
eBook - ePub
Nanotechnology Applications for Clean Water
Solutions for Improving Water Quality
- 704 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Nanotechnology Applications for Clean Water
Solutions for Improving Water Quality
About this book
Nanotechnology is already having a dramatic impact on improving water quality and the second edition of Nanotechnology Applications for Clean Water highlights both the challenges and the opportunities for nanotechnology to positively influence this area of environmental protection. This book presents detailed information on cutting-edge technologies, current research, and trends that may impact the success and uptake of the applications.
Recent advances show that many of the current problems with water quality can be addressed using nanosorbents, nanocatalysts, bioactive nanoparticles, nanostructured catalytic membranes, and nanoparticle enhanced filtration. The book describes these technologies in detail and demonstrates how they can provide clean drinking water in both large scale water treatment plants and in point-of-use systems. In addition, the book addresses the societal factors that may affect widespread acceptance of the applications.
Sections are also featured on carbon nanotube arrays and graphene-based sensors for contaminant sensing, nanostructured membranes for water purification, and multifunctional materials in carbon microspheres for the remediation of chlorinated hydrocarbons.
- Addresses both the technological aspects of delivering clean water supplies and the societal implications that affect take-up
- Details how the technologies are applied in large-scale water treatment plants and in point-of-use systems
- Highlights challenges and the opportunities for nanotechnology to positively influence this area of environmental protection
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Part 1
Contaminant Sensing Technologies
Outline
Chapter 1
Sensors Based on Carbon Nanotube Arrays and Graphene for Water Monitoring
Dan Du1,3, Weiying Zhang1,3, Abdullah Mohamed Asiri2 and Yuehe Lin3,4, 1Key Laboratory of Pesticide and Chemical Biology of Ministry of Education, College of Chemistry, Central China Normal University, Wuhan, PR China, 2Chemistry Department, King Abdulaziz University, Jeddah, Saudi Arabia, 3School of Mechanical and Materials Engineering, Washington State University, Pullman, WA, USA, 4Pacific Northwest National Laboratory, Richland, WA, USA
Nanomaterials, particularly carbon nanomaterials, have shown great promise in environmental applications. In this chapter, we focus only on the use of two carbon nanomaterials, carbon nanotubes (CNTs) and graphene-based sensors for water monitoring and detection of hazardous chemicals. The excellent electrochemical behaviors of CNTs and graphene indicate that they are promising electrode materials in electroanalysis. Sensors based on CNTs and graphene have shown excellent performance in electrochemical detection of metal ions, pesticides, and other pollutants.
Keywords
Sensors; carbon nanotubes; arrays; graphene; metals; pesticides
1.1 Introduction
Environmental monitoring of toxic pollutants in surface and subsurface water sources and wastewaters presently relies on the collection of discrete liquid samples for subsequent laboratory analysis. Sensors that are field-deployable and able to measure part-per-billion (ppb) or nanomolar levels of toxic pollutants will reduce time and costs associated with environmental monitoring of hazardous chemicals. Electrochemical sensors appear to be a very promising technique that offers desired characteristics such as field-deployability, selectivity, sensitivity, robustness, and inexpensiveness [1–3].
Nanomaterials, particularly carbon nanomaterials, have a significant role to play in new developments in each of the biosensor size domains [4–6]. They have shown great promise in many applications, such as bioscience and biotechnology, energy storage and conversion, environmental and biomedical applications. In this chapter, we focus only on the use of two carbon nanomaterials, carbon nanotubes (CNTs) and graphene-based sensors for water monitoring and environmental applications.
CNTs are well-ordered, hollow graphitic nanomaterials made of cylinders of sp2-hybridized carbon atoms. They have high aspect ratios, high mechanical strength, high surface areas, excellent chemical and thermal stabilities, and rich electronic and optical properties [7]. The latter properties make CNTs important transducer materials in biosensors: high conductivity along their length means they are excellent nanoscale electrode materials [8–10]. These materials are classed as single-walled carbon nanotubes (SWCNTs) (Figure 1.1A), which are single sheets of graphene “rolled” into tubes, or multiwalled carbon nanotubes (MWCNTs), each of which contains several concentric tubes that share a common longitudinal axis [11]. As one-dimensional (1D) carbon allotropes, CNTs have lengths that can vary from several hundred nanometers to several millimeters, but their diameters depend on their class: SWCNTs are 0.4–2 nm in diameter and MWCNTs are 2–100 nm in diameter. An important part of the success of CNTs for these applications is their ability to promote electron transfer in electrochemical reactions.
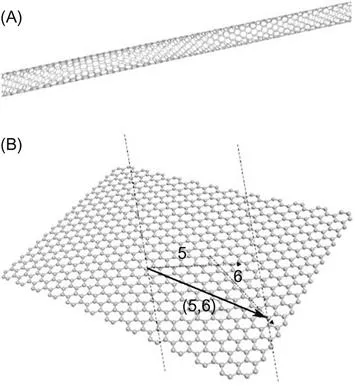
Graphene is a 2D sheet of carbon atoms in a hexagonal configuration with atoms bonded by sp2 bonds. These bonds and this electron configuration are the reasons for the extraordinary properties of graphene, which include a very large surface area (at 2630 m2/g, it is double that of SWCNTs), a tunable band gap, room-temperature Hall effect, high mechanical strength (200 times greater than steel), and high elasticity and thermal conductivity [12]. Graphene is the most recent member of the multidimensional carbon-nanomaterial family, starting with fullerenes as a 0D material, SWCNTs as 1D nanomaterials, and ending with graphite as a 3D material. Graphene fills the gap for 2D carbon nanomaterials (Figure 1.1B). Isolation of individual graphene sheets was long sought, but only in 2004, it was achieved by a surprisingly simple technique [13]. Since then, fundamental research and research on applications have increased rapidly. Graphene is an ideal material for electrochemistry [14–17] because of its very large 2D electrical conductivity, large surface area, and low cost. The use of graphene in electrochemical sensors and biosensors is particularly interesting, with the first articles emerging in 2008. Since then, their number has grown explosively.
1.2 CNT-based electrochemical sensors
1.2.1 Various methods for preparation of CNT-based sensors
Although CNTs are relatively new in analytical fields, their unique electronic (electron transfer rate similar to edge-plane graphite), chemical (biocompatibility and ability to be covalently functionalized), and mechanical properties (3 times stronger than steel) make them extremely attractive for chemical and biochemical sensors [18,19]. Various methods have been used to prepare CNT-based sensors: (a) casting of CNT thin films, from the suspensions of CNTs in solvents [20–28], such as sulfuric acid [21], Nafion [25], dihexadecyl hydrogen phosphate (DHP) [26], DMF [27], and acetone [28], prior to being coated on electrode surfaces, (b) using CNTs as paste electrodes or electrode composites [29–31], and (c) using aligned CNT as electrode substrates [32–40]. With specific to metal ion sensors, DHP [26] and Nafion [25] have been used to disperse MWCNTs under ultrasonication prior to being drop-coated on glassy carbon electrodes. The CNT film enables the development of mercury-free electrodes that can detect from 10−9 to 10−6 M of Cd and Pb.
Most CNT-based sensors take advantage of the bulk properties of CNTs, including increased electrode surface area [41], fast electron transfer rate [42], and good electrocatalytivity in promoting electron transfer reactions of many important species [43,44]. Using CNTs as nanoelectrod...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- List of Contributors
- Foreword
- Preface
- Acknowledgment
- Introduction: Water Purification in the Twenty-First Century—Challenges and Opportunities
- Part 1: Contaminant Sensing Technologies
- Part 2: Separation Technologies
- Part 3: Transformation Technologies
- Part 4: Stabilization Technologies
- Part 5: Societal Issues
- Part 6: Outlook
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Nanotechnology Applications for Clean Water by Anita Street,Richard Sustich,Jeremiah Duncan,Nora Savage in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Electrical Engineering & Telecommunications. We have over 1.5 million books available in our catalogue for you to explore.