
- 320 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Understand Electronics
About this book
In Understand Electronics, Owen Bishop covers the basics needed in all the main areas of electronics with a jargon-free, informative style, and numerous illustrations.
An essential book for the newcomer to electronics, this highly practical guide provides a thorough introduction to practical electronics and the basic principles of electronic components and circuitry. Owen Bishop, a leading international electronics author, takes a step-by-step approach, employing the minimum of mathematics, to create a text that is ideal for the student or enthusiast who has little previous knowledge of the subject area but wishes to find out more.
The new edition has been thoroughly updated throughout, with many new illustrations and coverage of recent advances in electronics, including a chapter on microcontrollers - the simple programmable devices that are transforming electronics project work. A new section on test equipment reinforces the practical emphasis of the book.
- A practical guide made highly accessible through clear explanations and numerous illustrations
- Assumes little previous knowledge of the subject area or mathematical background
- Covers the basics needed in all the main areas of electronics
Tools to learn more effectively

Saving Books

Keyword Search

Annotating Text

Listen to it instead
Information
1
Electrons and electricity
Electricity consists of electric charge. Though electricity has been the subject of scientific investigations for thousands of years, the nature of electric charge is not fully understood, even at the present day. But we do know enough about it to be able to use it in many ways. Using electric charge is what this book is about.
Electric charge is a property of matter and, since matter consists of atoms, we need to look closely at atoms to find out more about electricity. But, even without studying atoms as such, we are easily able to discover some of the properties of electricity for ourselves.
The simplest way to demonstrate electric charge is to take a plastic ruler and rub it with a dry cloth. If you hold the ruler over a table on which there are some small scraps of thin paper or scraps of plastic film, the pieces jump up and down repeatedly. If you rub an inflated rubber balloon against the sleeve of your clothing then place it against the wall or ceiling, for a while, the balloon remains attracted to the wall or ceiling, defying the force of gravity. The electric charge on the plastic ruler or wall is creating a force, an electric force. In effect, the energy of your rubbing appears in another form which moves the pieces of paper, or prevents the balloon from falling.
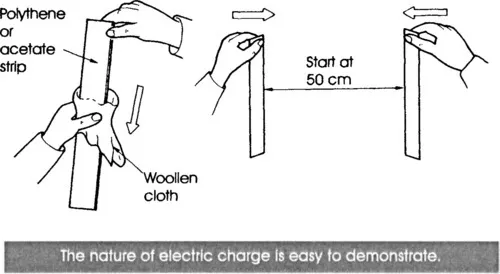
You can also charge a strip of polythene sheet (cut from a plastic food-bag) by rubbing it. If you charge two strips, then hold them apart and then try to bring them together, they are repelled by each other. As you try to push them together, their lower ends diverge, spreading away from each other.
A similar experiment is to charge a strip of acetate sheet (cut from a shirt-box) by rubbing it and bring it toward a charged polythene strip, the two strips attract each other. If they are allowed to, their lower ends come together and touch. From this behaviour, we reason that the charge on an acetate strip must be of a different kind from that on a polythene strip. Simple demonstrations such as these show that:
• There are two kinds of electric charge.
• Like kinds of charge repel each other.
• Opposite kinds of charge attract each other.
Electric charge and atoms
Now we are ready to link the basic facts about electric charge to what is known about the structure of matter.
Research has shown that atoms are built up of several different kinds of atomic particle. Most of these occur only rarely in atoms but two kinds are very common. These are electrons and protons. Although protons are about 2000 times more massive than electrons, protons and electrons have equal electric charges. The charge on an electron is opposite in its nature to the charge on a proton; these are the two kinds of electric charge mentioned above. The charge on an electron is said to be negative and that on a proton to be positive, but this is simply a convention. There is nothing positive on a proton which is ‘missing’ or ‘absent’ from an electron. The two terms merely imply that positive and negative charges are opposite.
We have said that electrons and protons carry equal but opposite charges. If an electron combines with a proton, their charges cancel out exactly and an uncharged particle is formed — a neutron. Since neutrons have no charge, they are of little interest in electronics.
Atomic structure
All atoms are composed of electrons and protons (ignoring the other rare kinds of particle). The simplest possible atom, the atom of hydrogen, consists of one electron and one proton. The proton is at the centre of the atom and the electron is circling around it in orbit. With one unit of negative charge and one of positive charge, the atom as a whole is uncharged.
Since the electron is moving at high speed around the proton, there must be a force to keep it in orbit, to prevent it from flying off into space. The force that holds the electron in the atom is the attractive electrical force between oppositely charged particles, which we demonstrated earlier. It acts in a similar way to the attractive force of gravity, which keeps the Moon circling round the Earth, and the planets of the solar system circling round the Sun.

Other atoms
There are more than a hundred different elements in nature, including hydrogen, helium, copper, iron, mercury and oxygen, to name only a few. Each element has its own distinctive atomic structure, but all are based on the same plan as the hydrogen atom. That is to say, there is a central part, the nucleus, where most of the mass is concentrated, which is surrounded by a cloud of circling electrons. However, atoms other than hydrogen have more than one proton and also some neutrons in the nucleus at the centre of the atom. The positive charge on the nucleus is due to the protons it contains. The electron cloud contains a number of electrons to equal the number of protons in the nucleus. In this way the positive charge on the nucleus is exactly balanced by the negative charges on the electrons and the atom as a whole has no electric charge.
The electrons are in orbits at different distances from the nucleus. These orbits are at definite fixed distances from the nucleus and there is room for only a fixed number of electrons in each orbit.
Electric fields
When an object is charged there is an electric field around it. This is a force field which makes charged objects move when they are in the field. Another more familiar force field is gravity, which affects us everywhere and at all times; but gravity is only attractive, it does not repel.
The drawing shows how we imagine the field around an electron. The lines of force show the way a positive charge moves when placed in the field; it moves towards the electron. Although lines of force are strictly imaginary (just as the lines of latitude and longitude on the Earth are imaginary) it helps to think of them as if they are like rubber bands under tension. This gives them two properties:
• They tend to be as short as possible.
• They tend to be as straight as possible.



When there is a proton in the vicinity of an electron, we see the fields of the electron and proton combining to give lines running from the proton to the electron. Making the lines as short as possible creates forces acting on the electron and proton, drawing them together until they meet. They are attracted to each other.
The field between two electrons is very different. The lines of force of one electron do not join with those of the other electron. Each electron maintains its own field.
Another property of lines of force is that they can not cross. So the fields become distorted, as shown. But lines of force tend to become straight and, for this to happen, the electrons are forced to move further apart. They are repelled by each other.
For the same reason, two positive charges repel each other.
Charge and energy
We can now begin to understand what happens when we charge a strip of plastic by rubbing it with a cloth. Although there is normally a fixed number of electrons circling around the nucleus of an atom, the electrons in the outer or...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright page
- Introduction
- 1: Electrons and electricity
- 2: E.m.f. and potential
- 3: Resistance
- 4: Capacitance
- 5: Inductance
- 6: Simple circuits
- 7: Semiconduction
- 8: Transistors
- 9: Semiconductor circuits
- 10: Power supply circuits
- 11: Sensors and transducers
- 12: Optoelectronic sensors
- 13: Light sources and displays
- 14: Test equipment
- 15: From components to circuits
- 16: Oscillating circuits
- 17: Amplifying circuits
- 18: Operational amplifiers
- 19: Logic circuits
- 20: Audio electronics
- 21: Computers
- 22: Microcontrollers
- 23: Telecommunications
- 24: Microwaves
- 25: Detection and measurement
- 26: Electronic control
- 27: Electronics and the future
- Acknowledgements
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Understand Electronics by Owen Bishop in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Industrial Design. We have over one million books available in our catalogue for you to explore.