
eBook - ePub
Handbook of Food Powders
Processes and Properties
- 688 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Handbook of Food Powders
Processes and Properties
About this book
Many food ingredients are supplied in powdered form, as reducing water content increases shelf life and aids ease of storage, handling and transport. Powder technology is therefore of great importance to the food industry. The Handbook of food powders explores a variety of processes that are involved in the production of food powders, the further processing of these powders and their functional properties.Part one introduces processing and handling technologies for food powders and includes chapters on spray, freeze and drum drying, powder mixing in the production of food powders and safety issues around food powder production processes. Part two focusses on powder properties including surface composition, rehydration and techniques to analyse the particle size of food powders. Finally, part three highlights speciality food powders and includes chapters on dairy powders, fruit and vegetable powders and coating foods with powders.The Handbook of food powders is a standard reference for professionals in the food powder production and handling industries, development and quality control professionals in the food industry using powders in foods, and researchers, scientists and academics interested in the field.
- Explores the processing and handling technologies in the production of food powders
- Examines powder properties, including surface composition, shelf life, and techniques used to examine particle size
- Focusses on speciality powders such as dairy, infant formulas, powdered egg, fruit and vegetable, and culinary and speciality products
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
1
Introduction to food powders
B. Bhandari, The University of Queensland Australia
Abstract :
This chapter introduces the recent advances on the understanding of properties of food powders and the fundamental mechanisms that govern their functional properties. The internal molecular- and micro-structure, and the surface properties of the powder particles that influence the bulk properties are highlighted. This chapter provides overall descriptions of the food powders but the specific details of each important property are discussed in the individual chapters of the book.
Key words
food powder structure
surface properties
mixing of powders
stickiness of powders
1.1 Introduction
Powders are particulate solid state materials containing discreet particles of size ranging from nanometres to millimetres. One gram of powder of the average particle size of 20 micron will contain around 108 particles. The bulk powder properties are the combined effect of particle properties. Food products in solid or liquid states are converted to powder form for ease of use, processing and keeping quality. Currently, many food products in the market are found in a powder form. The powder industries are growing tremendously around the globe aligned with the growth of industries that manufacture new products and powder premixes.
Various terms are used to indicate the particulate solids in bulk, such as powder, granules, flour and dust, though all these materials can be treated under powder category (Fig. 1.1, Table 1.1). These common terminologies are based on the size or the source of the materials. Granular products have a dimension on the order of millimetres while fine powder products are of average size less than 100 μm.
Table 1.1
Various terminologies used within the category of food powders
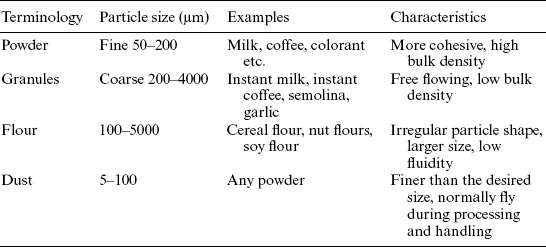

Fig. 1.1 Some examples of granules, powders and flour
All types of powders are generally free flowing, can be used in very small portions (in milligrams) and are easy to transport in pipes pneumatically. In many processing situations, the powder forms are essential, such as in mixing and dissolution. Particles are created from bulk liquid or solid materials by drying, grinding, crushing, attrition, pulverization, precipitation or crystallization (Table 1.2). The two main methods of conversion of liquid to powder form are drying and crystallization. Size-reduction processes such as grinding and milling also contribute to powder production. The particle size, distribution, shape and surface characteristics and the density of the powders are highly variable and depend on both the characteristics of the raw materials and processing conditions during their formations. These parameters contribute to the functional properties of powders, including flowability, packaging density, ease of handling, dust forming, mixing/segregation, compressibility and surface activity. Powders have a large surface area per unit volume and may be hygroscopic (e.g., high degree of moisture absorption). The stability of a powder, in terms of physical and chemical properties, is usually impaired by increased moisture sorption (Bhandari and Hartel, 2005).
Table 1.2
Different methods to produce and process powders

The properties of powder can be classified into three properties: fundamental, functional and defective (Fig. 1.2, Masters, 2002). The functional properties relate to the properties in relation to their application as a product or product ingredients and are directly influenced by the combination of fundamental properties. The defective properties are not desirable properties of the powders and often restrict their use for particular application.
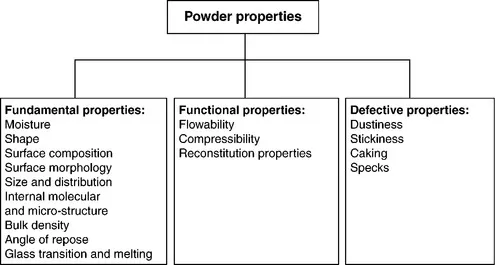
Fig. 1.2 Three classes of powder properties.
1.2 Crystalline and amorphous microstructure of powders
Food powders may be amorphous, crystalline or mixed (semi-crystalline) in their molecular level structure. Depending on the process applied, the powders can be produced in either of these forms. Powders in crystalline state possess defined molecular alignment in the long-range order; while amorphous state is disordered. The properties of food powders including their functionality and their stability are highly dependent on these structures. Many of the desired and important properties of the food materials can be achieved by altering these structures.
1.2.1 Crystalline structure
Common powders found in crystalline states are salts, sugars and organic acids. Crystalline powders are non-hygroscopic, stable and easy flowing. The crystalline form is characterized by a tightly packed molecular arrangement ; therefore, only the molecules at the air-crystals interface can interact with external materials such as water (absorption). Thermodynamically, the crystalline form is in the lowest energy level or stable equilibrium state (Hartel, 2001). Normally, the crystallization process involves concentration of the solute above super-saturation by removal of the solvent through evaporation and/or by cooling. The crystals’ shape and size changes with the species, type of the polymorphs and isomers of same species. All these properties influence the final powder fundamental and functional properties. The crystalline powders are slower to dissolve than the amorphous powders as more energy and time are needed to dissociate the strongly bound molecules in a crystalline structure.
1.2.2 Amorphous structure
Amorphous structure exists in many important food powders such as high or low molecular weight carbohydrates and proteins. Many food products such as dairy powders, fruit juice powders, honey powders and hydrolyzed protein powders are normally in amorphous state. Molecules in the amorphous state are disordered, more open and porous. Therefore, an individual molecule possesses more sites for external interactions that make them able to absorb volatiles, for example, an amorphous structure can absorb water easily. The microstructure of an amorphous solid may consist of short-range order and regions of high and low densities and have higher entropy than the corresponding crystals. As the amorphous state is a non-equilibrium state, its structure can undergo crystallization or structural relaxation to achieve an equilibrium condition (Yu, 2001). Amorphous powders are obtained by rapid supercooling or rapid removal of solvent. Very short processing condition does not provide enough time for the molecules to align themselves to become crystalline. Spray drying is one of the most common methods used to produce amorphous powders. A material with slow crystallizing tendency promotes amorphous structure formation. Addition of impurities, mainly molecules of similar molecular structure (such as fructose in glucose) or large molecules (such as maltodextrins in sucrose), can prevent or delay crystallization and promote amorphous structure formation during drying. Addition of additives to control crystallization can create a completely amorphous or mixed (crystalline + amorphous) structure.
Cereal starches have semi-crystalline structure. Grinding cereals while making flour can destroy some of their crystalline structure, still maintaining its semi-crystalline structure. During the cooking process this crystalline structure is destroyed. This event is called gelatinization. Processes of melting the crystalline solid (such as sugar and starch) and rapid cooling post-grinding can also yield an amorphous powder. Although it is an energy intensive process, partial or completely amorphous structure can be formed from crystalline solids by milling or grinding them. A micronization process can virtually convert nearly all the crystalline arrangement to a non-equilibrium amorphous structure, though some microcrystallinity may always be present. One example is grinding of sugar crystals to make icing sugar (Fig. 1.3) which is a process of conversion of crystalline structure to semi-crystalline structure (further discussed in Section 1.2.3 ).

Fig. 1.3 Crystalline structure of sugar (a) and mixed structure (amorphous + crys...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Contributor contact details
- Woodhead Publishing Series in Electronic and Optical Materials
- Preface
- Chapter 1: Introduction to food powders
- Part I: Processing and handling of technologies
- Part II: Powder properties
- Part III: Speciality food powders
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Handbook of Food Powders by Bhesh Bhandari,Nidhi Bansal,Min Zhang,Pierre Schuck,Bhesh R. Bhandari,NIDHI BANSAL in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Food Science. We have over 1.5 million books available in our catalogue for you to explore.