
- 816 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
The problem of stress corrosion cracking (SCC), which causes sudden failure of metals and other materials subjected to stress in corrosive environment(s), has a significant impact on a number of sectors including the oil and gas industries and nuclear power production. Stress corrosion cracking reviews the fundamentals of the phenomenon as well as examining stress corrosion behaviour in specific materials and particular industries.The book is divided into four parts. Part one covers the mechanisms of SCC and hydrogen embrittlement, while the focus of part two is on methods of testing for SCC in metals. Chapters in part three each review the phenomenon with reference to a specific material, with a variety of metals, alloys and composites discussed, including steels, titanium alloys and polymer composites. In part four, the effect of SCC in various industries is examined, with chapters covering subjects such as aerospace engineering, nuclear reactors, utilities and pipelines.With its distinguished editors and international team of contributors, Stress corrosion cracking is an essential reference for engineers and designers working with metals, alloys and polymers, and will be an invaluable tool for any industries in which metallic components are exposed to tension, corrosive environments at ambient and high temperatures.
- Examines the mechanisms of stress corrosion cracking (SCC) presenting recognising testing methods and materials resistant to SCC
- Assesses the effect of SCC on particular metals featuring steel, stainless steel, nickel-based alloys, magnesium alloys, copper-based alloys and welds in steels
- Reviews the monitoring and management of SCC and the affect of SCC in different industries such as petrochemical and aerospace
Tools to learn more effectively

Saving Books

Keyword Search

Annotating Text

Listen to it instead
Information
Part I
Fundamental aspects of stress corrosion cracking (SCC) and hydrogen embrittlement
1
Mechanistic and fractographic aspects of stress-corrosion cracking (SCC)
S.P. Lynch, Defence Science and Technology Organisation (DSTO), Australia
Abstract:
Basic aspects of stress-corrosion cracking (SCC) in metallic materials are outlined, followed by a summary of the numerous mechanisms that have been proposed for SCC. The characteristics of transgranular and intergranular SCC in model systems, e.g. pure metal and single-phase alloy single crystals and bi-crystals under testing conditions that facilitate discrimination between mechanisms, are then described. The applicability of the various proposed mechanisms, such as those based on dissolution, hydrogen embrittlement, film-induced cleavage, and adsorption, are discussed in detail for these systems. Mechanisms of SCC in complex commercial alloys are then considered in the light of these studies on model systems.
Key words
stress-corrosion cracking
cleavage-like cracking
intergranular cracking
mechanisms
fractography
adsorption-induced cracking
dissolution
hydrogen embrittlement
1.1 Introduction
Stress-corrosion cracking (SCC) is the generally accepted term for describing sub-critical cracking of materials under sustained loads (residual or applied) in most liquid and some gaseous environments. Sub-critical cracking of materials in gaseous hydrogen or hydrogen sulphide, and cracking due to internal hydrogen resulting from pre-exposure of materials to hydrogen-bearing environments, are considered to be forms of hydrogen embrittlement (HE) rather than SCC. However, SCC in some materials can involve generation and ingress of hydrogen at crack tips, and characteristics and mechanisms of SCC and HE have a lot in common. Sub-critical cracking in liquid-metal environments is also considered to be a separate phenomenon, usually called liquid-metal embrittlement (LME), but also has a number of similarities to SCC. An understanding of the mechanisms of HE and LME, which are not as complex as SCC, is therefore valuable in understanding SCC, and chapters on the fundamentals of HE (Chapter 2) and LME (Chapter 18) should be consulted in this regard.
SCC occurs in a wide range of materials/environments at rates varying from ~ 10− 2 m/s to < 10− 11 m/s (< 0.3 mm/yr) – with extremely low rates obviously significant in regard to the integrity of structures with projected lifetimes of 50 years or more. There are extensive databases regarding susceptible material:environment combinations [1–4], but failures involving SCC continue to occur, sometimes with catastrophic consequences. Many SCC failures occur because ‘old’ SCC-susceptible materials are present in ageing structures and components where it has not been economical to replace them with more recently developed SCC-resistant materials. In other cases, SCC failures occur because the detrimental environmental conditions have not been predicted, e.g. in crevices where impurities can concentrate due to evaporation/re-wetting cycles, or because testing conditions for determining SCC resistance have not been representative of service conditions.
Applied stress levels may also have been underestimated or residual stresses not considered. Transient conditions during start-up or shut-down of equipment, where environmental conditions, and stresses/strain-rates are often different from those during normal operation, are also not always taken into account during material selection and design. In other cases, the SCC resistance of welds (and associated heat-affected zones) may not have been fully considered, especially if welding of structures in practice has been carried out under different conditions than those used for the test specimens. Environmental conditions envisaged at the design stage can also be changed during service, e.g. to increase operational efficiencies or to slow down general corrosion, without fully considering the implications for SCC resistance.
A good example of the failure to take into account some of the above considerations led to SCC of an austenitic steel lever-arm-pin (worth about €10) in a military jet engine, leading to a series of events that caused the crash of the aircraft (worth about €10 million) (see Chapter 16). The particular alloy may not have been in any SCC databases, but it could (arguably) have been predicted that a (hot) concentrated aqueous chloride environment would have developed in crevices, that residual stresses would have been present, and that most austenitic alloys would be susceptible to SCC under such conditions. A better fundamental understanding of SCC by those who select materials and design structures and components would probably help prevent many failures.
In this chapter, various proposed processes and mechanisms of SCC are outlined, and then the applicability of these mechanisms for particular materials and environments are discussed. The present review of SCC takes a somewhat different approach from previous reviews on the topic by focusing on understanding SCC in model systems, e.g. pure metal and single-phase alloy single crystals and bi-crystals, in order to provide a sounder basis for understanding SCC in complex commercial alloys. Fractographic aspects of SCC are also emphasised more than in previous reviews. Before discussing SCC mechanisms, some basic aspects of SCC are summarised. More detailed coverage of these basic aspects and electrochemical/thermodynamical fundamentals can be found in previous reviews, conference proceedings, and books [3–23].
1.2 Quantitative measures of stress-corrosion cracking (SCC)
SCC can occur at remarkably low stresses in tensile specimens for some materials and environments, as illustrated by time-to-failure versus applied-stress data where threshold stresses for SCC can be as low as 5% of the yield stress. For pre-cracked specimens, threshold stress-intensity factors (Kth), can also often be only ~ 5% of the K value for fast fracture (KIc). Plots of crack velocity, v, versus K often show two regimes – region-I, just above KISCC, where the crack velocity increases rapidly with increasing K, and region-II where there is little or no dependence of crack velocity on K (termed the plateau velocity) (Fig. 1.1). In a few cases, several plateau velocities are observed, and there is sometimes a third region where crack velocity increases rapidly with increasing K just below KIc.
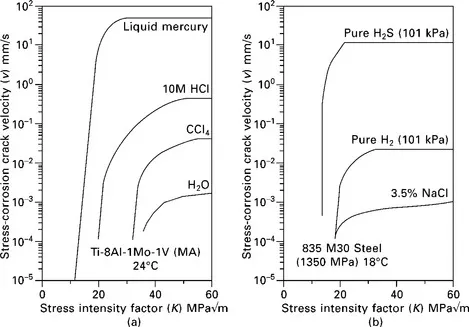
1.1 Plots of SCC velocity versus stress-intensity factor for (a) Ti alloy in various environments, (from data in [24]), and (b) high-strength steel in NaCl solution (from data in [25]). Data for LME (of a Ti alloy in mercury) and HE (of a steel in H2S and H2) are included for comparison.
Data from slow-strain-rate testing of smooth or notched tensile specimens are often used as a measure of SCC susceptibility, and are popular since these tests generally do not take as long as other tests. The time-to-failure, reduction-of-area, or extent of SCC on fracture surfaces, are used to assess the degree of susceptibility. Slow, rising-load tests on pre-cracked fracture-mechanics specimens are also used to obtain SCC data. Threshold K values may be lower under rising-load conditions than for sustained-load conditions, and may sometimes be more applicable to practical situations.
Threshold stresses can also be lower and crack-growth rates can be higher when small cyclic loads are superimposed on sustained loads. These so-called ‘ripple-load’ tests are equivalent to corrosion-fatigue at high R-ratios (0.9–0.95). For corrosion fatigue at low R ratios, SCC processes can be superimposed on fatigue processes when there are hold times at maximum loads. Laboratory testing rarely simulates the precise conditions in service so that the relevance of data from specific tests to structural-integrity and remaining-life estimations needs to be assessed on a case-by-case basis. Further details of test methods and their relevance can be found in Chapter 3 and elsewhere [26].
1.3 Basic phenomenology of stress-corrosion cracking (SCC)
1.3.1 Crack-initiation
Initiation of cracks is sometimes not necessary since structures and components may contain pre-existing crack-like defects, e.g. porosity, hot tears. Welds are especially prone to such defects. Machining can also produ...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Contributor contact details
- List of reviewers
- Foreword
- Preface
- Part I: Fundamental aspects of stress corrosion cracking (SCC) and hydrogen embrittlement
- Part II: Test methods for determining stress corrosion cracking (SCC) susceptibilities
- Part III: Stress corrosion cracking (SCC) in specific materials
- Part IV: Environmentally assisted cracking problems in various industries
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Stress Corrosion Cracking by V S Raja,Tetsuo Shoji in PDF and/or ePUB format, as well as other popular books in Technology & Engineering & Mining Engineering. We have over one million books available in our catalogue for you to explore.