
- 400 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Biomaterials and Devices for the Circulatory System
About this book
Cardiovascular disease is one of the leading causes of death in the world today. Thanks to major advances in circulatory biomaterials and medical devices over the past few decades, many complications of this prevalent disease can be managed with great success for prolonged periods.Biomaterials and devices for the circulatory system reviews the latest developments in this important field and how they can be used to improve the success and safety in this industry.Part one discusses physiological responses to biomaterials with chapters on tissue response, blood interface and biocompatibility. Part two then reviews clinical applications including developments in valve technology, percutaneous valve replacement, bypass technologies and cardiovascular stents. Part three covers future developments in the field with topics such as nanomedicine, cardiac restoration therapy, biosensor technology in the treatment of cardiovascular disease and vascular tissue engineering.With its distinguished editors and international team of contributors Biomaterials and devices for the circulatory system is a vital reference for those concerned with bioengineering, medical devices and clinicians within this critical field.
- Reviews the latest developments in this important field and how they can be used to improve success and safety in the industry
- Both current clinical advances as well as future innovation are assessed taking a progressive view of the role of biomaterials in medical applications
- An examination of the physiological responses to biomaterials features tissue responses to implanted materials and strategies to improve the biocompatibility of medical devices
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
MedicineSubtopic
Medical Technology & SuppliesPart I
Physiological responses to biomaterials
1
Tissue responses to implanted materials
I. Dimarakis and S.M. Rehman, University Hospital of South Manchester, UK
G. Asimakopoulos, University Hospitals Bristol NHS Foundation Trust, UK
Abstract:
Advances in cardiovascular procedures would have not been possible without the concomitant developments in biotechnology. Materials and devices used for the restoration of anatomical and physiological circulatory properties are associated with risks and potential complication. This chapter discusses the commonest forms of tissue response to implanted biomaterials.
Key words
cardiovascular procedures
implanted biomaterials
tissue responses
1.1 Introduction
Advances in cardiovascular procedures would have not been possible without the concomitant developments in biotechnology. The application of biomaterials spans from prosthetic heart valves, ventricular assist devices, cardiac pacemakers and endovascular implants, to suture material and bio-adhesives. Materials and devices used for the restoration of anatomical and physiological circulatory properties are not without associated risks and potential complication, both in the immediate as well as the long-term period, following implantation within the recipient.
Implant building blocks vary from an array of metals and their alloys, polymers and ceramics. Naturally occurring materials such as collagen, hyaluronan and dextran are commonly used. Synthetic polymers used for the construction of implantable devices include polylactic acid, polylactic-co-glycolic acid, polyvinyl alcohol and others (Onuki et al., 2008). The majority of implanted devices are composed of more than one type of material and are referred to as complex composition implants. Although these materials have been used extensively with good functional results, there are issues of biocompatibility. The need to surpass physical limitations and improve the biomechanical profile of metals has led to the development of novel metal alloys. In addition to superior physical and chemical properties regarding strength, durability and resistance to corrosion, certain alloys have very unique properties. An example is nickel-titanium (nitinol) alloys that exhibit shape memory and superelasticity, making it one of the most widely used materials in medical prostheses.
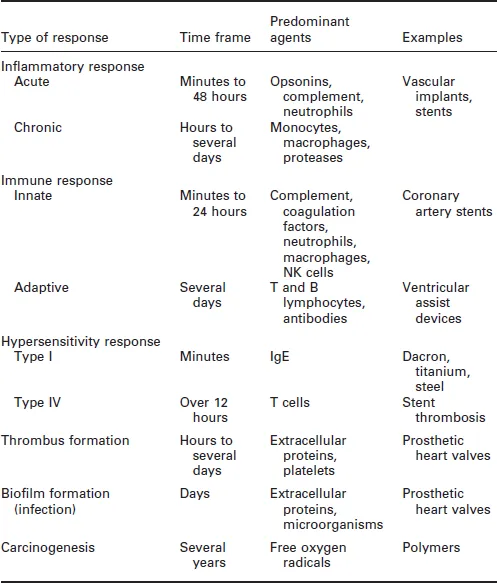
The tissue response to implanted materials is a complex process. It entails a variety of pathways that may include the generation and activation of interacting humoral and molecular components. These processes may be acute, taking place over minutes, or long term, spanning many years (Table 1.1). This chapter aims to present the commonest types of tissue response to materials used for the construction of implantable medical devices. It describes processes such as inflammation, immune response, hypersensitivity, formation of thrombus or biofilm and carcinogenesis. Each section contains a description of the molecular and cellular pathways involved in these processes.
Table 1.1
Commonest types of tissue response to implanted materials
1.2 Inflammatory response
Inflammation is the initial, non-specific response of vascularised tissue to a variety of injuries. It represents the attempt of the body to eliminate the injuring agent. Our knowledge of the complicated network of molecules that interconnects different inflammatory pathways during cardiac surgery started to expand at a later time following advances in basic sciences. The inflammatory response may be local or systemic. Although inflammation aims to exercise a healing effect against injury, the immune system may erroneously attack body tissues at a larger scale than necessary. The inflammation process comprises primarily activation of serum molecular cascades and responses of endothelium and leucocytes. It may be triggered by stimuli such as infection, immune reaction, physical injury or foreign bodies (Rock and Kono, 2008). Figure 1.1 illustrates mediators participating in acute inflammatory response.
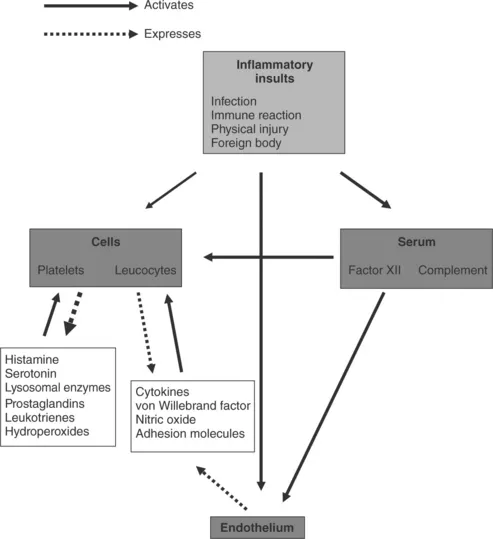
1.1 Mediators participating in the acute systemic inflammatory response.
Implantable materials are perceived as foreign bodies and elicit inflammatory response through injury to local tissues, which prompts the host to repair the damage. The immediate injury results in activation of endothelial cells and increased interaction with activated neutrophils. This process results in damage of endothelial integrity, increased endothelial permeability and tissue oedema. There is also activation of the coagulation cascade and components of the immune system. The products of these responses collectively form a provisional matrix consisting of fibrin, activated platelets, inflammatory cells and endothelial cells. The provisional matrix acts as a site for cell adhesion and provides activating and inhibiting substances to facilitate proliferation within the lesion. The process is well established and clearly illustrated in coronary stent insertion where the immediate injury is to vascular endothelium caused by stent strut penetration (Edelman and Rogers, 1998). The inflammatory response is a part of normal healing. However, abnormal proliferation of cells and formation of an extracellular matrix may result in intimal hyperplasia (Farb et al., 2004), which can cause significant stenosis and subsequent morbidity. A similar process is recognised at anastomotic sites in vascular bypass grafts (Glagov et al., 1991). One realises the importance of this fact as significant efforts have been made to design and produce anastomotic devices facilitating construction of coronary graft anastomoses (Falk et al., 2005; Gummert et al., 2007).
The inflammatory response consists of an acute and a chronic phase and is mediated by a vast array of chemicals released from the injured tissue, plasma and cells. These include vasoactive agents, plasma proteases (kinins,complement and coagulation systems), leukotrienes, lysosomal proteases, oxygen-derived free radicals, platelet activating factors, cytokines and growth factors (Anderson, 2004).
Acute inflammation lasts from minutes to 24 to 48 hours. It is characterised by changes in vascular calibre and endothelial permeability. The predominant cell type involved at this stage is the neutrophil, which migrates and binds to the site of injury via chemotactic agents and adhesion molecules respectively. This process results in extravasation of fluid and proteins causing oedema. Opsonins, especially IgG and C3b, promote the recognition and attachment of neutrophils to the surface of a device and may play a role in their activation and subsequent phagocytosis. However, normal phagocytosis does not occur due to the large molecules that tend to make up device materials. Instead leucocytes simply release chemicals in an attempt to degrade the device.
Chronic inflammation may occur naturally in response to prosthetic implants and may persist over long periods due to the continual presence of foreign material. It may follow acute inflammation. Furthermore, displacement or infection of a device are additional stimuli that promote a chronic inflammatory process. This is characterised by the presence of monocytes, macrophages and lymphocytes and proliferation of blood vessels and connective tissue. Monocytes and macrophages, along with cell types from various other tissues in the body, form the reticuloendothelial system. Macrophages are the most important cell in chronic inflammation as they release a variety of products that are involved in the tissue response to prosthetic implants and wound healing such as neutral proteases, chemotactic factors, arachidonic acid metabolites, reactive oxygen metabolites, complement components, coagulation factors, growth-promoting factors and cytokines (Anderson, 2004).
Granulation tissue, the hallmark of wound healing, can be seen as early as 3 to 5 days after device implantation. It is characterised by neovascularisation and proliferation of fibroblasts, which produce proteoglycans and collagen to form a fibrous capsule. Macrophages are also almost always present in granulation tissue. The extent of granulation tissue formation depends on the extent of tissue injury and the area of the defect, caused by injury that must be filled. The foreign-body reaction includes granulation tissue and foreign-body giant cell (FBGC) formation to varying degrees depending on an implant's size and surface properties. The presence of FBGCs and monocytes/macrophages may influence the strength of the implant-tissue integration and more monocytes/macrophages have been found to rest on smooth surfaces than on rough surfaces (Anderson, 2000). It has been further shown that surface smoothness may induce the differentiation of cells of the monocytic lineage and the formation of FBGCs out of the ce...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Contributor contact details
- Foreword
- Part I: Physiological responses to biomaterials
- Part II: Clinical application of biomaterials
- Part III: Future developments
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Biomaterials and Devices for the Circulatory System by Terence Gourlay,Richard A Black in PDF and/or ePUB format, as well as other popular books in Medicine & Medical Technology & Supplies. We have over 1.5 million books available in our catalogue for you to explore.