
- 336 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Dyeing is one of the most effective and popular methods used for colouring textiles and other materials. Dyes are employed in a variety of industries, from cosmetic production to the medical sector. The two volumes of the Handbook of textile and industrial dyeing provide a detailed review of the latest techniques and equipment used in the dyeing industry, as well as examining dyes and their application in a number of different industrial sectors.Volume 2 deals with major applications of dyes and is divided into two parts. Part one covers textile applications, with chapters dealing with the dyeing of wool, synthetic and cellulosic fibres, and textile fibre blends. In part two, industrial applications of dyes are examined, with topics including dyes used in food and in the cosmetics industry.With its distinguished editor and contributions from some of the world's leading authorities, the Handbook of textile and industrial dyeing is an essential reference for designers, colour technologists and product developers working in a variety of sectors, and will also be suitable for academic use.
- Provides a detailed review of the latest techniques and equipment used in the dyeing industry
- Industrial applications of dyes are examined, with topics including dyes used in food and in the cosmetics industry
- Is appropriate for a variety of different readers including designers, colour technologists, product developers and those in academia
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Part I
Textile applications
1
The colouration of wool
D.M. Lewis, The University of Leeds, UK
Abstract:
Perceived and real environmental threats have greatly influenced wool dyeing trends and much of these effects are detailed. In particular chrome dyeing has been under scrutiny and methods to avoid the use of extremely toxic Cr(VI) compounds are discussed. Damage in wool dyeing is an important theme and the use of anti-setting agents in wool dyebaths is fully described along with their modus operandi. In the latter case reactive dyes have much to offer especially in the case of those reacting by a Michael addition mechanism. Shrink-resist treatments to give machine-washable wool are discussed in terms of required dye selection and also the environmental hazards of Absorbable Organo Halogen (AOX) production if pre-chlorination is used. Recent developments in ink-jet printing and how they should favour wool fabric printing are described.
Key words
wool chemistry
acid dyes
chrome dyes
reactive dyes
dyeing theory
dyeing machine-washable wool
damage and wool setting
ink-jet printing
1.1 Introduction
Animal fibres such as wool, cashmere and alpaca are highly prized when made into garments since these exhibit desirable properties such as soft touch, warmth, beautiful drape, excellent comfort in wear and, using modern synthetic dyes, unparalleled colouration possibilities with very few shade restrictions. Leeder1 points out that wool has been bio-engineered over millions of years to be worn next to an animal’s skin, and is thus better than other types of fibres in terms of comfort in wear; it possesses the properties of absorbing up to 30% of its own weight of water without feeling wet, and even giving out heat when it absorbs water. Despite the above strong positives it has to be noted that wool makes up less than 5% of the textile market, cotton and synthetic fibres making up the bulk – it would not be possible for wool growing to increase significantly and therefore wool must position itself mainly in the desirable ‘luxury’ item sector of the market. There are also performance disadvantages, which are not present in garments made from the other textile fibres; these include felting shrinkage (in knitwear this can be as high as 60%) during household laundering procedures, the need to scour the raw wool to remove lanolin, soil and seeds, and attack by moths and beetles during garment storage.
Wool scientists are well aware of the beautiful composite structure of the wool fibre; a single fibre is made up of many thousands of different proteins and even some 15% of non-protein material; nature has designed, through evolution, each of these components to play a role in protecting the animal from the elements and in maximising the animal’s comfort. The widely acclaimed, classic diagram showing the amazing complexity of the fibre, produced by Fraser and co-workers at Commonwealth Industrial Scientific Organisation (CSIRO)’s Division of Protein Chemistry in Melbourne, is shown in Fig. 1.1.
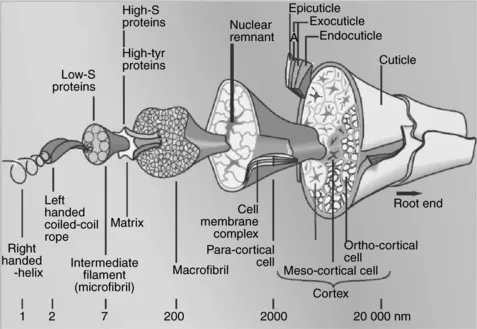
1.1 The microscopic structure of a wool fibre.
Not shown in this diagram is the F-layer which coats the epicuticle with a hydrophobic barrier; the classic studies by CSIRO scientists and others have established that this layer is made up of fatty acids covalently bonded to the fibre through thiol ester bonds at the cysteine site. The major constituent of this lipid material is 18-methyleicosanoic acid. The proteins in the epicuticle are highly disulphide cross-linked (i.e. cystine rich); in fact in the A-layer one in every 2.5 residues is cystine, making these surface proteins as hard as hoof material. These observations are crucial in understanding dye-diffusion processes when dyeing wool from aqueous solution as the surface structure creates a barrier to dye diffusion.
The marketplace is increasingly sensitive to possible negative impact of processing products on the environment; this has implications across the whole range of manufacturing processes used. This statement actually has profound implications for the finisher, dyer and printer of wool materials. In this context heavy metals present in dyes or in dyeing processes are important issues; although reactive dyes are being used more and more as replacements to produce dyed materials having high wet-fastness properties they do not give the same level of light-fastness in pale depths as do dyeings produced with pre-metallised or after-chrome dyes.
Compared to cotton and synthetic fibres, wool lacks brilliance in white and pastel shades and this has been seen as a major market issue; resolution of this problem will come from further research into improving the photo-stability of the base wool fibre2,3 and developing novel bright chromophores, even those not normally suited for dyeing wool.
To prevent felting shrinkage processes based on chlorination pretreatments are often used but these give relatively high amounts of Absorbable Organo Halogen (AOX) residues – the colourist has to be aware that up to 1000 mg/L AOX can be discharged from dye-houses dyeing these pre-treated substrates; it is vital that such pre-treatments are replaced by non-chlorine surface oxidation processes without adversely affecting dyeing properties.
Wool and other animal fibres undergo ‘setting’ reactions during dyeing, the extent of these depending on bath pH, time and temperature; the amount of set imparted influences final fibre strength and other properties such as yarn bulk. The interesting sulphur chemistry behind these reactions will be reviewed, emphasising the value of using so-called anti-setting agents in the dyeing process to improve final wool quality.
Ink-jet printing textile fabrics is now well established; in particular this procedure is ideally suited to wool printing; wool printing was a very small-scale activity in the past since screen printing required considerable investment in preparing and storing screens which could not be justified in terms of the short run-lengths involved. The special factors limiting the rapid adoption of such digital technologies for wool fabric printing will be considered.
1.2 Overview of industry practice
Prior to dyeing wool has to be scoured in the so-called raw state to remove dirt, soil, some vegetable matter, suint and lanolin or wool grease; scouring is usually carried out with non-ionic detergent and optionally sodium carbonate at 40–50 °C. This cleaned loose wool is then combed or carded to remove residual vegetable matter; combing is done on longer fibres to produce tops which are then spun on the worsted yarn system; the woollen system tends to use carded, shorter, coarser fibres. Wool may be dyed in the raw, loose fibre form, top form, or in yarn form or in piece form; dyeing is typically carried out at the boil for usually 1 h, although with current high energy costs there is increasing interest in reducing dyeing temperatures.
In the current high fashion industry it is more desirable to leave colouration to the l...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Contributor contact details
- Woodhead Publishing Series in Textiles
- Part I: Textile applications
- Part II: Industrial applications
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Handbook of Textile and Industrial Dyeing by M Clark in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Chemical & Biochemical Engineering. We have over 1.5 million books available in our catalogue for you to explore.