
eBook - ePub
Surgical Retina
ESASO modules 2009 and 2010: Selected contributions
- 188 pages
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Surgical Retina
ESASO modules 2009 and 2010: Selected contributions
About this book
In the new book series 'ESASO Course Series', the essentials of the courses of the European School for Advanced Studies in Ophthalmology (ESASO) are made available to interested ophthalmologists, optometrists, technicians and residents all over the world. In this volume, the seminars on surgical retina presented by renowned experts during ESASO's activities are collected. Many specialists have contributed their knowledge to make this volume a device to give practical support. The topics range from prevention to state-of-the-art diagnostic techniques and the latest surgical treatment options for many eye conditions such as diabetic retinopathy and rhegmatogenous retinal detachment. This publication provides the ophthalmologist with the main aspects of surgical retina in a simple and practical update.
Tools to learn more effectively

Saving Books

Keyword Search

Annotating Text

Listen to it instead
Information
Bandello F, Battaglia Parodi M (eds): Surgical Retina.
ESASO Course Series. Basel, Karger, 2012, vol 2, pp 1–34
ESASO Course Series. Basel, Karger, 2012, vol 2, pp 1–34
______________________
Diabetic Retinopathy Management
Marco A. Zarbina · William E. Smiddyb
aInstitute of Ophthalmology and Visual Science-New Jersey Medical School, Newark, N.J., and bBascom Palmer Eye Institute, Miami, Fla., USA
______________________
Abstract
Strict control of blood glucose and blood pressure is critical for reduction of the incidence and progression of diabetic retinopathy (DR). Follow-up of patients with diabetes mellitus is protocol based and not based solely on the presence of symptoms. Staging of the level of DR (mild, moderate, or severe nonproliferative DR vs. proliferative DR, PDR) drives the follow-up interval. The most common cause of visual loss in diabetic patients is diabetic macular edema (DME). The results of multicenter, randomized studies suggest that the best visual results for DME currently are achieved with intravitreal ranibi-zumab injections ± focal laser photocoagulation. Results using bevacizumab seem quite comparable to those with ranibizumab. In addition to treating DME, this approach also seems to reduce the likelihood of progression of DR. Selected patients also may benefit from intravitreal steroid treatment + focal laser therapy, but there is a relatively higher rate of glaucoma and cataract formation. Panretinal photocoagulation is currently the most effective treatment for high-risk PDR. Panretinal photocoagulation also should be considered for patients with severe nonproliferative DR and early PDR, particularly if follow-up cannot be assured and/or if the patient has type 2 diabetes mellitus. Pars plana vitrectomy is used to manage severe complications of DR such as nonclearing vitreous hemorrhage, severe fibrovascular proliferation, and retinal detachment. Adjunctive anti-vascular endothelial growth factor agents might enhance those results in selected subsets of patients.
Copyright © 2012 S. Karger AG, Basel
A substantial body of scientific data, much of it in the form of randomized, controlled clinical trials, underlies current treatment recommendations for patients with diabetic retinopathy (DR) [1–36]. These data define recommendations for patient follow-up, indications for focal and scatter laser photocoagulation, indications for pharmacological intervention (intraocular), and indications for vitrectomy. The main purpose of this chapter is to review these data in the context of contemporary treatment options. In addition, the pathophysiology of DR and associated clinical findings will be mentioned in the context of clinical management paradigms.
Pathological Processes
A detailed review of the pathophysiology of DR is beyond the scope of this chapter but has been published elsewhere (fig. 1, 2) [34, 37, 38]. Briefly, hyperglycemia leads inter alia to high intracellular levels of glucose, free radical formation (oxidative stress), and protein kinase C activation [39]. Chronic hyperglycemia generates advanced glycation end products (AGEs), which may incite processes leading to DR and maculopathy. Hypoxia, altered blood flow, retinal ischemia, and inflammation also are associated with DR. Increased vascular endothelial growth factor (VEGF) levels, decreased pigment epithelium-derived factor levels, increased protein kinase C production, endothelial dysfunction, and leukocyte adhesion are associated with breakdown of the blood-retinal barrier and, thus, increased diabetic retinal vascular permeability. In preclinical models, AGEs seem to be involved in of all these processes [40–42]. There is evidence that neuronal dysfunction precedes the retinal vascular abnormalities in DR [43]. For example, abnormalities of the ERG and multifocal ERG as well as apoptosis of retinal neurons have been demonstrated early in patients with diabetes mellitus, before the appearance of retinopathy [44–46]. Accordingly, early DR may be a neurovascular disease of the retina [47]. Eventually, ischemia may result in retinal neovascularization (mediated in part by the 121 and 165 amino acid isoforms of VEGF), vitreous hemorrhage (VH), retinal detachment, rubeosis iridis, and neovascular glaucoma.
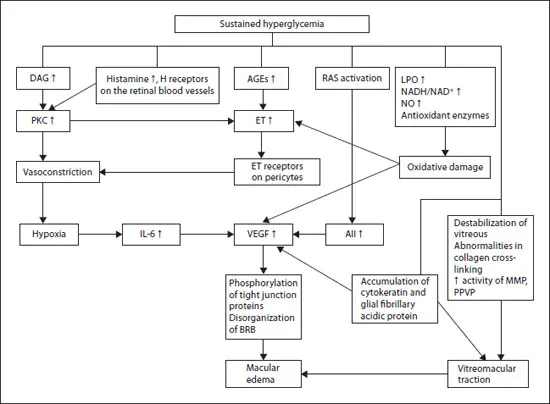
Fig. 1. Pathogenesis of DME. AII = Angiotensin II; DAG = diacylglycerol; ET = endothelin; LPO= lipo-oxygenase; NO = nitric oxide; PKC = protein kinase C; RAS = renin-angiotensin system. Reproduced with permission from Bhagat et al. [34].
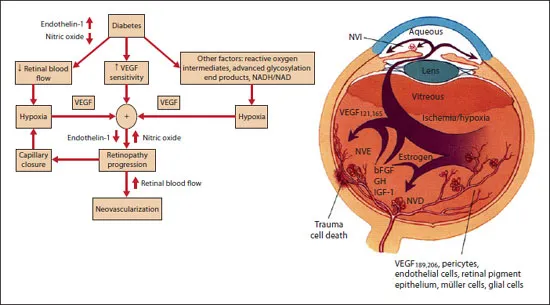
Fig. 2. Pathogenesis of retinal neovascularization in DR. VEGF and its isoforms, basic fibroblast growth factor (bFGF), growth hormone (GH), insulin-like growth factor-1 (IGF-1), inflammation, and ischemia all play important roles in disease progression. Reproduced with permission [114–116],
Clinical Findings
Microaneurysms (MAs) are foci of retinal capillary endothelial proliferation and are the earliest clinical sign of DR (fig. 3). Vascular damage resulting in ischemia and increased vascular permeability include cotton wool spots (signs of nerve fiber layer ischemia), intraretinal microvascular abnormalities (IRMA, areas of retinal vascular remodeling that occur within the plane of the retina), venous loops (areas of endothelial cell proliferation associated with partition of the venous lumen) [48], and venous beading, as well as in the development of capillary non-perfusion (fig. 3). Macular capillary non-perfusion can be associated with visual loss (particularly if the foveal avascular zone (FAZ) is enlarged to more than 875 μm in diameter) [49]. More severe degrees of ischemia induce proliferation of abnormal blood vessels arising from the retinal vasculature or, in severe cases, on the iris. These vessels may bleed, giving rise to preretinal or vitreous hemorrhage or hyphema, depending on their location (fig. 4). With time, fibrous tissue proliferation (cicatrization) results from the abnormal blood vessels (fig. 4). These cells have myofibroblastic properties and can contract to the point of causing traction and combined traction-rhegmatogenous retinal detachment (compounding the ischemia), causing severe visual loss. Cellular proliferation within the premacular vitreous cortex can cause opacification of the posterior hyaloid face (recognizable clinically and with optical coherence tomography, OCT) and macular edema. Growth of abnormal vessels in the anterior chamber angle is associated with neovascular glaucoma.
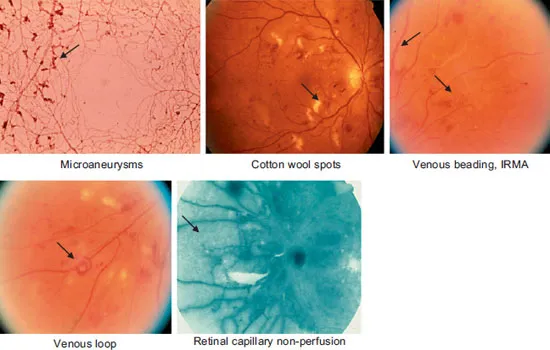
Fig. 3. Signs of DR. Arrows point out MAs (shown here in an India ink histopathology preparation), cotton wool spots (areas of superficial retinal whitening), venous beading, IRMA, venous loops, and retinal non-perfusion (shown on fluorescein angiography).
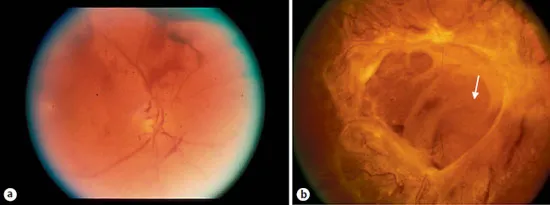
Fig. 4. a VH arising from retinal neovascularization. b Untreated retinal neovascularization has undergone cicatrization and contracture resulting in traction retinal detachment.
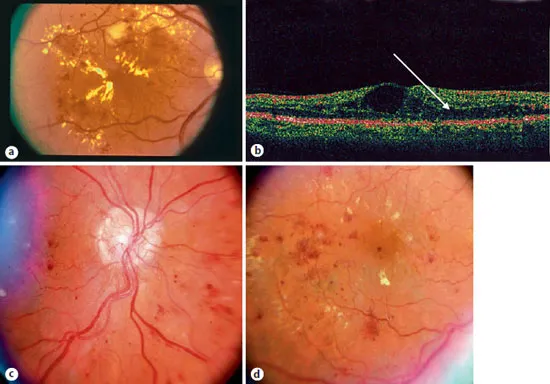
Fig. 5. a Hard exudates in a case of NPDR. b OCT of a case with macular edema showing fluid accumulation in the outer plexiform layer (arrow). c Neovascularization at the optic nerve head. d Same eye as c showing macular edema with loss of the foveal light reflex and cystic change in fovea.
Increased vascular permeability is evidenced by lipoprotein exudates; hemorrhages and edema develop in the early stages of DR and may occur to remarkable degrees in later stages, especially when systemic hypertension coexists (fig. 5). Breakdown of the inner blood-retinal barrier results in retinal edema (most prominent in the outer plexiform layer, nerve fiber layer of Henle) as well as intraretinal hemorrhage. Severe lipoprotein deposits (hard exudates) can be a manifestation of hyperlipidemia as well as vascular incompetence [50]. Hard exudates normally are cleared by phagocytosis, and edema is corrected by dehydration of the retina via RPE active transport and, in some cases (e.g. after ph...
Table of contents
- Cover Page
- Front Matter
- Diabetic Retinopathy Management
- Nonsurgical Options for the Treatment of Diabetic Macular Edema
- Use of Anti-Vascular Endothelial Growth Factor Drugs for Diabetic Macular Edema
- Proliferative Diabetic Retinopathy: Laser Treatment and Intravitreal Medication
- Proliferative Diabetic Retinopathy: Surgical Treatment
- Complications and Management of Diabetic Vitrectomy
- Diabetic Macular Edema: Cases and Diagnostics
- Retinal Venous Occlusions: Diagnosis and Choice of Treatment
- Rhegmatogenous Retinal Detachment: Clinical Preoperative and Postoperative Handling
- Making Sense of Prophylaxis for Retinal Detachment
- Ab externo Surgery of Rhegmatogenous Retinal Detachment: Handling of Intraoperative and Postoperative Complications
- Vitrectomy for Rhegmatogenous Retinal Detachment
- Scleral Buckling Materials
- Pneumatic Retinopexy
- Retinal Detachment by Giant Tear or Dialysis
- Macular Hole Surgery
- Vitrectomy for Epiretinal Membrane
- Irvine-Gass Syndrome
- Endophthalmitis
- Subject Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Surgical Retina by F. Bandello,M. Battaglia Parodi,F., Bandello,M., Battaglia Parodi, Francesco Bandello,Borja Corcóstegui,Francesco, Bandello,Borja, Corcóstegui in PDF and/or ePUB format, as well as other popular books in Medicine & Endocrinology & Metabolism. We have over one million books available in our catalogue for you to explore.