Establishing ethical and privacy protection aspects in scientific research, especially in medical research, has a long history. Medical data are usually more sensible than other personal data and require therefore an even higher degree of protection than other personal data. In recent research projects genetic evaluations become more and more important and trigger thereby new and continuing activities in the context of data protection. Genetic data as a subset of medical data are the most sensible category of personal data and require therefore the highest degree of data protection.
The book provides a systematic and itemized approach to data protection in clinical research including the handling of genetic material, genetic samples as well as derived genetic data and the subsequent secure storage of them. The set up of different kinds of clinical trials having in addition a genetic part, the concept of a genetic informed consent as well as collection schemes of samples are described in detail. Technical requirements and aspects of data protection including pseudonymization and anonymization procedures taking into account ethics committees requirements as well as the underlying legal framework are also presented.
Without any exception, all principles and methods presented are best practices, repeatedly applied in different clinical environments and by no means theoretical considerations.

eBook - ePub
G3P - Good Privacy Protection Practice in Clinical Research
Principles of Pseudonymization and Anonymization
- 210 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
G3P - Good Privacy Protection Practice in Clinical Research
Principles of Pseudonymization and Anonymization
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
Medicine1 Introduction
When evaluating clinical data and related samples from patients within a clinical trial, we are subjected to the Good Clinical Practice (GCP) principles, i.e. we are accustomed to provide a certain and appropriate degree of privacy with respect to the results of that trial (cf. (1)).
Guideline for good clinical practice E6 (R1)
2.11 The confidentiality of records that could identify subjects should be protected, respecting the privacy and confidentiality rules in accordance with the applicable regulatory requirement (s).
Now that the genetic mapping technologies have gradually become so revealing, dealing with genetic data requires an even higher degree of privacy to be provided to all steps of the clinical trial process, especially to the results obtained. We are becoming more and more able to identify genetic factors that may indicate increased risks of diseases but are not uniquely the determinants of diseases. These new possibilities of predicting the development of diseases did simultaneously trigger ethical and legal initiatives toward solutions that will protect individuals against the use of genetic data against their interests, e.g. for the aptitude for employment, insurances for health and life as well as credit-worthiness (cf. (2), (3)).
These thoughts provide only a few arguments why genetic data are considered to be highly sensitive and thus require a different approach with respect to the data protection process as is usually implemented in the context of clinical data only. The special status of human genetic data has been already emphasized in Article 4 of the International Declaration on Human Genetic Data (cf. (4))
Article 4: Special status
- (a) Human genetic data have a special status because:
- (i) they can be predictive of genetic predispositions concerning individuals;
- (ii) they may have a significant impact on the family, including offspring, extending over generations, and in some instances on the whole group to which the person concerned belongs;
- (iii) they may contain information the significance of which is not necessarily known at the time of the collection of the biological samples;
- (iv) they may have cultural significance for persons or groups.
There are numerous publications all focusing on the topics and terms arising in the context of genetic data processing, but there is no common terminology yet in that upcoming new field of clinical trial data processing; “the most universal quality is diversity”, (Michel de Montaigne). Even the same terms are defined differently by various sources, and only few regulations are available that cover the area of handling genetic data within clinical trials. (cf. (5), (6), (7), (8))
Bear in mind that authorities in general publish only very few regulations. Most of the publications are conceived as guidances containing nonbinding recommendations.
Nevertheless, those recommendations are widely understood as a broad hint how authorities expect procedures to be implemented.
New procedures must meet the recommendations of authorities to be considered as implicit regulations, as well as the needs of industry itself. An important additional factor that must not be underestimated and that must be considered before coming to a final decision is the public opinion.
It might be a wise decision if the procedures being implemented provide a certain degree of conformity with the public opinion with respect to the sensitive area of handling genetic data.
Possibly the decision to be taken is a balance between rational necessities on the one hand and political/public expectations on the other hand.
In any case, procedures to be implemented must allow adjustments to upcoming new regulations without major efforts.
When working in this context your interest should be focused on but not be limited to the following topics:
- – Agreed Terminology in genetic data processing, i.e. unambiguously define the terms you use.
- – Relevant Legal Requirements, laws, directives, regulations, i.e. be informed about legal requirements and/or boundaries that must be taken into account.
- – Data Protection – protect the privacy of personal data, i.e. develop sensitivity and respective measures to guarantee a necessary and high level of data protection.
- – Data Security – control the access to personal data, i.e. protect the physical security of personal data, adjust data security measures to the actual standards.
- – Informed Consent for the participation in the clinical as well as in the genetic part of a clinical study – agreement which evaluations may be performed.
In addition, processes must be in place to handle:
- – withdrawals from either part of a clinical study, clinical as well as genetic one at any time and if ever possible,
- – and survey the workflow of samples collected for genetic analysis,
- – possible restrictions to genetic analysis required most likely by Ethics Committees resp. IRBs,
- – the coding or anonymization of data and samples,
- – the secure storage of genetic data, original as well as derived data,
- – requests for a common statistical analysis of coded (e.g. anonymized) clinical and related genetic data.
2 Study Modes
Prior to starting a clinical trial intended to have a genetic part, there must be a clear concept about which role genetics should play within the upcoming trial.
Four different approaches are currently imaginable:
2.1 Screening Mode – Pharmacogenetic Information for Screening
If genetic information is only used in order to select patients to be enrolled in a clinical study, we usually speak of Screening Mode, because patients are screened according to some genetic pattern, and the verification of a special pattern is mandatory for inclusion in the planned study.
The decision process when using screening mode is visualized in Fig. 2.1.
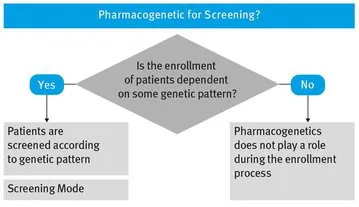
Fig. 2.1 Study Screening Opportunities
2.2 Pre-Unspecified Mode – Pharmacogenetic as Part of a Study
When planning to supplement the clinical part of a study by some kind of genetic analysis, two cases must be distinguished:
- – We know for certain, prior to the start of the study, which kind of genetic analysis (genetic tests) should be performed. In this case, all genetic tests to be carried out must be prespecified in the Clinical Trial Protocol (CTP). We speak of Prespecified Mode.
- – We do not know for certain, prior to the start of the study, which kind of genetic analysis should be performed. In this situation we cannot specify genetic tests to be carried out in the CTP, i.e. genetic tests to be carried out later remain unspecified at that moment. Therefore, we speak of Unspecified Mode.
The decision process when to use which mode is visualized in Fig. 2.2.
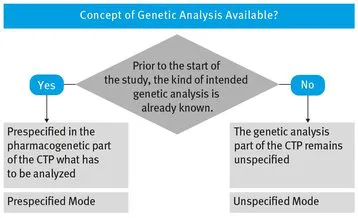
Fig. 2.2 Opportunities, if Genetic Analysis is Defined
Is it possible to apply both modes within one study?
Yes, it is.
- – If you have a concrete idea with respect to genetic analysis, start with prespecified mode, and if you think there could be some more genetic analysis needed in the future, collect additional samples for later analysis (unspecified mode).
What does this mean in practice, how to proceed?
- – Prespecified Mode – essentially „business as usual“.
- – Unspecified Mode – anonymization of data and samples is required prior to the start of a genetic analysis (cf. later).
2.3 Possible Approaches, Summary – Clinical Trial With Genetic Part
There are four possible approaches to supplement a clinical study with a genetic part (Fig. 2.3):
- – If the genetic part is used as inclusion criteria, i.e. the verification of a specific genetic pattern is needed for the inclusion of a subject, you have to apply Screening Mode (genotyping)
- – If you know in advance (prior to the start of the study), which genetic tests are to be evaluated later, you apply Prespecified Mode
- – If you do not want or cannot specify in advance which genetic tests are to be evaluated later, then samples are collected, the DNA is extracted and stored for later analysis. You have to apply Unspecified Mode
- – If you know in advance (prior to the start of the study), which genetic tests are to be evaluated later, and if you want to collect in addition samples for later analysis, then you have to apply Prespecified Mode on the first stage, andUnspecified Mode on the second stage

Fig. 2.3 Alternative Study Modes
When applying prespecified mode, the following facts should be known:
- – A trace back to the patient‘s identity is possible, but only the investigator knows the patient‘s identity.
- – Data (clinical as well as genetic data) are de-identified by a code, e.g. PAT_NO (Patient Number).
- – Essentially, samples will be destroyed after reporting of results.
- – Genetic data analysis can be part of a submission....
Table of contents
- Also of Interest
- Title Page
- Authors
- Copyright Page
- Preface
- Table of Contents
- About the Authors
- 1 Introduction
- 2 Study Modes
- 3 Protection Masks and Procedures
- 4 Coding Methods for De-identified Samples/Data
- 5 Relationships Among the Protection Masks
- 6 Data Types
- 7 Anonymization
- 8 Validation – a Brief Introduction
- 9 Request Management
- 10 Legal Requirements & Regulations
- 11 Informed Consent
- 12 Selected Data Protection & Medical Sites
- 13 Impact of External Services on Data Protection
- 14 Practical Approach to Clinical Trials with SupplementaryGenetic Parts
- Appendix
- 20 Abbreviations
- 21 References
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access G3P - Good Privacy Protection Practice in Clinical Research by Karl-Heinz Schriever,Markus Schröder in PDF and/or ePUB format, as well as other popular books in Medicine & Medical Theory, Practice & Reference. We have over 1.5 million books available in our catalogue for you to explore.