![]()
Chapter 1
Thermal comfort and assessment
Nomenclature
| A | area (m2) |
| ASHRAE | American Society of Heating Refrigeration and Air Conditioning Engineers |
| Clo | unit of thermal resistance of clothing (m2K/W) |
| dt | temperature difference (K) |
| ehr | heat transfer coefficient for radiation (W/m2K) |
| emf | electromotive force (volts) |
| h | height (m) |
| hc | heat transfer coefficient for convection (W/m2K) |
| m | mass (kg) |
| Met. | metabolic rate (W/m2) of body surface |
| mwet | mean weighted enclosure temperature (°C) |
| P | pressure (Pa) |
| PMV | predicted mean vote |
| PPD | predicted percentage dissatisfied |
| Q | rate of heat loss (W) |
| Qc | rate of heat loss/gain by convection (W) |
| Qcd | rate of heat loss/gain by conduction (W) |
| Qe | rate of heat loss by evaporation (W) |
| Qr | rate of heat loss/gain by radiation (W) |
| t | temperature (°C) |
| ta | air temperature (°C) |
| tam | ambient temperature (°C) |
| tc | comfort temperature (°C) |
| te | environmental temperature (°C) |
| tg | globe temperature (°C) |
| tr | mean radiant temperature (°C) |
| u | mean air velocity (m/s) |
| V | volume (m3) |
1.1 Introduction
This first chapter introduces you to temperature, the variations of which provides the motive force in heat transfer, and heat energy, the flow and transport of which in air, water and steam is the essence of much of heating, ventilating and air conditioning design. Its main focus, however, is on the topic of thermal comfort and the assessment of indoor climates, in which people live and work, to establish levels of comfort. The American Society of Heating Refrigeration and Air Conditioning Engineers (ASHRAE) defines thermal comfort as ‘that condition of mind in which satisfaction is expressed with the thermal environment’. The accurate assessment of building heat losses and gains, the type of comfort systems selected and the regimes of control of the comfort systems are all directed towards achieving this definition.
The effect that the amount of clothing, which is worn, has on different levels of activity also impinges on the comfort of the individual. Because thermal comfort is also a subjective assessment, a minority of individuals may feel uncomfortable even in thermal environments which are well regulated.
1.2 Heat energy and temperature
A definition of energy is the capacity a substance possesses which can result in the performance of work. It is a property of the substance. Heat on the other hand is energy in transition. Heat is one form of energy and can be expressed, for example, as a specific heat capacity in kJ/kgK. In this form, it is expressing the potential of a substance for storing heat which it has absorbed from its surroundings. It can also express the potential for the intensity of heat transfer from the substance to its surroundings.
Up until the end of the eighteenth century, heat energy, known as ‘caloric’, was considered as a fluid which could be made to flow for the purposes of space heating among other things or it flowed of its own volition as a result of friction which was generated as a result of a process or work done such as boring out a cannon. The idea of heat being a form of energy rather than a fluid was developed by an American named Benjamin Thompson, subsequently known as Count Rumford, during the process of boring out cannons for his arsenal as war minister of Bavaria. His conclusions were that the amount of heat liberated depended upon the work done against friction by the boring device. A partial definition of heat energy is, therefore, the interaction between two substances which occurs by virtue of their temperature difference when they communicate. However, heat energy does not always initiate a rise in temperature as in the cases of the latent heat of vaporisation and condensation which occur when substances change in state. This is a qualification of the definition.
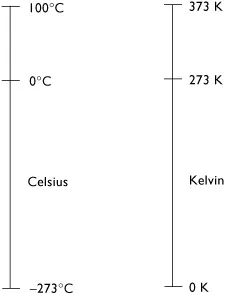
Figure 1.1 Scales of temperatures.
Heat is a transient commodity like work; it exists during communication only, although like work its effect may be permanent. The primary need for burning fuel oil might be the generation of heat energy and the permanent result of the process are the products of combustion. The combustion products cannot return to fuel oil. The transient result is the generation of heat.
A definition of temperature is a scaled measurement of relative hot and cold sensations. It can be described as an intensity of hotness or coldness. Kelvin found that absolute coldness is reached when the agitation of the molecules and atoms of a substance ceases at −273.15°C (0.0 K). Temperature scales have been advanced by various authorities. The scales commonly in use now are the Celsius scale and the Kelvin scale. Figure 1.1 shows these scales from absolute zero to the upper fixed point.
1.3 Thermometry
In the seventeenth century it was proposed that two fixed points should be used to determine a temperature scale:
• The lower fixed point was taken as melting ice at atmospheric pressure; the ice being distilled water. The ice point is the temperature at which ice and water can exist in equilibrium.
• The upper fixed point was taken as steam generated from distilled water when boiling at atmospheric pressure. Since the temperature tends to vary depending upon geographical location, the steam point is the temperature of boiling water and steam at atmospheric pressure on latitude 45.
Table 1.1 Fixed points of the International Temperature Scale
| Fixed points | Temperature (°C) |
| Boiling point of liquid oxygen | −182 |
| Ice point | 0 |
| Steam point | 100 |
| Boiling point of sulphur | 444.6 |
| Freezing point of silver | 960.8 |
| Freezing point of gold | 1063.0 |
Table 1.1 lists the fixed points of the International Temperature Scale at standard atmospheric pressure (101 325 Pa).
1.4 Types of thermometer
There are six main types of thermometer, namely
1 constant volume gas thermometer,
2 resistance thermometers,
3 thermocouples,
4 liquid thermometers,
5 bimetallic thermometer,
6 pyrometers.
The constant volume gas thermometer was selected in 1887 as the standard: it did not give a pointer reading, however. For a perfect gas, Boyle’s Law states that P ∝ 1/V at constant temperature and the Kelvin scale (Figure 1.1) agrees exactly with the scale of a perfect gas thermometer.
The resistance thermometer consists of a platinum wire wound onto two strips of mica and the coil is attached to leads, which is connected in turn to a wheatstone bridge.
Thermocouples have the measuring element as the junction between ...