![]()
1
Dynamical information processing in the CA1 microcircuit of the hippocampus
Bruce P. Graham and Vassilis Cutsuridis
A major challenge to understanding cortical function is the complexity found at both the single-cell and microcircuit levels. Here we outline what is known about the microcircuitry of the CA1 region of the mammalian hippocampus. We then explore the possible functional roles of the variety of neuronal types within this microcircuit during dynamic information processing. This is considered within the framework of CA1 acting as an associative storage device during encoding and retrieval of episodic memories.
1 Introduction
The local circuitry to be found in many parts of mammalian nervous systems consists of a complex architecture involving many different neuronal types connected in feedforward and feedback loops. Synaptic connections may be excitatory or inhibitory and target specific spatial locations on a neuron. In addition to synaptic input, a neuron and the microcircuit it is a part of are subject to diffuse neuromodulatory signals. Neural synaptic transmission and neuromodulation combine to provide a complex dynamics of neural activity and presumed information processing in a neuronal microcircuit.
Computational models of cognitive behaviour generally seek to provide a simple but cogent explanation of the functionality required to produce a particular behaviour. A model may be more or less interpretable in terms of the workings of a particular brain area, or set of connected areas. Often an artificial neural network (ANN) approach is used in which the simple computing units may correspond to populations of neurons rather than to individual biological neurons. The next level of biological detail is to use spiking neuron models where the identification with real neurons may be one-to-one. Such spiking models are of the integrate-and-fire type, or they may include explicit biophysical properties of a neuron in a compartmental model. Typically the neuronal types in such models are restricted to the principal excitatory cells, plus one or two sources of inhibition.
As we learn more about the details of real neural microcircuitry, it is clear that our current models lack the richness in spatial and temporal information processing that brain circuits possess. The challenge is to build models that include more of the known biological details – such as further cell types and more complex models of individual neurons – but remain simple enough that they are understandable and provide explanatory power for cognitive function. To explore the ways forward, here we outline what is known about a particular neuronal microcircuit: the CA1 region of the mammalian hippocampus. We then try to relate aspects of this microcircuit directly to the general cognitive function of the storage and recall of information in an associative memory.
2 The hippocampal CA1 microcircuit
For both historical and experimental reasons, the hippocampus is among the most widely studied of mammalian brain regions, yielding a wealth of data on network architecture, cell types, the anatomy and membrane properties of pyramidal cells and some interneurons, and synaptic plasticity (Andersen, Morris, Amaral, Bliss, & O’Keefe, 2007). Its basic functional role is hypothesized to be the formation of declarative, or episodic, memories (Andersen et al., 2007; Eichenbaum, Dudchenko, Wood, Shapiro, & Tanila, 1999; Wood, Dudchenko, & Eichenbaum, 1999). Various subsystems, such as dentate gyrus, CA3 and CA1, may be involved in the storage of information in context, such as location in a particular spatial environment (Andersen et al., 2007), with appropriate recoding of afferent information depending on familiarity or novelty (Treves & Rolls, 1994).
The mammalian hippocampus contains principal excitatory neurons (pyramidal cells in CA3 and CA1) and a large variety of inhibitory interneurons (Freund & Buzsaki, 1996; Somogyi & Klausberger, 2005). The circuitry they form exhibits different rhythmic states in different behavioural conditions. Multiple rhythms, such as theta (4–7 Hz) and gamma (30–100 Hz) oscillations, can coexist (Whittington & Traub, 2003). This dynamic complexity presumably corresponds to specific functional processing of information (Axmacher, Mormann, Fernandez, Elger, & Fell, 2006). Much work has been devoted to trying to understand the cellular and network properties that generate these rhythms (Buzsaki, 2002; Traub, Jefferys, & Whittington, 1999), but much is still to be been done to decipher the function of the detailed microcircuits. In particular, how is plasticity controlled so that it does not interfere with previously stored memories while appropriately assimilating familiar and new information? This is the fundamental question that we will address, concentrating on the operation of the CA1 area.
2.1 External inputs to CA1
The CA1 region is one of several stages of information processing in the hippocampus. Its major sources of input are from the CA3 region of the hippocampus and the entorhinal cortex. It sends excitatory output back to the entorhinal cortex, both directly and via the subiculum, and sends diverse outputs to a variety of other brain regions, such as the olfactory bulb. In addition, there are inhibitory projections from CA1 to the medial septum (MS) and back to CA3 (Sik, Ylinen, Penttonen, & Buzsaki, 1994). In turn, CA1 receives GABAergic inhibition and cholinergic neuromodulation from the MS (Freund & Antal, 1988; Frotscher & Lenrath, 1985). CA1 also receives a variety of other neuromodulatory inputs, including dopaminergic and noradrenergic pathways. Much of this neuromodulation is directed to the distal apical dendrites of CA1 pyramidal cells, where it coincides with the entorhinal glutamatergic input (Otmakhova & Lisman, 2000).
2.2 Neuronal types and their connectivity
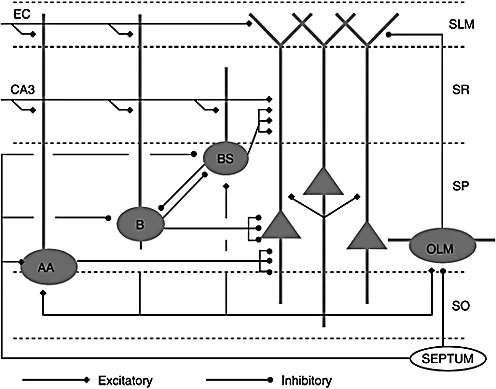
The basic hippocampal CA1 microcircuit is shown in Figure 1.1. The single excitatory cell type is the pyramidal cell (PC), which is the putative major
information processor for signals entering this brain region and is the major source of output from CA1. Pyramidal cells, here and elsewhere in the hippocampus and neocortex, have a large dendritic tree that is divided into apical and basal dendrites. These dendrites are the target for synaptic inputs that have distinct spatial segregation depending on the neuronal source.
Excitatory inputs from outside CA1 make connections on specific portions of the apical and basal dendrites of PCs (Ishizuka, Cowan, & Amaral, 1995). The Schaffer collateral input from pyramidal cells in the CA3 region of the hippocampus is exclusively to the proximal region of the apical dendrites constituting stratum radiatum (SR) and to the basal dendrites in stratum oriens (SO). Perforant path input from layer III of entorhinal cortex (EC) reaches the distal part of the apical dendritic tree in stratum lacunosum-moleculare (SL-M). Recurrent collaterals from other CA1 PCs synapse on the basal dendrites. Such collaterals are rather sparse in CA1, with only about 1% recurrent connectivity between pyramidal cells (Deuchars & Thomson, 1996). There are additional excitatory inputs from the thalamus to SL-M and from the amygdala to SO (Somogyi & Klausberger, 2005).
The pyramidal cells are surrounded by a variety of inhibitory interneurons (INs). These INs differ in morphology, pharmacology and connectivity (Freund & Buzsaki, 1996; Maccaferri & Lacaille, 2003; McBain & Fisahn, 2001; Somogyi & Klausberger, 2005). Though a complete catalogue of interneuronal types remains to be determined, at least 16 classes can be distinguished on anatomical, electrophysiological and pharmacological grounds (Somogyi & Klausberger, 2005). The most clear-cut types are basket cells, bistratified cells, axo-axonic (chandelier) cells and oriens lacunosum-moleculare (horizontal) cells. However, basket cells in particular consist of at least two subtypes: one that expresses parvalbumin and one that expresses cholecystokinin. Others include horizontal and radial trilaminar cells and INs that only synapse onto other INs (Freund & Buzsaki, 1996). A subclass of horizontal trilaminar cells (HTCs) sends axon collater...