Abdomen
Fatima H. Naeima1, Zainab H. Ibrahim1, Sarah N. Ahmed1, Hayder D. Abbas2, Karrar Z. Sadoun2, Houreleen H. Salman3, Sultan M. Ghanim4, * 1 6th Grade Medical College, Kufa University Diwanya, Iraq
2 5th Grade Jabir ibn Hayyan Medical University Najaf, Iraq
3 6th Grade Medical College, Kufa University Dhi-Qar, Iraq
4 ME Unit, Medical College, University of Kufa, Iraq
Abstract
In this chapter we discuss common and unusual conditions of the abdomen that are treated surgically, related to the stomach, small intestine and colon. Such as Hypertrophic pyloric stenosis, is one of the most common surgical conditions of the newborn. Congenital intestinal obstruction occurs in approximately 1 in 2000 live births and is a common cause of admission to a neonatal surgical unit. Normal rotation of the intestine requires transformation from a simple, straight alimentary tube into the mature fixed and folded configuration normally present at birth. Through precise embryologic events, the duodenojejunal junction become fixed in the left upper abdomen while the cecum is anchored in the right lower quadrant. The midgut, defined as the portion of the intestine supplied by the superior mesenteric artery (SMA), is thus suspended from a wide mesenteric base.
Keywords: Intestine, Stomach, Surgery.
* Corresponding author Sultan M. Ghanim: ME Unit, Medical College, University of Kufa, Iraq;
Tel: +9647816669997; E-mail: [email protected] HYPERTROPHIC PYLORIC STENOSIS
HPS is one of the most common surgical conditions of the newborn [1]. It occurs in approximately 1 in 300 live births and commonly in infants between 3 and 6 weeks of age. Male-to-female ratio is nearly 5:1, and there are several risk factors for HPS include family history, gender, younger maternal age, being a first-born infant, and maternal feeding patterns [2, 3].
Etiology
- Genetic
- 2. Environmental factors such as (method of feeding (breast vs. formula), seasonal variability, and exposure to erythromycin, environmental pesticides, and Trans pyloric feeding in premature infants).
Clinical Presentation
Infants with hypertrophic pyloric stenosis (HPS) typically present with non- bilious vomiting that becomes increasingly projectile, over the course of several days to weeks due to progressive thickening of the pylorus muscle.
Physical Examination
The neonate usually appears well if the diagnosis is made early depending on the duration of symptoms and degree of dehydration, the neonate may be gaunt and somnolent. Visible peristaltic waves may be present in the mid to left upper abdomen. So, the pylorus is palpable in 70–90% of patients [4, 5]. To palpate the pyloric mass (i.e. , “olive”), the neonate must be relaxed. Techniques for relaxing the patient include bending the newborn’s knees and flexing the hips, using a pacifier with sugar water. These techniques should be attempted after the stomach has been decompressed with 10 French to 12 French oro-gastrictubes,after palpating the liver edge, the examiner’s fingertips should slide underneath the liver in the midline. Slowly, the fingers are pulled backdown, trying to trap the “olive. ” Palpating the pylorus requires patience and an optimal examination setting. If palpated, no further studies are needed.
Imaging
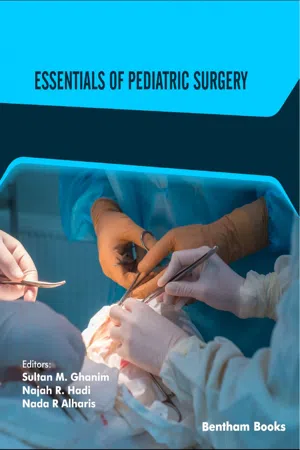
By ultrasound, the diagnostic criteria for pyloric stenosis are a muscle thickness of ≥4 mm and a pyloric length of ≥16 mm (Fig. 4.1) [6]. A thickness of >3 mm is considered positive if the neonate is younger than 30 days of age.
Fig. (4.1)) Ultrasonography has become the standard imaging study for diagnosing pyloric stenosis and has supplanted physical examination at most institutions. The (A) transverse and (B) longitudinal views of hypertrophic pyloric stenosis are seen here. Muscle thickness ≥4 mm on the transverse view or a length ≥16 mm on the longitudinal view is diagnostic of pyloric stenosis. On this study, the pyloric wall thickness was 5 mm and the length (arrows) was 20 mm.
Treatment
The mainstay of therapy is typically resuscitation followed by pyloromyotomy (Fig. 4.2). There are reports of medical treatment with atropine and pyloric dilation, but these treatments require long periods of time and are often not effective [7, 8].
Preparation for Surgery
Feedings should be withheld. Gastric decompression is usually not necessary but occasionally may be required in extreme cases. If a barium study was performed, it is important to remove all of the contrast material from the stomach to prevent aspiration. The hallmark metabolic derangement of hypochloremic, hypokalemic metabolic alkalosis is usually seen to some degree in most patients. Profound dehydration is rarely seen today, and correction is usually achieved in <24 hours after presentation. A basic metabolic panel should be ordered, and the resuscitation should be directed toward correcting the abnormalities. Most surgeons use the serum carbon dioxide (<30 mmol/L), chloride (>100 mmol/L), and potassium (4.5–6. 5 mmol/L) levels as markers of adequate resuscitation. Initially, a 10- to 20-mL/kg bolus of normal saline should be given if the electrolyte values are abnormal. Then D5/½NS with 20–30 mEq/L of potassium chloride is started at a rate of 1. 25–2 times the calculated maintenance rate. Electrolytes should be checked every 6 hours until they normalize and the alkalosis has resolved. Subsequent fluid boluses are given if the electrolytes remain abnormal. Then the patient can safely undergo anesthesia and operation. It is important to appreciate that HPS is not a surgical emergency and resuscitation is of the utmost priority. Inadequate resuscitation can lead to postoperative apnea due to decreased respiratory drive secondary to metabolic alkalosis [9].
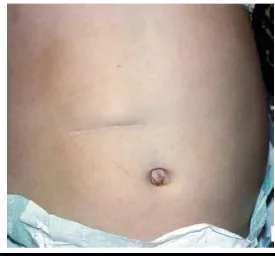
Fig. (4.2)) This child underwent opens pyloromyotomy through a right upper quadrant transverse incision. Over time, the cosmetic appearance of his incision is not as attractive as that seen after the laparoscopic operation.
Types of Surgery
- ● Open pyloromyotomy
- ● Laparoscopic pyloromyotomy
Postoperative Care
IV fluids are continued for several hours. After which Pedialyte is offered, followed by formula or breast milk, which is gradually increased to 60 cc every 3 hours. Most infants can be discharged home within 24 to 48 hours following surgery. Recently, several authors have shown that ad lib feeds are safely tolerated by the neonate and result in a shorter hospital stay.
Complications
- mucosal perforation
- wound infection
- incision hernia
- prolonged postoperative emesis
- incomplete myotomy
- duodenal injury
Intestinal Obstruction
Duodenal and Intestinal Atresia and Stenosis
The duodenum is the most common site for intestinal obstruction in neonate, occurs in 1 per 500010,000 live births, affecting boys more commonly than girls [10]. Trisomy 21 is found in almost half the cases. About 20% of patients with duodenal atresia have cardiac anomalies. Esophageal atresia may accompany duodenal atresia. In congenital duodenal atresia, the obstruction is distal to the ampoule of Vater in 85% of patients. A complete obstruction may be a luminal web (most common), an atresia connected by a fibrous cord, or complete separation of proximal and distal segments. The ampoule is located close to the medial wall of the web, and care must be taken to avoid injuring it during repair. A fenestrated duodenal Fig. (4.5) web with incomplete obstruction may not be noted at birth but can be seen later with feeding intolerance or obstruction when solid foods are started.
Etiology
Congenital duodenal obstruction can occur due to an intrinsic or extrinsic lesion [11]. The most common cause of duodenal obstruction is atresia and other causes are duodenal web, duodenal stenosis, annular pancreas, duodenal duplication cyst, preduodenal portal vein may cause obstruction of the duodenum. A “windsock” deformity can suggest a more distal obstruction.
Classification System
Anatomically, duodenal obstructions are classified as either atresia or stenosis. An incomplete obstruction, due to a fenestrated web or diaphragm, is considered a stenosis. Most stenosis involves the third and/or fourth part of the duodenum. Atresia is a complete obstruction.
Atresia is classified into three morphologic types (Fig. 4.3).