1 Overview of anatomy and physiology of the central nervous system
The nervous system is a fascinating, complex and remarkable structure. It is one of the two major regulatory systems of the body, the other being the endocrine system. However, the nervous system not only regulates internal body functions, it facilitates the body’s ability to interact with the environment. The nervous system is the body’s most rapid means of maintaining homeostasis and to achieve this it must be able to constantly react and adjust to internal and external stimuli. These stimuli are detected and conveyed by nerves to the brain where they are analysed and interpreted resulting in a coordinated response. Neuroanatomy and physiology are the foundations upon which an understanding of nervous system disorders is based (Crossman and Neary 2000). The aim of this chapter is to provide a clear and concise overview of anatomy and physiology of the nervous system, with particular emphasis upon changes that occur as part of normal childhood growth and development.
At the end of reading this chapter you will be able to:
Learning outcomes
- Describe the main divisions of the nervous system
- Outline the key anatomical structures of the brain
- Describe the physiology of nerve conduction
- Appreciate embryological development of the nervous system
1.1 Introduction and overview of the organization of the nervous system
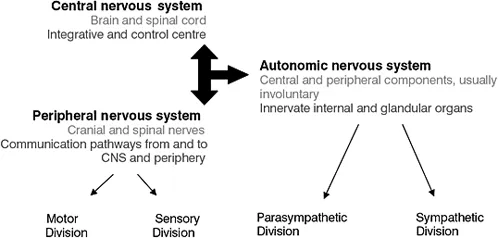
Although the nervous system functions as a whole, for descriptive purposes it is divided into the central nervous system (CNS) and the peripheral nervous system (PNS) (Sugarman 2002).
Central nervous system
The CNS consists of the brain and spinal cord which are respectively enclosed in the cranial vault and spinal column. The three main functions of the nervous system are sensory (detect changes), integrative (interpret changes) and motor (respond to changes initiating action). The CNS is the control centre for the entire nervous system (Figure 1.1a).
Figure 1.1a Relationship between the components of the nervous system
Peripheral nervous system
The PNS consists of all the nerve networks which link the CNS with the periphery, such as the body’s receptors, muscles, glands and organs. Peripheral nerve pathways are further differentiated into ascending or afferent pathways that carry information towards the CNS, and descending or efferent pathways that carry information away from the CNS to the effector organs (Sugarman 2002). Clinically the PNS has two main subdivisions, the somatic nervous system (SNS) and autonomic nervous system (ANS) (Sugarman 2002). Actions carried out by the SNS are both voluntary and involuntary with sensations consciously perceived, while actions carried out by the ANS are involuntary and are not usually consciously perceived.
Somatic nervous system
The SNS can be divided into:
- Sensory afferent nerve receptors which are located in the skin and transmit impulses to the brain. Impulses are perceived as general senses such as touch, pressure, temperature and pain;
- Special sense organs transmit impulses to the brain. Impulses are perceived as smell, taste, vision and hearing;
- Motor efferent nerves that conduct impulses away from the CNS to the skeletal muscles;
- Mixed nerves containing both sensory and motor neurons;
- The 12 pairs of cranial nerves (Table 1.1a) and 31 pairs of spinal nerves.
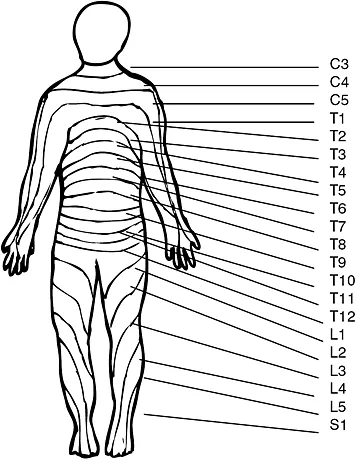
Sensory spinal nerves carry information from specific areas of the body to the CNS. These regions can be graphically represented by known as a dermatome (Figure 1.1b). Similarly each group of muscles innervated by nerves from a particular spinal nerve can be represented graphically known as a myotome. A plexus is a network of nerves, for example the brachial plexuses is formed from C 5, 6, 7, 8 and T1 spinal nerves, which innervate the skin and muscles of the upper limbs and chest muscles.
Table 1.1a Summary of the cranial nerves and their functions
Figure 1.1b Dermatome map (anterior view) (Source: Crossman and Neary 2000)
Autonomic nervous system
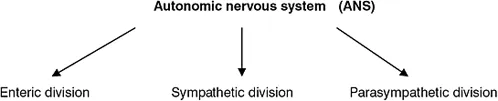
The ANS has components within both the central nervous system and the peripheral nervous system (Crossman and Neary 2000). The ANS can be further divided into the sympathetic and parasympathetic divisions, and the enteric nervous system (Figure 1.1c).
The ANS is primarily concerned with the innervation and control of visceral organs, smooth muscle and secretory glands with the overall aim to maintain homeostasis in the internal environment (Crossman and Neary 2000). Sensory neurons of the ANS carry impulses to the central nervous system from the receptors of the body’s organs. Motor neurons of the ANS carry impulses from the central nervous system to smooth muscle, cardiac muscle and the glands. Where organs have both sympathetic and parasympathetic nerve innovation their actions have an antagonistic effect. Table 1.1bprovides examples of the opposing actions of the ANS.
Figure 1.1c Divisions of the autonomic nervous system
Table 1.1b Actions of the sympathetic and para-sympathetic divisions of the ANS
Enteric nervous system
The enteric nervous system is an interconnected network of neurons organized in two cylindrical sheets embedded along the length of the gut wall. Nerves of the enteric system innervate the pancreas, liver and gall bladder (Crossman and Neary 2000; Longstaff 2005). The enteric nervous system assists in controlling peristaltic activity, glandular secretions and water and ion transfer, in essence regulating gut functioning.
1.2 Development of the nervous system
An understanding of the development of the nervous system is important because the cause of many neurological problems in infancy may be a result of a malformation that has occurred during embryological development (Padgett 2002). Following fertilization, cell division rapidly occurs and at three days a hollow ball of cells, known as a blastocyte, embeds in the uterine wall. After implantation the inner mass of the blastocyte begins to differentiate into the three primary germ layers: the ectoderm, endoderm and mesoderm. These germ layers form different structures of the body. The endoderm differentiates primarily into epithelial tissues such as the lining of the gastrointestinal tract and internal organs. The mesoderm differentiates primarily into muscles and connective tissues. The ectoderm differentiates primarily into all nervous tissue, the ear, the eye and the epidermis.
The first obvious sign of nervous system development is during the third week of embryonic life when the dorsal midline of the ectoderm thickens to form the neural plate (Padgett 2002). The lateral margins of the plate become elevated resulting in a midline depression known as the neural groove. Eventually the folds become apposed and fuse together creating the neural tube. The neural tube is completely closed by the end of the forth week of embryonic development and becomes the CNS (Longstaff 2005).
Points to consider
Failure of the neural tube to fuse results in abnormalities such as: anencephaly, which occurs if the rostral (or uppermost) portion of the tube fails to close and spina bifida if the caudal (or hind) portion fails to fuse.
The central fluid-filled canal persists into adulthood, the rostral portion becoming the ventricular system in the brain and the caudal portion becoming the central canal in the spinal cord. As the neural tube is forming some of the cells become isolated, grouping together to form the neural crests (Figure 1.2a). These eventually form most of the cells of the peripheral nervous system, and the sensory cells of the spinal and cranial nerves.
Development of the brain
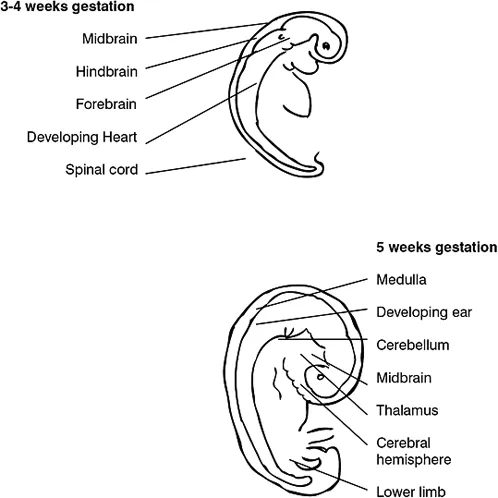
By the fifth week of gestation the rostral portion of the neural tube undergoes extensive differentiation, with the three primary brain vesicles, the forebrain, midbrain and hindbrain, becoming identifiable. The forebrain (cerebrum) is by far the largest of these divisions and eventually gives rise to the two cerebral hemispheres and the thalamus (Figure 1.2b). The midbrain remains relatively undifferentiated. The hindbrain develops into the pons, cerebellum and medulla oblongata. By convention the midbrain, medulla and pons are collectively known as the brain stem (Table 1.2a).
Figure 1.2a Formation of the neural tube from the embryonic ectoderm
Table 1.2a Relationship between embryonic development and final structures
Up to about six weeks of gestation the single layer of neuroepidermal cells increase in number and are the precursors of both the supporting glial cells and the neurons. Cell differentiation and final function is determined by genetic coding and extracellular signalling, which is influenced by the position of the cells within the blastocyte (Longstaff 2005). Early neurons are known as neuroblasts, and initially proliferate at a faster rate than the glial cells (Longstaff 2005). In the final stages of differentiation the neuroblast’s eventual structure, function and position are established, and during the last division neuroblasts lose the ability to divide.
Figure 1.2b Development of the brain and spinal cord
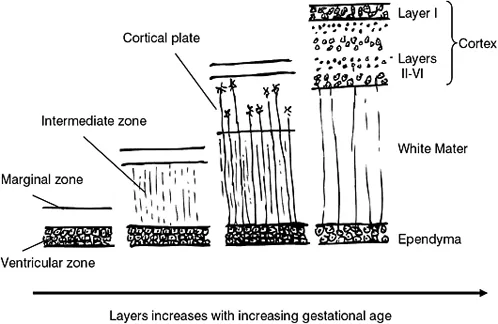
The increase in cell numbers results in the formation of layers of cells, which in turn form discrete zones, the first being the inner ventricular zone (Longstaff 2005). The inner ventricular zone is situated nearest to the neural tube. As new cells are formed at the base of the neural tube they migrate through the layers of previously formed cells, the new daughter cells adding to the rapidly expanding cerebral cortex (Figure 1.2c). Each zone of cells become specialized in their role, and the mass organization of cells eventually forms the cerebral hemispheres. This cell migration results in the development of the typical appearance of the brain: an outer cortex of grey matter (consisting primarily of cell bodies) and the white matter of the inner cortex consisting primarily of nerve tracts. Neuronal migration, which occurs as early as the second month of gestation, is controlled by a complex assortment of chemical mediators and cell signalling.
Figure 1.2c Schematic representation of cell migration and the development of the layers of the brain
Points to consider
Neuronal migration disorders, for example lissencephaly, are a group of defects caused by the abnormal migration of neurons in the developing brain. These disorders can have a profound effect upon a child’s ability to develop normally.
By the 28th week of gestation the major fissures (the lateral, central and calcarine sulci) have appeared on the surface of the brain. As the brain develops, the central cavity in the rostral portion of the neural tube also undergoes considerable changes in shape and size, and forms the ventricular system (FitzGerald and Folan-Curran 2002). Although the neuronal cell numbers do not increase after birth, continued glial cell proliferation, increased neuronal connections and...