
- 414 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Mechanisms of Morphogenesis
About this book
Morphogenesis is the set of processes that generate shape and form in the embryo--an important area within developmental biology. An exciting and up-to-the-minute account of the very latest research into the factors that create biological form, Mechanisms of Morphogenesis, second edition is a text reference on the mechanisms of cell and tissue morphogenesis in a diverse array of organisms, including prokaryotes, animals, plants and fungi.
By combining hard data with computer modeling, Mechanisms of Morphogenesis, second edition equips readers with a much broader understanding of the scope of modern research than is otherwise available. The book focuses on the ways in which the genetic program is translated to generate cell shape, to direct cell migration, and to produce the shape, form and rates of growth of the various tissues. Each topic is illustrated with experimental data from real systems, with particular reference to gaps in current knowledge and pointers to future
- Includes over 200 four-color figures
- Offers an integrated view of theoretical developmental biology and computer modelling with laboratory-based discoveries
- Covers experimental techniques as a guide to the reader
- Organized around principles and mechanisms, using them to integrate discoveries from a range of organisms and systems
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
SECTION III
Cell Migration
Chapter 7 Cell Migration in Development
Chapter 8 The Nano-Machinery of Locomotion
Chapter 9 Guidance by Chemotaxis
Chapter 10 Guidance by Galvanotaxis
Chapter 11 Guidance by Contact
Chapter 12 Waypoint Navigation in the Embryo
Chapter 13 Cooperative Migration of Mesenchymal Cells
Chapter 14 Condensation of Cells
Chapter 7
Cell Migration in Development
A Brief Overview
Cell migration takes place at least at some point in the life cycle of almost all animals. For some, such as coelenterates, it is an important but rather rare event, whereas for others, such as vertebrates, cell migration is involved in the morphogenesis of most parts of the body. This chapter will present an overview of the broad types of cell migration that are common in development: the chapters that follow will examine in detail the cellular mechanisms involved in their guidance and regulation.
Cell migration of one sort or another is an almost universal attribute of sexual reproduction, for the obvious reason that two gametes produced in different places, and usually in different individuals, have to unite. Some organisms, usually ones that are anatomically simple, produce gametes that are morphologically identical (isogamous) even if they happen to be divided into two biochemically distinct ‘mating types’. In these cases, the gametes share the task of finding each other. This does not always involve cell migration; in the filamentous alga Spyrogyra, for example, gametes remain in the organism that produces them and find each other simply by extending a cell process towards the gametes of an adjacent individual1 (Figure 7.1a). In most cases, though, adults are not present at a high enough density for this to be feasible, and gametes have to migrate by crawling or swimming. In both plants and animals, migration has favoured the loss of isogamy and the specialization of gametes either to be nutrient-rich and relatively immobile (ova), or numerous, small and specialized for migration (sperm).
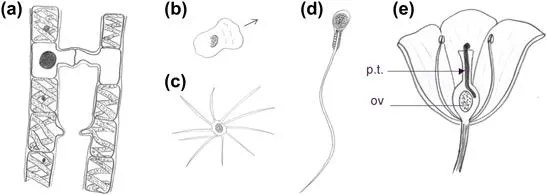
FIGURE 7.1 Mechanisms for bringing gametes together. (a) Union of gametes by extension of cell processes in the isogametic alga Spyrogyra. The upper conjugation is seen at a late stage, after union of gametes, while the lower one is just beginning. (b) Amoeboid sperm of the nematode Caenorhabditis elegans, drawn from a phase contrast micrograph.2 (c) Sperm of the decapod Inacnus, showing actin-rich extensions. (d) Human sperm, showing a single long flagellum. (e) The pollen tube (p.t.) of a flowering plant, growing towards the ovule (ov); the size of the pollen tube is exaggerated for clarity.
Sperm use a variety of methods for migration (Figure 7.1). Those of nematodes are approximately amoeboid in shape and move by amoeboid motion (Figure 7.1b). Those of decapod crustaceans (crabs, lobsters, and so on) have a large number of actin-containing processes3 (Figure 7.1c). Mammalian sperm, and those of many other animals and algae, move by the whiplash action of flagella (Figure 7.1d). In many species, sperm is attracted towards eggs of the correct species by chemotaxis (see Chapter 9). Male gametes of primitive plants, such as liverworts (Hepaticae), also swim using flagella but in flowering plants the meeting of sperm and egg nuclei is achieved by the extension of a tube from the pollen, which invades the female parts of the flower and brings the male gamete nucleus to the ovum (Figure 7.1e).
Important as it is to development, fertilization is not morphogenesis. During post-fertilization development, cell migration and locomotion contribute directly to morphogenesis in four main ways:
1. Bringing dispersed cells together to aggregate in one place
2. Moving a group of cells from one place in the body to another
3. Dispersing cells created in one place to a variety of sites
4. Connecting cells and cell processes together in a specific network, such as the nervous system.
There are also many cell rearrangements that operate on too short a range to be considered true migrations and are therefore discussed in Chapter 16 rather than here.
Morphogenesis by Coalescence of Dispersed Cells
One of the simplest examples of cell migration effecting true morphogenetic change is seen in the social amoeba Dictyostelium discoideum. Where food sources are plentiful, D. discoideum exists as a community of scattered myxamoebae, which are amoeba-like cells that roam decaying wood on forest floors and prey on bacteria. When food becomes scarce, the myxamoebae begin to emit a chemotactic signal and, through sensing and information processing systems that will be discussed in detail in Chapter 9, they move toward sources of this signal and therefore toward each other. Their migration results in the formation of streams of moving cells that converge to produce a tight, conical aggregate (Figure 7.2). This aggregate then behaves as a single organism, and later processes of differentiation within it turn it into a migratory grex, sometimes called a ‘slug’ because of its superficial resemblance to these animals. The grex moves away and, as it does so, some cells differentiate to form spores. It then stops migrating, raises these spores into the air atop a long stalk, and releases them so that they disperse. Where they land, they form further myxamoebae and the cycle repeats.
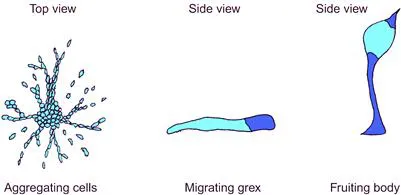
FIGURE 7.2 Formation of the grex of D. discoideum by chemotaxis-driven aggregation of myxamoebal cells.
Morphogenesis by coalescence of migrating cells is seen many times in the development of higher animals too. The great blood vessels of vertebrates, for example, form in this way. In embryos of the frog Xenopus laevis, precursor cells of the vascular system arise in the lateral mesoderm, 200–300 μmref4 from where they will eventually be needed (Figure 7.3). They migrate from these sites towards the axis of the embryo, specifically towards the hypochord area, which lies just ventral to the notochord. Once they have converged on the midline, the cells differentiate into endothelium and adhere to each other to form the dorsal aorta, the vessel that carries high-pressure blood from the heart to the trunk of the body. The hypochord expresses a diffusible growth factor (VEGF) and the precursor cells express a receptor for it (flk-1), suggesting that the cells navigate up the concentration gradient of VEGF. This hypothesis is strengthened by the observations that if an ectopic source of VEGF is applied to an X. laevis embryo, the vessel precursor cells migrate towards this as well,4 and that dorsal aorta development fails in vegf −/− mice.5 The same general pool of vascular precursors in the lateral mesoderm also gives rise to other parts of the vasculature, and the process by which the precursor pool is divided is not yet understood. Morphogenesis of blood vessels by coalescence of migratory cells, usually called ‘vasculogenesis’, is not the only way that blood vessels form; smaller vessels often form by branching from existing vessels in a process that is called ‘angiogenesis’ and that will be described in Chapter 20.
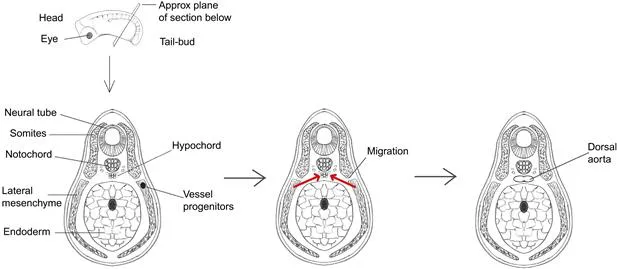
FIGURE 7.3 Morphogenesis of the dorsal aorta of Xenopus laevis by coalescence of migrating angioblasts, the precursors of blood vessels, which arise in the lateral mesoderm.
Coalescence of cells is not confined to foetal development; the coming together of migrating cells is an important mechanism in the morphogenesis of lymphatic tissue, which continues to develop and remodel through adult life. In the spleen, arterioles that derive from the splenic artery ramify through the organ and become sheathed by lymphoid cells, mainly in the form of lyphoid follicles. The follicles form when stromal cells at the sites of presumptive follicles secrete a chemokine, CXCL13.6 B lymphocytes bear a receptor for CXCL13, CXCR5,6 and it is assumed that CXCL13, perhaps with other chemokines, is responsible for attracting migrating B cells to the presumptive follicle. Certainly, B cells will migrate towards sources of CXCL13 in culture7 and mice lacking CXCR5 fail to form follicles.8 Relatively short-range movements of mesenchymal cells are also involved in the formation of cell ‘condensations’, a process so important to development that it will be discussed separately in Chapter 14.
Translocation of Groups of Cells from One Place to Another
As well as driving morphogenesis through cell aggregation, cell migration can be used as a means of cell dispersal or for the translocation of cells from the place of their birth to the place of their final use. An illustration is provided by the primordial germ cells of vertebrate embryos, which give rise to the gametes discussed at the beginning of this chapter. Primordial germ cells arise, early in development, far outside the gonads that will eventually be their home (Figure 7.4). For instance, in frogs, they develop in the posterior region of the gut and then migrate anteriorly along its dorsal surface, out along the dorsal mesentery (a sheet of tissue that connects the gut to the rest of the abdomen), up the abdominal walls into the genital ridges and along the ridges to the developing gonads.9–11 In mammals, primordial germ cells arise near the junction between the embryo-proper and the allantois, an extraembryonic structure, and invade the endoderm of the embryo, which will form the gut.12 Once there, they migrate anteriorly along the gut and enter the genital ridges13 (Figure 7.4). It used to be believed that mammalian primordial germ cells migrated via the allantois, but it now seems that those cells taking this route are ‘lost’ and do not contribute to the germ line; cells migrating into the endoderm are the true source of mammalian gametes.12,14
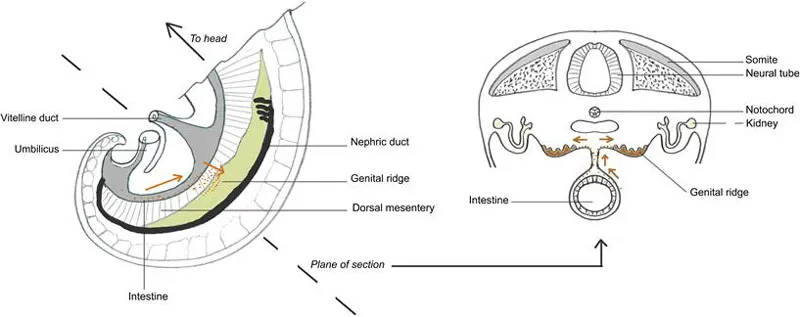
FIGURE 7.4 Migration of primordial germ cells in the mouse embryo.
Sometimes, groups of migrating cells really do move as a coherent, cooperative group, integrating the guidance cues they receive and passing ‘traffic control’ signals to one another to avoid cellular traffic jams. Although the existence of collective cell migration has been suspected for decades, there has been a sudden surge of interest in the phenomenon, stimulated partly by studies of the principles of efficient traffic flow in the human-scale world. For this reason, a new chapter (13), devoted to the topic, has been added to this second edition of this book.
Dispersal of Cells from One Site to the Rest of the Body
One of the most spectacular demonstrations of cell migration in development is provided by the vertebrate neural crest. The neural crest arises from the dorsal-most zone of the neural tube (presumptive brain and spinal cord) (Figure 7.5), and it gives rise to a multitude of cell types in locations often far removed from the neural tube itself. The face, for example, is produced primarily fr...
Table of contents
- Cover image
- Title page
- Table of Contents
- Copyright
- Dedication
- Preface
- A Note on References
- SECTION I: Introductory Section
- SECTION II: Cell Shape and Cell Morphogenesis
- SECTION III: Cell Migration
- SECTION IV: Epithelial Morphogenesis
- SECTION V: Morphogenesis by Cell Proliferation and Death
- SECTION VI: Modelling Morphogenesis
- SECTION VII: Conclusion and Perspectives
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Mechanisms of Morphogenesis by Jamie A. Davies,Jamie Davies in PDF and/or ePUB format, as well as other popular books in Biological Sciences & Biology. We have over 1.5 million books available in our catalogue for you to explore.