
- 796 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Physical Properties of Textile Fibres
About this book
First published in 1962, and now in its fourth edition, Physical properties of textile fibres has become a classic, providing the standard reference on key aspects of fibre performance. The new edition has been substantially reorganised and revised to reflect new research.After introductory chapters on fibre structure, testing and sampling, the book reviews key fibre properties, their technical significance, factors affecting these properties and measurement issues. Each chapter covers both natural and synthetic fibres, including high-performance fibres. The book first reviews properties such as fineness, length and density. It then considers thermal properties and reaction to moisture. A further group of chapters then reviews tensile properties, thermo-mechanical responses, fibre breakage and fatigue. Finally, the book discusses dielectric properties, electrical resistance and static, optical properties and fibre friction.Written by one of the world's leading authorities, the fourth edition of Physical properties of textile fibres consolidates its reputation as a standard work both for those working in the textile industry and those teaching and studying textile science.
- A standard reference on key aspects of fibre performance
- An essential read and reference for textile technologists, fibre scientists, textile engineers and those in academia
- Provides substantial updated material on fibre structure and new test methods, data and theories regarding properties of textile fibres
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
1
An introduction to fibre structure
1.1 General introduction
1.1.1 The nature of matter
Fibre physics is the study of the structure and physical properties of fibres. These two aspects are not, however, independent: the properties must be explained by the structure, which they also help to elucidate. Because of this connection, it is appropriate to start this book on the physical properties of fibres with a review of what is known about their structure. There is much detail, only partly superseded by more recent work, in the book edited by Hearle and Peters [1] and the review by Hearle and Greer [2]. Other information is in the general references given at the end of the chapter.
Matter is composed of atoms linked together by bonds of varying strength. It is the arrangement of these atoms and the strength of the bonds between them that determine the physical properties of materials. Thus with light atoms, such as those of helium, attracted to one another by very weak forces, the energy of the atoms is sufficient (except at very low temperatures) to cause them to move about independently, and the material is a gas. The material will also be a gas (though with a higher liquefaction point) if it is made up of heavier atoms, or of molecules composed of two or three atoms held together by strong forces (valency bonds), provided that the forces between the individual molecules are weak. These weak forces are often called van der Waals forces, since they are the cause of one of the deviations of a real gas from an ideal gas, which were considered by van der Waals in his modification of the gas laws. If the molecules are heavy enough, and the attractive forces strong enough, then the atoms will not have sufficient energy at room temperature to move freely away from one another, and the substance will be a liquid or a solid.
Some materials are made up of giant molecules. For example, in a crystal of diamond, all the atoms are linked to one another by valency bonds in a regular three-dimensional network. This gives a very hard, non-fusible material. In graphite, which is also pure carbon, the atoms are linked only in single planes by valency bonds; the forces between the planes are weak, so the material is one that easily splits up into sheets, and these will slide over one another, giving a lubricating action. In linear polymers, the linking is in only one dimension. If there is flexibility in the main-chain covalent bonds and only weak bonding between the long-chain molecules, there is nothing to prevent thermal energy from causing the chains to take up a disordered, random, tangled arrangement, as suggested in Fig. 1.1(a). When the material is tensioned, the molecules straighten out, giving a large extension, Fig. 1.1(b). This extension is reversible, since on releasing the tension the molecules return to the random tangle. This is the rubbery state, though some crosslinks are introduced in vulcanisation to give cohesion to the material. At lower temperatures or with stronger bonding, such an amorphous polymer material is a glassy plastic. Other linear polymers can crystallise into regular lattices to give plastics such as polyethylene of intermediate stiffness. From these examples, we see that the characteristics of matter are determined by its molecular arrangement.
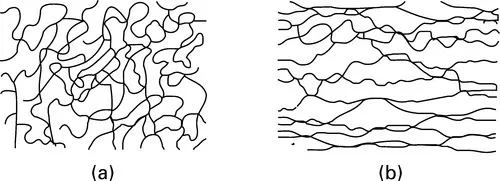
1.1 (a) Disordered arrangement of long-chain molecules in rubber. (b) Oriented arrangement of molecules in stretched rubber.
1.1.2 Intermediate bonds: hydrogen bonding
In addition to the ordinary covalent bonds that link atoms in a molecule and the usual weak van der Waals interactions between molecules, there is another class of bonds of intermediate strength, which are very important in influencing fibre properties. The best-known example is the hydrogen bond, which forms between hydroxyl (—OH) groups. Figure 1.2(a) gives a schematic representation of this bond, and Fig. 1.2(b) illustrates, in a very inexact manner, the way in which it might arise by a sharing of electrons from the outer rings of the hydrogen and oxygen atoms.
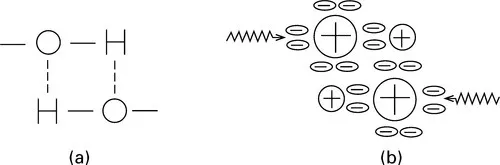
1.2 Two schematic representations of a hydrogen bond: (a) bonding; (b) electron arrangement in outer rings.
Water illustrates the importance of hydrogen bonding, both in its own properties and in its occurrence as the commonest ‘fibre plasticiser’. Below 0 °C, the hydrogen bonds are strong enough to hold the water molecules together as a crystalline solid, although the mass of the molecules is less than that of many substances (propane, butane, hydrogen sulphide, chlorine, nitrogen, oxygen, to name but a few) that are gases at this temperature. At atmospheric pressure, between 0 and 100 °C, water is a liquid of limited volume in equilibrium with water vapour. The water molecules are in a mobile dynamic equilibrium, with hydrogen bonds continually breaking and reforming. Above 100 °C (at atmospheric pressure), all the molecules disperse into a gas. At ordinary temperatures, therefore, hydrogen bonds are in a very sensitive state: they are on the verge of breaking and thus are easily affected by changes of temperature, by applied stresses and by chemical and structural changes. Deliberately or inevitably, this results in considerable effects in some fibre materials, such as cellulose and nylon. This affects fibre behaviour, processing and usage; the fibre molecules under various conditions may be held rigidly together, be free to move in a dynamic equilibrium or be completely free of one another, except for chain entanglements.
Hydrogen bonds can also form, as illustrated in Fig. 1.3, between —CO · NH— groups, which are found in polyamide and protein fibres.
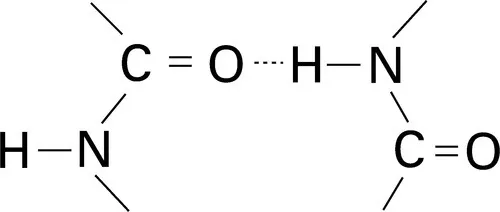
1.3 Schematic indication of hydrogen bonds between —CO · NH— groups.
In some fibres, there may be other bonds of intermediate strength. Thus, in the acrylic fibres, the asymmetry of electron arrangement in the —C ≡ N group, illustrated in Fig. 1.4, results in a moderately strong electrical interaction. In polyester fibres, and others based on aromatic polymers, there is an interaction between benzene rings.
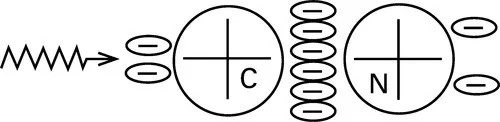
1.4 Electric dipoles in the acrylonitrile side group.
1.1.3 The nature of fibres
Fibres have been defined by the Textile Institute [3] as units of matter characterised by flexibility, fineness and a high ratio of length to thickness. To these characteristics might be added, if the fibre is to be of any use for g...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright page
- Preface to the first edition
- Preface to the fourth edition
- Woodhead Publishing in Textiles
- 1: An introduction to fibre structure
- 2: Testing and sampling
- 3: Fibre fineness and transverse dimensions
- 4: Fibre length
- 5: Fibre density
- 6: Thermal properties
- 7: Equilibrium absorption of water
- 8: Heats of sorption
- 9: Rate of absorption of moisture
- 10: The retention of liquid water
- 11: Swelling
- 12: Theories of moisture sorption
- 13: Tensile properties
- 14: The effects of variability
- 15: Elastic recovery
- 16: Rheology
- 17: Directional effects
- 18: Thermomechanical responses
- 19: Fibre breakage and fatigue
- 20: Theories of mechanical properties
- 21: Dielectric properties
- 22: Electrical resistance
- 23: Static electricity
- 24: Optical properties
- 25: Fibre friction
- Appendix I: Units
- Appendix II: Fibre names
- Appendix III: Standard test methods
- Name index
- Subject index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Physical Properties of Textile Fibres by J. W. S. Hearle,W E Morton in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Materials Science. We have over 1.5 million books available in our catalogue for you to explore.