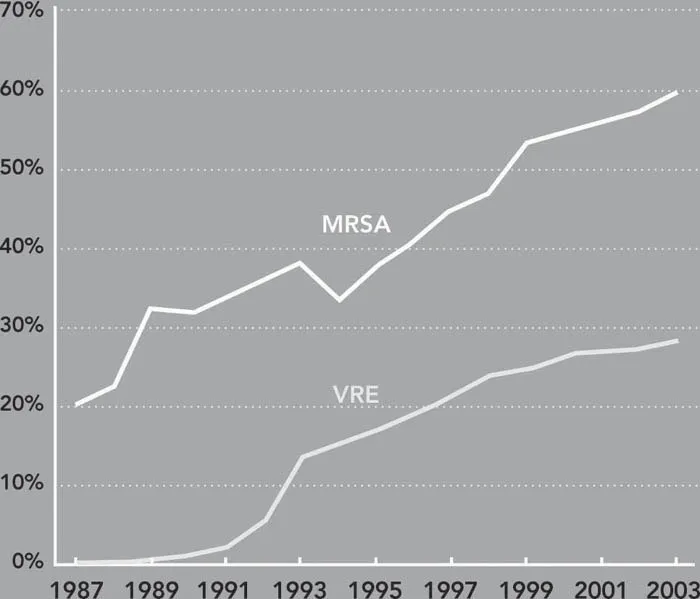
The timeline of emergence of drug resistance is best illustrated by the case of Staphylococcus aureus (S. aureus), a common pathogen that causes life-threatening infections and is transmitted in both health care and community settings. The mortality rate from a S. aureus infection was as high as 82 percent in the preantibiotics era (Skinner and Keefer 1941) but fell dramatically after the introduction of penicillin. Resistance to penicillin emerged soon after its introduction and was linked to patient deaths in the early 1950s (Abboud and Waisbren 1959). In 1960, penicillin was replaced with a beta-lactam compound, methicillin, which was effective against penicillin-resistant S. aureus, but methicillin-resistant S. aureus (MRSA) emerged in the 1970s in Europe and soon after in the United States. MRSA prevalence in U.S. hospitals, which was 2.4 percent in 1975, increased to 29 percent in 1991 (Archer 1998) (Figure 1.2), and 59.5 percent in 2003 (CDC 2004), growing at an average rate of more than 12 percent per year. Vancomycin is the main and potentially last available drug that can reliably treat MRSA infections, and the massive use of vancomycin for treating MRSA is believed to be an important reason for the emergence and spread of vancomycin-resistant enterococci (VRE) (Weinstein 2003). Meanwhile, strains of MRSA resistant to vancomycin have been detected, providing the first glimpse of medical outcomes in a post-antibiotics era (Chang, Sievert et al. 2003).
Figure 1.1 Crude infectious disease mortality rate in the United States, 1900–1996
Source: Adapted from Armstrong, Conn et al. (1999).
Figure 1.2 The proportion of methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant nterococcal (VRE) infections is increasing (1987–2003)
Sources: VRE and MRSA data, 1998–2000, 2002–2003 (CDC 1999; CDC 2000; CDC 2001; CDC 2003; CDC 2004); data for 2001 are the average of 2000 and 2002 data. MRSA data from 1987–1997 are estimated from (Lowy 1998). VRE data for 1989 and 1993 are from (CDC 1993). VRE data for 1990–1992 and 1994–1997 are interpolated based on geometric mean.
The fast evolution of S. aureus from a bacterium that was easily treatable at pennies a dose to a pathogen that now requires powerful, expensive antibiotics is paralleled by other predominantly hospital-acquired infections, like those caused by VRE, Enterobacter and Pseudomonas aeruginosa. In each case, the ability to treat bacterial infections has been rolled back by the evolution of resistance. In this chapter, we describe the medical and economic impacts of resistance and explore why drug resistance is a compelling problem that, if left unaddressed, has the potential to derail the health care system by returning us to a world where children, the elderly, and other vulnerable populations routinely die from simple bacterial infections.
Trends in resistance
The pathogens that are transmitted in hospitals and communities are different in their ecology and epidemiology, as explained in Chapter 2. Drug resistance is growing in both types of pathogens. Common hospital-acquired, or nosocomial, infections include Gram-positive infections, such as those caused by S. aureus and enterococci, whose resistance has been increasing at a rapid pace. In 1998, MRSA was detected among patients without recent health care exposure or other predisposing risk factors (Herold, Immergluck et al. 1998), and it has since become an important threat to community health.
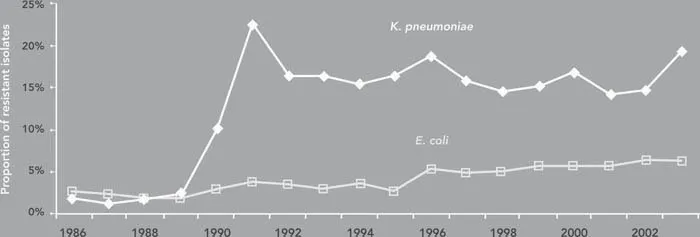
Resistance has become a serious problem among hospital-acquired Gram-negative pathogens, such as Escherichia coli, Acinetobacter baumannii, Klebsiella pneumoniae, and Pseudomonas aeruginosa, as shown in Figure 1.3 (Gaynes and Edwards 2005). Gram-negative pathogens are even more challenging than MRSA because there are fewer antibiotics available to treat infections caused by them.
Figure 1.3 Resistance to third-generation cephalosporins among Klebsiella pneumoniae and E. coli, 1986–2003
Source: Adapted from Gaynes and Edwards (2005).
Comparison with other countries
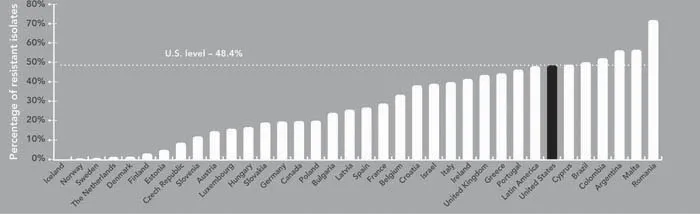
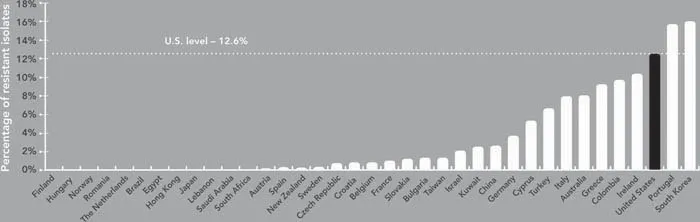
Hospitals in the United States have among the highest rates of MRSA in the world: on average 60 percent of patients infected with S. aureus in intensive care units of U.S. hospitals cannot be treated with methicillin or older antibiotics (CDC 2004). Surveillance for drug-resistant hospital-acquired infections has been less successful than in Europe, where a concerted effort has been made to identify antimicrobial rates in both hospital and community settings. Figure 1.4 shows MRSA rates for the United States and other high- and middle-income countries for 2004. In Europe, only Romania and Malta had higher rates of MRSA than the United States in that year. MRSA levels were high in East Asia, specifically South Korea, Japan, and Taiwan, probably because of high levels of antibiotic use, but not much higher than for the United States. In the Americas, only Argentina, Brazil, and Colombia had a higher MRSA prevalence than the United States.
Figure 1.4 Methicillin-resistant Staphylococcus aureus infection rates in the United States and other countries
Sources: Canada and United States, 2000–2002 (Jones, Draghi et al. 2004); Latin America, 1998 (Diekema, Pfaller et al. 2000); Brazil, 1998 (Melo, Silva-Carval-ho et al. 2004); Colombia, 2001–2002 (Arias, Reyes et al. 2003); Argentina, 2002 (Bantar, Famiglietti et al. 2004); European countries, 2004 (RIVM 2005).
Prevalence of vancomycin-resistant enterococci (VRE) in U.S. hospitals is estimated to be roughly 12 percent on average across all hospital patients (McDonald 2006) and according to the CDC is more than 28 percent in intensive care units (CDC 2004). VRE rates in the United States and other countries are shown in Figure 1.5. In Europe, only Portugal had a higher prevalence of VRE than did the United States. Data on VRE prevalence outside Europe are less reliable but show lower rates than the United States, with the exception of South Korea. Reliable studies from Japan have found isolated outbreaks but no evidence of VRE transmission (Arakawa, Ike et al. 2000; Matsumoto, Muratani et al. 2004).
Figure 1.5 Vancomycin-resistant enterococci rates in the United States and other countries
Sources: Brazil, 2002 (Titze-de-Almeida, Filho et al. 2004); Egypt, Lebanon, Saudi Arabia, South Africa, and Turkey, 2001–2002 (Bouchillon, Johnson et al. 2004); Hong Kong, 2000 (Ho 2003); Japan, 2000 (Arakawa, Ike et al. 2000); New Zealand, 2000 (Briggs, Upton et al. 2002); Taiwan and United States, 2000 (McDonald, Lauderdale et al. 2004); Kuwait, 1999–2001 (Udo, Al-Sweih et al. 2003); Australia, 1999 (Nimmo, Bell et al. 2003); Colombia, 2001–2002 (Arias, Reyes et al. 2003); China (Liu, Xu et al. 2003); South Korea, 2002 (Lee, Kim et al. 2004); European countries, 2004 (RIVM 2005).
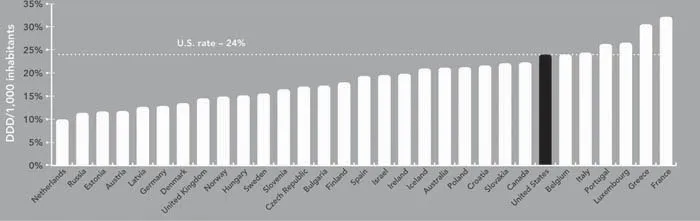
One reason for the higher prevalence of resistance in the United States may be the higher levels of antibiotic prescribing in this country (see Box 2.5, Chapter 2). Although antibiotic prescribing has fallen in the United States since 1994, it remains among the highest in the world (Steinman, Gonzales et al. 2003). Data from the European Surveillance of Antibiotic Consumption (Goossens, Ferech et al. 2003) show prescribing rates for most countries from 1997 to 2002 (Figure 1.6). The prescribing rate in the United States was 24 defined daily doses per 1,000 population per year. Only five countries—France, Luxembourg, Italy, Greece, and Portugal—had higher rates, but some of these countries had lower rates of resistance than the United States, indicating that there may be other casual factors.
Figure 1.6 Antibiotic prescribing rates for the United States and other countries
Sources: Canada, Australia, and United States, 1994 (McManus, Hammond et al. 1997); Russia, 1998 (Cizman, Beovic et al. 2004); Australia, 2002 (National Prescribing Service 2005); European countries, 2004 (Goossens, Ferech et al. 2003).
Health impact of drug resistance
Patients who have a hospital infection have a lower probability of survival (Osmon, Warren et al. 2003), as shown in Figure 1.7. This survival disadvantage is worse if the infection is due to a drug-resistant pathogen. Studies have s...