Publisher Summary
This chapter discusses the topography of chemically identified neurons in the central nervous system (CNS) and its progress during 1981–1983. The large number of substances that occurred in CNS neurons at first gave a chaotic impression rather than the feeling of increased understanding. Although, there was no doubt that the classical views based on the assumption of homogeneous brain centers and unidirectional interconnections between brain regions were challenged by later neurochemical findings, the conventional view of innervations of a certain brain area, nucleus, or even a single cell by one or two types of nerve terminals was a rough oversimplification. The degree of neuronal convergence was surprisingly high and median eminence of more than 40 amines, amino acids, and peptides were demonstrated. Increasing numbers of immunohistochemical observations provided evidence against the Dale principle—one transmitter/one neuron—demonstrating double or multiple coexistence of amines and peptides in the same neuronal cell body or terminal. A similar degree of neuronal divergence was revealed by histofluorescence and immunohistochemistry—a single neuron can innervate hundreds, thousands, and in certain cases ten thousands of other neurons.
INTRODUCTION
The chemical characterization of central nervous system (CNS) neurons began in the 1960′s and is continually expanding. This advancement is based on two factors: (1) important achievements in amine and peptide biochemistry and (2) the availability of sensitive and specific biochemical and histochemical techniques.
In the past few years a growing number of brain neurons has been identified chemically. This review is the third in the last 6 years. Papers published before 1978 were summarized in the first (Palkovits, 1978) and the progress between 1978–1980 in the second (Palkovits, 1980a). Here the latest data, published before August 1983, are briefly summarized.
In construction and form this review is confined to the topography of chemically identified neuronal cell bodies, nerve fibers and terminals in the CNS. It is a morphological summary with no neurophysiological or neuropharmacological comments. For mapping, only those morphological techniques were considered by which neurons could be visualized and localized light- or electronmicroscopically. Data obtained from biochemical [only radioimmunoassay (RIA) data] mapping of neuropeptides on microdissected brain nuclei are summarized in Tables 1.2–1.6. For didactical reasons chemical substances present in neurons are divided into four groups: transmitter amino acids, acetylcholine, biogenic amines and neuronal peptides.
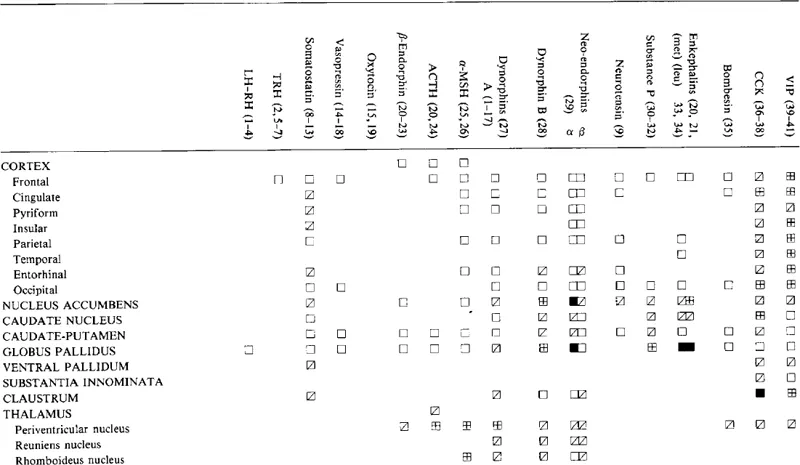
Table 1.2
CONCENTRATIONS OF NEUROPEPTIDES IN CEREBRAL CORTEX, BASAL GANGLIA AND THALAMIC NUCLEI
Concentrations: = very high;
= high;
= moderate, and
= low.
References: 1 = Palkovits et al. (1974); 2 = Kizer et al. (1976); 3 = Selmanoff et al. (1980); 4 = Samson et al. (1980); 5 = Brownstein et al. (1974); 6 = Eskay et al. (1983); 7 = Kerdelhue et al. (1981); 8 = Brownstein et al. (1975); 9 = Kobayashi et al. (1977); 10 = Palkovits et al. (1980b); ll = Epelbaum et al. (1979); 12 = Palkovits et al. (1976); 13 = Douglas and Palkovits (1982); 14 = George and Jacobowitz (1976); 15 = Dogterom et al. (1978); 16 = Hawthorn et al. (1980); 17 = Laczi et al. (1983); 18 = Zerbe and Palkovits (1984); 19 = George et al. (1976); 20 = Palkovits et al. (1978); 21 = Dupont et al. (1980); 22 = Dorsa et al. (1981); 23 = Kerdelhue et al. (1983); 24 = Krieger et al. (1977); 25 = O’Donohue et al. (1979); 26 = Eskay et al. (1979); 27 = Zamir et al. (1983a); 28 = Zamir et al. (1983b); 29 = Zamir et al. (in prep.); 30 = Brownstein et al. (1976); 31 =Kanazawa and Jessell (1976); 32 = Douglas et al. (1982); 33 = Hong et al. (1977); 34 = Kobayashi et al. (1978); 35 = Moody et al. (1981); 36 = Beinfeld et al. (1981); 37 = Beinfeld and Palkovits (1981); 38 = Beinfeld and Palkovits (1982); 39 = Palkovits et al. (1981a); 40 = Eiden et al. (1982); 41 = Rostène et al. (1982).
Table 1.3
CONCENTRATIONS OF NEUROPEPTIDES IN THE PREOPTIC, HYPOTHALAMIC, MAMMILLARY AND SUBTHALAMIC NUCLEI
Symbols and references are as in Table 1.2.
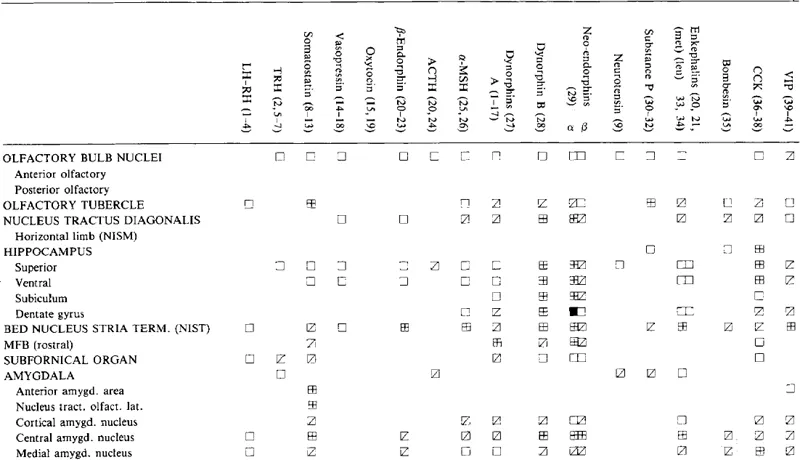
Table 1.4
CONCENTRATIONS OF NEUROPEPTIDES IN THE LIMBIC SYSTEM
Symbols and references are as in Table 1.2.
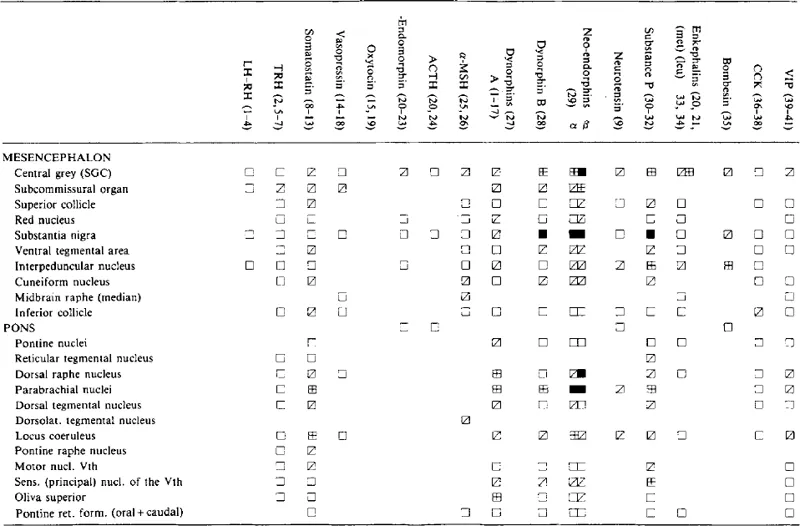
Table 1.5
CONCENTRATIONS OF NEUROPEPTIDES IN THE MIDBRAIN AND PONTINE NUCLEILH-RH
Symbols and references are as in Table 1.2.
Table 1.6
CONCENTRATIONS OF NEUROPEPTIDES IN THE MEDULLA OBLONGATA, THE CEREBELLUM AND THE SPINAL CORDLH
Symbols and references are as in Table 1.2.
Although this review aims to summarize progress since 1980, some basic data discussed in earlier reviews are also incorporated; 45 putative transmitter substances (some of them with subclasses or variants) listed in Table 1.1 are reviewed in the order given. The reader is referred for details and further references to recently published reviews (Elde and Hökfelt, 1978; Hökfelt et al., 1978a,b; Palkovits, 1978, 1980a,b, 1982a,b).
Table 1.1
CHEMICALLY IDENTIFIED SUBSTANCES (PUTATIVE NEUROTRANSMITTERS) IN THE CNS OF RAT
AMINO ACIDS
A considerable wealth of evidence suggests that some CNS amino acids may act as neurotransmitters (cf., Palkovits 1980a; Perry, 1982; Fagg and Foster, 1983). Neutral amino acids (GABA, glycine, taurine) are strong candidates to be inhibitory, the acidic amino acids (glutamate and aspartate) excitatory neurotransmitters. Although amino acids are widely distributed in the CNS, as has been detected biochemically, their definitive transmitter role cannot automatically be suggested since the presence of amino acids in the brain as the substrates of protein synthesis should also be taken into account. Beside biochemical mapping indirect immunohistochemical methods have been introduced in recent years to visualize biosynthetic enzymes of amino acids for use as markers for topographical localization of amino acid-containing cell bodies, nerve fibers and terminals.
Knowledge about brain amino acid pools (Perry, 1982) and amino acid transmitter pathways (Fagg and Foster, 1983) has been recently reviewed.
1 GABA
γ-Aminobutyric acid (GABA) is the most widely examined amino acid in the brain. It is unevenly distributed in all major brain areas but its level is generally lower than the concentrations of other transmitter amino acids except glycine. GABA is present in highest concentrations in the substantia nigra, globus pallidus, hypothalamic nuclei, midbrain colliculi and cerebellum, especially in the cerebellar nuclei (cf., Palkovits, 1978, 1980a). GABA itself cannot yet be visualized by immunohistochemistry. Its assumed distribution in the CNS is based on immunohistochemical demonstration of its rate-limiting enzyme, the glutamic acid decarboxylase (GAD).
In the 1970′s GAD-containing neuronal perikarya were demonstrated in various brain regions such as cerebral cortex, olfactory bulb, hippocampus, cerebellum, geniculates, superior collicle, reticular formation, spinal cord and spinal ganglia (cf., Palkovits, 1978, 1980a). Many of these observations were proved by recent immunohistochemical studies. In the rat cerebellum, GAD immunoreactivity was localized in all inhibitory (Purkinje, stellate, basket and Golgi cells) neurons (Oertel et al., 1981). Large, GAD-containing neurons were found in the spinal cord, predominantly within layers I and II and II–III border (Hunt et al., 1981b) and in neurons of all other laminae except motoneuron...