
eBook - ePub
Concerning Amines
Their Properties, Preparation and Reactions
- 208 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Concerning Amines: Their Properties, Preparation and Reactions attempts to describe the accumulated knowledge on the properties of amines. This text first discusses nitrogen atom contained in amines, along with the basicity, geometry, nomenclature, occurrence, reaction, and preparation of these derivatives of ammonia. This book then explains the stereochemistry of the amino group and of quaternary ammonium compounds. Amine salts; absolute configuration of asymmetric amines; and influence of amino group are also tackled. This text also looks into the amino group in heterocyclic aromatic systems. This book concludes by explaining the syntheses of heterocyclic amines, enamines, and further reactions of carbonyl compounds with amines, including the Eschweiler, Vilsmeyer, and von Braun reactions. This publication will be invaluable to those interested in studying the properties of amines, such as chemists and students of chemistry.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
Ciencias físicasSubtopic
QuímicaTHE NITROGEN ATOM
Publisher Summary
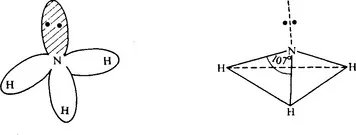
Nitrogen has atomic number 7 and the structure ls2 2s2 2p3. The three 2p electrons are disposed singly in the 2px, 2py, and 2pz orbitals. The nitrogen atom may form three covalent bonds with three hydrogen atoms resulting in stable ammonia molecule, NH3, in which the nitrogen atom still owns a lone pair of electrons.
NITROGEN having atomic number 7 has the structure 1s22s22p3, the three 2p electrons disposed singly in the 2px, 2py and 2pz orbitals. The ground state of a nitrogen atom is represented as follows:
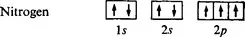
It is therefore not surprising that the nitrogen atom may form three covalent bonds with each of three hydrogen atoms with the resulting stable ammonia molecule NH3 in which the nitrogen atom still owns a lone pair of electrons.
TYPES OF AMINES
Publisher Summary
This chapter discusses types of amines. Amines are the derivatives of ammonia in which one or more hydrogen atoms have been replaced by carbon-containing groups. It does not mean that the only way of preparing amines from ammonia is by the substitution of the hydrogen atoms by carbon-containing groups. The chapter discusses different methods of preparing amines and in those methods, ammonia is not one of the reactants. Any organic substance in which the —NH2 group is present is called a primary amine; this group is attached to a carbon-containing group R that may be as simple as CH3 or quite a bit more complex in structure. An organic substance that contains an >NH group attached to two carbon-containing groups of varying complexity is called a secondary amine, and one in which three carbon-containing groups are attached to a nitrogen atom is called a tertiary amine. In all the cases, the lone pair of electrons is present on the respective nitrogen atoms.
We can consider various organic compounds containing a nitrogen atom at the same oxidation level as in ammonia, as derivatives of ammonia in which, formally, one or more hydrogen atoms have been replaced by carbon-containing groups. Because of this way of looking at these derivatives these substances have been given the name amines.
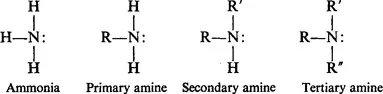
This formal representation is useful only insofar as it emphasizes the relationship of the various classes of amines to ammonia; it does not mean necessarily that the only way of preparing amines is by substitution of the hydrogen atoms in ammonia by carbon-containing groups. Quite the contrary, we shall discuss many methods of preparing amines in which ammonia is not one of the reactants.
The above four formulae show that any organic substance in which the —NH2 group is present is called a primary amine; this group is attached to a carbon-containing group R which may be as simple as CH3 or quite a bit more complex in structure. Whatever the case, it should be noted that the nitrogen atom still owns, of itself, a lone pair of electrons. (This last statement will be somewhat qualified in our later discussion.)
An organic substance which contains an 〉NH group attached to two carbon-containing groups of varying complexity is called a secondary amine and one in which three carbon-containing groups are attached to a nitrogen atom is called a tertiary amine. Here also it should be emphasized that the lone pair of electrons is present on the respective nitrogen atoms.
BASICITY OF AMINES
Publisher Summary
Amines are important bases of organic chemistry. This chapter discusses the basicity of amines. It is the lone pair on nitrogen atom that confers the basic character of the amines. If the delocalization of the lone pair occurs through its involvement with other parts of the molecule because of resonance, the basicity of the amine may be low indeed. The chapter illustrates the ionization of primary amines in aqueous solution. When ammonia reacts with a proton to give an ammonium ion, the ammonium ion must have a single positive charge to obey the law of conservation of energy. A proton is a hydrogen atom that has lost its single electron or single unit of negative charge and therefore, it is positively charged. The nitrogen atom contributes its lone pair to form a two-electron covalent bond between it and the proton. Once this has happened, one cannot tell the difference between the three nitrogen–hydrogen bonds already existing in ammonia and the fourth newly-created nitrogen–hydrogen bond.
The presence of the lone pair cannot be overemphasized since it is the lone pair which confers upon the amines, whether they be primary, secondary or tertiary, their basic character. The amines are the important bases of organic chemistry. They may be stronger or weaker bases but the fact that they are bases is due to the lone pair on nitrogen. If delocalization of the lone pair occurs through its involvement with other parts of the molecule due to resonance, the basicity of the amine may be very low indeed and we shall exemplify this in our discussion of certain aromatic and heterocyclic amines. However, in principle, the basicity of the amines ma...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- PREFACE
- EDITORIAL PREFACE
- Chapter 1: THE NITROGEN ATOM
- Chapter 2: TYPES OF AMINES
- Chapter 3: BASICITY OF AMINES
- Chapter 4: GEOMETRY OF AMINES AND AMMONIUM COMPOUNDS
- Chapter 5: NOMENCLATURE OF AMINES
- Chapter 6: PHYSICAL PROPERTIES OF AMINES
- Chapter 7: OCCURRENCE OF AMINES IN NATURE
- Chapter 8: PREPARATION OF AMINES
- Chapter 9: PRACTICAL CONSIDERATIONS IN PREPARATION OF AMINES
- Chapter 10: REACTIONS OF AMINES
- Chapter 11: STEREOCHEMISTRY OF THE AMINO GROUP
- Chapter 12: STEREOCHEMISTRY OF QUATERNARY AMMONIUM COMPOUNDS
- Chapter 13: AMINE SALTS—RESOLUTION OF RACEMATES OF ASYMMETRIC CARBON COMPOUNDS
- Chapter 14: ABSOLUTE CONFIGURATION OF ASYMMETRIC AMINES
- Chapter 15: DIRECTIVE INFLUENCE OF AMINO GROUP IN ELECTROPHILIC AROMATIC SUBSTITUTION
- Chapter 16: THE AMINO GROUP IN HETEROCYCLIC AROMATIC SYSTEMS
- Chapter 17: SYNTHESES OF HETEROCYCLIC AMINES
- Chapter 18: ENAMINES (= ene-amines)
- Chapter 19: FURTHER REACTIONS OF CARBONYL COMPOUNDS WITH AMINES
- INDEX
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Concerning Amines by David Ginsburg, Robert Robinson in PDF and/or ePUB format, as well as other popular books in Ciencias físicas & Química. We have over 1.5 million books available in our catalogue for you to explore.