Chapter 1 Multidimensionality in Gas Chromatography: General Concepts
Lourdes Ramos and Udo A.Th. Brinkman
1. Introduction
Gas chromatography with open-tubular capillary columns (GC) is a powerful separation technique that is particularly suitable for determining (semi-)volatile compounds. Application to less volatile analytes is also possible provided that these analytes are first transformed into volatile derivatives. Since its introduction by Golay [1], many examples have illustrated the potential of GC for accurate identification and quantification of individual analytes in many types of difficult real-life mixtures. The excellent resolution provided by present-day one-dimensional (1D) GC, combined with its accuracy and robustness, makes this technique the preferred separation approach in a variety of application areas. However, the improved detection capabilities provided by state-of-the-art detectors have also shown that, in many cases, aroma, food, petrochemical, and environmental samples are much more complex than was assumed a decade ago. Improved knowledge about the composition of such samples demands enhanced resolution — that is, adding an extra separation/identification capability over that achieved with 1D GC. For example, considering that 1D GC separation often relies on a single (“bulk”) separation criterion (e.g., the different volatilities of the target analytes), if the vapor pressures of several analytes in a mixture do not differ sufficiently, co-elution will occur. Separation of these co-eluting analytes from each other will require the application of another separation criterion through which these analytes can be differentiated, for example, their different polarities. In principle, if co-elution remains, a third separation mechanism can subsequently be applied until complete resolution is achieved. Proper combination of the information provided by the several separation procedures will yield accurate information about the composition of the extracts.
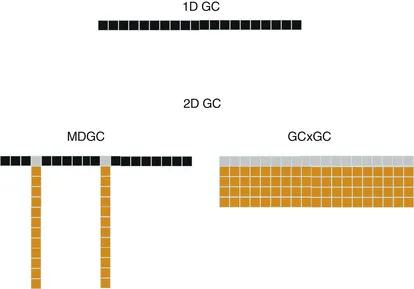
If each separation mechanism applied to the resolution of a mixture is defined as a separation “dimension,” the subsequent application of different separation mechanisms can be called a multidimensional approach. This chapter briefly introduces the concept of multidimensionality in the context of GC separations and discusses how switching from traditional multidimensional GC (MDGC or GC–GC) to comprehensive two-dimensional GC (GC×GC) dramatically enhances the potential of the technique.
2. Basic Concepts of Multidimensionality
An enormous variety of combinations of different separation mechanisms can be used to create multidimensional separation systems. A significant number of these combinations have already been implemented successfully, and the experimental results nicely illustrate the potential of high separation power that is typically associated with these techniques. For other combinations, especially those involving the cross-coupling of widely different separation techniques (e.g., liquid to gas; severe miniaturization; different flow regimes), development remains much more limited. That is, despite the promising advances made in the past two decades, in practice we are certainly far away from being able to set up the 104 to 106 binary combinations initially estimated by Giddings as possible multidimensional combinations based on known separation techniques and their variations [2]. The main stumbling block no doubt is the proper (on-line) coupling of the various dimensions or systems required for the multidimensional approach. Even for the relatively straightforward coupling of two modes of GC operation — and specifically for comprehensive GC — designing and properly using coupling devices or interfaces has been found to be a challenging task (see Chapter 2 for full details). The instrumental setup will rapidly become more complex when more than two separation systems are combined, and two-dimensional gas chromatography (2D GC) — whether of the MDGC or GC×GC type — can be considered the only combination of practical value.
The basic requirements for a multiple separation to be considered multidimensional were already discussed by Giddings in 1990
[2]. Two conditions should be fulfilled:
– The components of a mixture should be subjected to two (or more) separation steps in which their displacement is governed by different factors.
– Analytes that have been resolved in an earlier step should remain separated until the separation process is completed.
If, as the first condition requires, two (or more) independent separation mechanisms have to be used, this will result in a similar number of parameters to define the identity of an analyte [3]. That is, compared to 1D GC, each analyte in 2D GC is characterized by two independent retention times rather than by a single one, and characterization is done by indicating the proper analyte location in a 2D plane in which the two axes correspond to the retention time scales of the two separations. Obviously, preliminary identification of analytes becomes correspondingly more reliable.
The second condition requires the separate analysis of relatively small fractions of eluate from the first column on the second one, in order to maintain the resolution already achieved on the first column. Preferably, a cryogenic device should be used in between the two columns, acting as an interface to trap, focus, and release the subsequently arriving fractions onto the second column.
3. 2D GC: From MDGC to GC×GC
The standard procedure in gas chromatography is to perform 1D GC on a 30- to 60-m-long capillary column, under temperature-programming conditions, thereby achieving a peak capacity of some 100–150. If more resolution is desired, the higher potential of 2D GC is invoked (Figure 1).
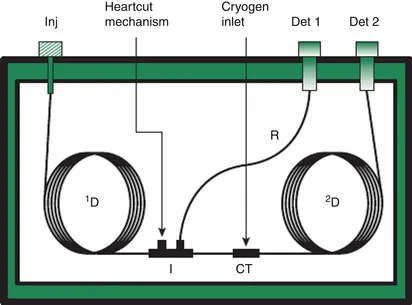
The basic setup of the multidimensional system that is now required is, in principle, fairly simple. The 30- to 60-m GC column is coupled in series, via a cryotype interface (cf. above), to a second column packed with a stationary phase providing another type of selectivity. The second column is often somewhat shorter than the first one (e.g., 10 to 20m) in order to limit the overall runtime while, of course, effecting the desired resolution. The typical setup for the first class of 2D separations, introduced by Deans in 1968 [4], MDGC, is shown in Figure 2.
The typical procedure in MDGC is to select the fraction — or, the few fractions — of interest eluting from the first column — that is, those that contain the target compounds — and to subject these, one after the other, to the second, independent, separation (see Figure 1, bottom left). MDGC was used quite widely in the final decades of the twentieth century, but it never became very popular. It probably did not catch on because the instrumental setup, though considered “fairly simple” today, was thought to be too complex for routine use at the time. (For one important exception, see the discussion of the PIONA analyzer in Chapter 7.)
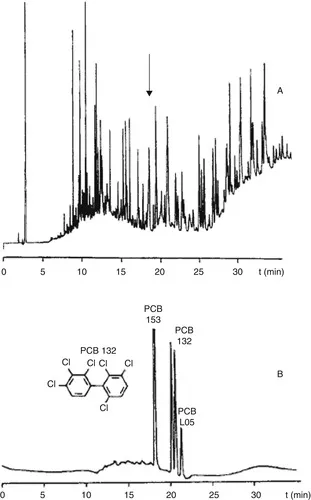
The main advantages of the MDGC approach are that, in principle, the most powerful second-dimension column can be selected for each individual target-analysis problem, and that there are no time constraints: as regards duration of the run, the second separation is not “coupled” to the first one. Successful applications include the separation of several groups of polychlorinated biphenyls (PCBs) co-eluting on a conventional nonpolar column and, then, adequately separated from each other, and from interfering sample constituents, on a more polar second column [6]. Admittedly, this frequently had the disadvantage that several heart-cuts had to be taken during one run. Another application of interest was, and remains, the use of enantioselective MDGC in food and fragrance analysis for determining enantiomeric ratios, for example, in order to detect adulteration (see, e.g. [7]). In this case, the absence of time constraints in the second dimension is a distinct advantage: because of their rather poor selectivity, chiral separations almost always require long columns. As an illustration, Figure 3 shows the (enantioselective) analysis of CBs 153 and 105, and the two atropisomers of CB 132, in a purified sediment extract using a 30m DB-5–10m Chirasil-Dex column set [8].
MDGC also has serious limitations. Its main problem is that subjecting more than one or two fractions to a second analysis dramatically increases the total runtime. After all, each second-dimension analysis will easi...