![]()
1 Introduction
Jie Jack Li
CONTENTS
1.1 Nomenclature, Structure, and Stability
1.2 Generation of Carbocations
1.3 The Nonclassical Ion Controversy
1.4 Electrophilic Addition to Alkenes
1.4.1 Addition of Hydrohalides and Acetic Acid
1.4.2 Hydration
1.4.3 Addition of Halogens
1.4.4 Hydroboration of Olefins
1.4.5 Oxymercuration of Olefins
1.5 Electrophilic Aromatic Substitution
1.5.1 Mechanism and Orientation
1.5.2 Nitration
1.5.3 Halogenation
1.5.4 Friedel–Crafts Alkylation
1.5.5 Friedel–Crafts Acylation
1.6 β-Elimination Reactions
1.7 Rearrangement Reactions of Carbocations
1.7.1 Pinacol Rearrangement
1.7.2 Beckmann Rearrangement
1.7.3 Demjanov and Tiffeneau–Demjanov Rearrangement
1.7.4 Meyer–Schuster Rearrangement
1.7.5 Schmidt Reactions
1.7.6 Wagner–Meerwein Rearrangement
References
1.1 NOMENCLATURE, STRUCTURE, AND STABILITY
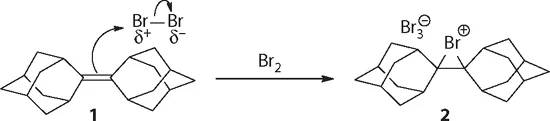
In sophomore organic chemistry, we learned that many organic compounds bear positive charges. For instance, bromonium ion was invoked as the putative intermediate when explaining the mechanism of electrophilic addition of bromine to alkenes. In 1985, Brown at Alberta isolated and determined the crystal structure of the bromonium ion 2 during the course of electrophilic bromination of adamantylideneadamantane (1).1
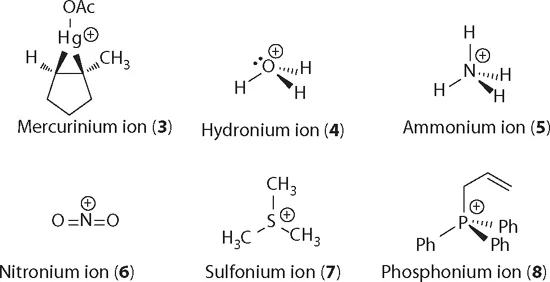
Other positive species in organic chemistry include mercurinium ion (3), hydronium ion (4), ammonium ion (5), nitronium ion (6), sulfonium ion (7), and phosphonium ion (8), just to name a few.
But none of the aforementioned -nium ions are as prevalent as carbonium ions. After all, organic chemistry is the study of carbon-containing compounds. As time passes, the term carbonium ions have been largely replaced by the more popular term, carbocations. This was probably inevitable since carbocation is one word and carbonium ion are two!
As a matter of fact, initially proposed by Olah2 and later accepted by the IUPAC (International Union of Pure and Applied Chemistry), the term carbocation encompasses two species, one is the carbenium ion, which represents the “classical” trivalent ions with CH3⊕ as the parent, and the other is the carbonium ion, which represents the “nonclassical” penta- (or higher) coordinate ions with CH5⊕ as the parent.
This book titled Carbocation Chemistry will cover both carbonium ions and carbenium ions. And in this chapter, carbocation and carbenium ion are used interchangeably because nearly all of the carbocations mentioned here are carbenium ions.
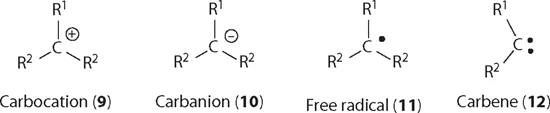
Carbocations depicted below as 9, with a cation on the carbon atom, is one of the important reaction intermediates in organic chemistry. The other intermediates include carbanions (10), radicals (11), carbenes (12), etc. For a trivalent carbocation, it is sp2 hybridized with a geometry of trigonal planar and all three bond angles are 120°.
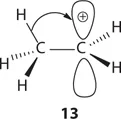
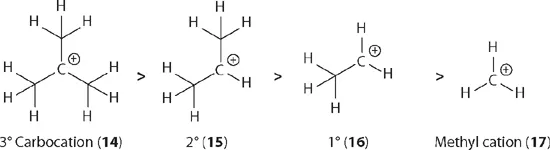
The stability of a carbocation is determined by its structure: the more alkyl substituents, the more stable. For tertiary (3°), secondary (2°), primary (1°), and methyl carbocations, their corresponding stability may be explained using the concept of hyperconjugation, which refers to the donation of a pair of bonding electrons into an unfilled or partially filled orbital. As depicted by ethyl cation (13), the two bonding electrons of the C–H σ-bond adjacent to the carbocation may donate part of its electron cloud to the carbocation's empty p orbital. As a consequence, cation 13 is more stabilized than it would if the α C–H bond did not exist. In all, ethyl cation 13 has three α C–H bonds, thus three hyperconjugations (a short hand to describe three possibilities of having hyperconjugation).
As clearly depicted below, t-butyl cation (14) as a representative of tertiary (3°) carbocations possesses nine α C–H bonds, therefore it has nine hyperconjugations. Meanwhile, isopropyl cation (15) as a representative of secondary (2°) carbocations possesses six α C–H bonds, therefore it has six hyperconjugations. In the same vein, primary (1°) cation 16 has three hyperconjugations and the methyl cation (17) has none. We can thus conclude that tertiary (3°) carbocations are the most stable in this series, followed by secondary (2°), primary (1°), and methyl carbocations. The stability of cations decreases in this sequence from the left to the right. This trend is, not surprisingly, consistent with experimental data generated from their dissociation energies in the gas phase.3
In addition to evoking the concept of hyperconjugation, the stability of carbocations may be explained using the concept of inductive effects. The carbon atom is more electronegative than the hydrogen atom with electronegativity values of 2.55 and 2.20, respectively. Therefore, the methyl group is considered electron donating. The t-butyl cation (14) has three electron-donating methyl groups; isopropyl cation (15) has two electron-donating methyl groups; ethyl cation 16 has one electron-donating m...