![]()
David J. Nelson, Catherine S. J. Cazin, Steven P. Nolan
1Grignard Reagents and Palladium
1.1Introduction
One contributor to the success of palladium-catalyzed cross-coupling as a technique in academia and industry has been the ability to make use of a wide range of reagents that are commercially available and/or relatively straightforward to prepare. Grignard reagents [1]are some of the most common and widely known organometallic reagents in chemistry [2]. While these are relatively reactive species, they do not typically require the cryogenic conditions that their organolithium counterparts frequently demand. Yet, many coupling reactions of Grignard reactions occur at, or close to, room temperature.
The use of these valuable reagents in cross-coupling has been known for decades, since the studies of Kharasch et al. in the early 1940s [3, 4]. The cross-coupling of Grignard reagents with aryl halides is often referred to as the “Kumada-Tamao-Corriu reaction” (or simply, the “Kumada coupling”), but we have chosen not to ascribe a name to the reaction here.
In this chapter, we briefly examine the historical development of the cross-coupling of Grignard reagents, and discuss the modern applications of the palladium-catalyzed variants. While we focus here only on palladium catalysis, Shinokubo and Oshima have published a micro review that includes examples of catalysis with a wider range of metals [5], and Tamao has reviewed the development of the nickel-catalyzed variant [6]. We consider the reactions of different classes of Grignard reagent (aryl, vinyl, alkyl and alkenyl) with different classes of electrophile (aryl, vinyl, alkyl and alkenyl), with the aim of providing the chemist with an appreciation of what is possible using this method. We also briefly describe and discuss some examples from target syntheses, and from larger-scale industrial applications.
1.2The Discovery and Development of Catalytic Cross-Coupling Reactions of Grignard Reagents
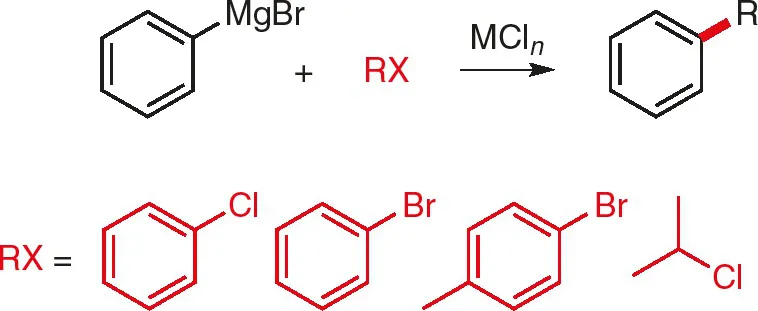
The earliest studies originated from the laboratory of Kharasch, who demonstrated that various metal salts catalyzed the coupling of some aryl halides (and one alkyl halide) with phenyl magnesium bromide (Fig. 1.1) [3]; CoCl2 was shown to be the most efficacious (at loadings of 2.5–9 mol %), although MnCl2, FeCl2 and NiCl2 also showed some (lesser) catalytic activity.
Fig. 1.1: Cross-coupling of phenyl magnesium bromide; MCln = CoCl2, MnCl2, FeCl2, or NiCl2 [3].
A subsequent study further probed the cobalt-catalyzed protocol, achieving the cross-coupling of phenyl-, cyclohexyl-, benzyl-, butyl- and naphthyl-magnesium bromide with several vinyl bromide and chloride compounds in the presence of typically 5 mol % CoCl2 [4]. However, the corresponding styrenes were not the only product, and in many cases were not even the major product; polymers and protodehalogenated products were also obtained. Therefore, while these studies represented a significant step towards catalytic coupling of Grignard reagents, these protocols were not practically useful for synthetic processes. Alternative systems based on iron(II) and iron(III) chloride complexes were disclosed by Kochi, in which alkylmagnesium bromides were coupled with vinyl bromides [7].
A watershed moment in the development of Grignard cross-coupling was the use of catalytic quantities of nickel complexes, at loadings as low as 0.1 mol % in some cases, to selectively prepare the desired products. It was at this point that the technique began to emerge as a potentially g...