![]()
Free Radical Biology
Naito Y, Suematsu M, Yoshikawa T (eds): Free Radical Biology in Digestive Diseases.
Front Gastrointest Res. Basel, Karger, 2011, vol 29, pp 35-54
______________________
Neutrophil-Dependent Oxidative Stress in Inflammatory Gastrointestinal Diseases
Yuji Naito · Toshikazu Yoshikawa
Molecular Gastroenterology and Hepatology, Kyoto Prefectural University of Medicine, Kyoto, Japan
______________________
Abstract
Gastrointestinal inflammation is a highly complex biochemical protective response to cellular/tissue injury. When this process occurs in an uncontrolled manner, the result is excessive tissue damage that results chronic inflammation and destruction of normal tissue, and may associate with carcinogenesis. Current evidence suggests that neutrophil-dependent oxidative stress plays a crucial role in the pathogenesis of gastrointestinal inflammation associated with Helicobacter pylori infection, nonsteroidal anti-inflammatory drug ingestion, ischemic condition and inflammatory bowel disease. It has been shown that the neutrophil-vascular endothelial cell interaction is regulated by various cell adhesion molecules, and that this interaction is directly or indirectly modified by many factors, including bacterial products, chemokines and gaseous mediators. Activated neutrophils can produce reactive oxygen/nitrogen species, myeloperoxidase, and elastases. This review describes the potential role of neutrophils and neutrophil-dependent oxidative stress in gastrointestinal disease.
Copyright © 2011 S. Karger AG, Basel
Although it is clear that genetic, environmental, and immunological factors affect the pathophysiology of the gastrointestinal diseases, recent studies have demonstrated that oxidative stress induced by the infiltration and activation of neutrophils plays a crucial role in the pathophysiology of inflammation of the gastrointestinal tract. Many experimental studies using neutrophil-deficient animals have clearly indicated the important role of neutrophils in the pathogenesis of the gastrointestinal inflammation, including Helicobacter pylori-induced gastritis, gastrointestinal inflammation induced by nonsteroidal anti-inflammatory drugs (NSAIDs) ingestion, and inflammatory bowel disease. The sequence of events in the infiltration and the activation of neutrophils have also been clarified, and some candidates have been proposed as a therapeutic target for these diseases during the last decade. More importantly in the clinical field, several studies have showed that granulocyte adsorptive apheresis therapy could induce the remission stage especially in patients with ulcerative colitis and Crohn's disease. Here, we review recent advances in molecular mediators of the infiltration and activation of neutrophils in gastrointestinal inflammatory disease.
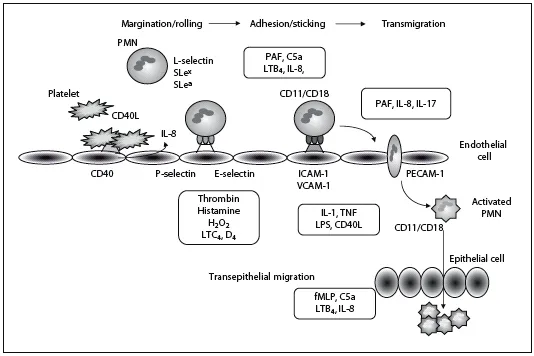
Fig. 1. Neutrophil-endothelial interaction mediated by adhesion molecules and chemical mediators. PAF = Platelet activating factor; PECAM-1 = platelet endothelial adhesion molecule 1; fMLP = N-formyl methionyl phenylalanine; LTC4 = leukotriene C4.
Molecular Mediators in Neutrophil Infiltration
Leukocyte and Vascular Endothelial Cell Interaction
The sequence of events in the extravasation of neutrophils from the vascular lumen to the extravascular space is divided into (1) margination and rolling, (2) adhesion and transmigration, and (3) migration in interstitial mucosal tissues towards chemotactic stimulants (fig. 1), which are regulated by the interaction of adhesion molecules located on the surface of neutrophils and endothelial cells [1–3]. With stimuli, such as various cytokines and inflammatory mediators, neutrophils roll slowly on endothelial cells through interactions between L-selectin and carbohydrate antigen on neutrophils, and P-and E-selectin on endothelial cells. P-selectin is present in the Weibel-Plade bodies of endothelial cells and in the α-granules of platelets. Stimulation with histamine, thrombin, or reactive oxygen species (ROS) leads to rapid degranulation and translocation of P-selectin to the cell surface within a few minutes [4]. Eventually, the neutrophils adhere strongly to endothelial cells via CD11/CD18 glycoproteins and endothelial adhesion molecules of immunoglobulin superfamily, including the intercellular adhesion molecule 1 (ICAM-1) and the vascular cell adhesion molecule 1 (VCAM-1). ICAM-1 is expressed by activated and nonactivated endothelial cells and by lymphocytes, monocytes, and epithelial cells including gastric/intestinal epithelial cells. Its expression is stimulated by molecules such as interleukin (IL)-1, IL-17, tumor necrosis factor-α (TNF-α), interferon-γ (IFN-γ), and lipopolysaccharide (LPS). ICAM-1 binds β2-integrins lymphocyte function-associated antigen-1 (LFA-1; CD11a/CD18) and Mac-1 (CD11b/CD18), which are stored in the intracellular granules of leukocytes. VCAM-1 is found in a membrane-bound form on various cells, including endothelial cells and monocytes/ macrophages and also exists in a soluble form. The expression of VCAM-1 is stimulated by TNF-α, IL-1β, IL-4, IFN-γ, and LPS. After firmly binding to the endothelial surface (primarily CD11/CD18 binding to ICAM-1), the leukocytes transmigrate between cells along the intercellular junction. Platelet endothelial cell adhesion molecule 1, a cell-cell adhesion molecule, is a likely candidate for mediating this process. After passing the endothelial junctions, leukocytes are able to cross the basement membrane by focally degrading it with secreted collagenases. Due to the crucial role played by these adhesion molecules in the pathogenesis of gastrointestinal inflammatory diseases, targeting of these molecules has recently been proposed as a new direction for the development of anti-inflammatory strategies for these diseases. However, systemic treatment with alicaforsen (ISIS 2302), an antisense to ICAM-1 for Crohn's disease has not revealed significant effect, which was demonstrated by a randomized, double-masked, placebo-controlled study [5].
Cytokines and Mediators
After extravasation, neutrophils emigrate towards the site of gastrointestinal mucosal injury along a chemical gradient of chemotaxis. In the gastrointestinal inflammation, exogenous and endogenous substances are able to act as chemotactic agents for neutrophils including (1) cytokines especially IL-8, (2) soluble bacterial products, particularly peptides with N-formyl-methionine termini, (3) components of complement system, and (4) products of the lipoxygenase pathway of arachidonic acid metabolism, particularly leukotriene B4 (LTB4). IL-8, a member of the CXC chemokine family, is an important activator and chemoattractant for neutrophils, and has been implicated in the pathogenesis of a variety of inflammatory disease, including reflux esophagitis [6, 7], H. pylori-induced gastritis [8–11], NSAID-induced gastrointestinal injuries [12, 13], ischemic intestinal injury [14–16], and inflammatory bowel disease [17–19]. Especially, it has been indicated that a strong correlation exists between mucosal IL-8 contents and the level of polymorphonuclear cell infiltration in H. pylori-associated antral inflammation [20].
Current data suggest that Toll-like receptors (TLRs), which recognize specific pathogen-associated molecular patterns, are differentially expressed on both leukocytes and mucosal epithelial cells while serving to modulate leukocyte-epithelial interactions. Exposure of epithelial TLRs to microbial ligands has been shown to result in transcriptional upregulation of inflammatory mediators, whereas ligation of leukocyte TLRs modulates specific antimicrobial responses. Recently, Watanabe et al. [21] have reported that TLR4-deficient and MyD88-deficient mice are resistant to the intestinal damage induced by indomethacin. It has also been reported that treatment with CRX-526, a synthetic TLR4 antagonist, inhibits the development of moderate-to-severe disease in two mouse models of colonic inflammation: the dextran sodium sulfate model and multidrug resistance gene 1α-deficient mice [22]. These data indicate the significant role of TLR4-dependent pathway in the pathogenesis of the intestinal inflammation. A better understanding of these events will hopefully provide new insights into the mechanisms of epithelial responses to microorganisms and ideas for therapies aimed at inhibiting the deleterious consequences of mucosal inflammation in gastrointestinal disease.
As one of effectors of mucosal inflammation, a new lineage of effector CD4+ T cells characterized by production of IL-17, the T-helper (Th)-17 lineage, was recently described [23]. It has been shown that IL-17-expresing T cells are increased in the inflamed mucosa of inflammatory bowel disease patients, and that serum IL-17 concentrations are elevated in patients with active inflammatory bowel disease [24]. Recent studies have demonstrated that IL-23 and transforming growth factor-β can induce the differentiation of naïve T cells into Th-17 cells, and that this differentiation is most efficiently induced when Th1 and Th2 effector functions are simultaneously inhibited [25–27]. Interestingly, IL-17 appears to stimulate predominantly the production of cytokines that either specifically attract neutrophils to the site of inflammation (IL-8, GROα, GCP-2) or stimulate granulopoiesis in bone marrow (IL-6, G-CSF, GM-CSF) [28]. While IL-17 itself does not affect neutrophil chemotaxis, the supernatants from IL-17-treated fibroblast, epithelial and endothelial cells stimulate neutrophil migration. IL-17 appears to be an important mediator of inflammation, especially in neutrophil-dominated response to bacterial challenge, and may enhance faster and more effective recruitment of neutrophil as an important element of host defense.
Role of Platelets
Recently, a great deal of attention has been given to the role of platelets in gastrointestinal pathophysiology [29]. Patients with inflammatory bowel disease exhibit an increased expression of CD40L on platelets and increased plasma levels of soluble CD40L, which is largely derived from the activate CD40L+ platelets [30–32]. The most prominent activities mediated by the platelet CD40/CD40 ligand pathway include inf...