Covering newsworthy aspects of contemporary biology—gene therapy, the Human Genome Project, DNA testing, and genetic engineering—as well as fundamental concepts, this book, written specifically for nonbiologists, discusses classical and molecular genetics, quantitative and population genetics—including cloning and genetic diseases—and the many applications of genetics to the world around us, from genetically modified foods to genetic testing.
With minimal technical terminology and jargon, Genes and DNA facilitates conceptual understanding. Eschewing the organization of traditional genetics texts, the authors have provided an organic progression of information: topics are introduced as needed, within a broader framework that makes them meaningful for nonbiologists. The book encourages the reader to think independently, always stressing scientific background and current facts.

- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Topic
BiowissenschaftenSubtopic
BiologieCHAPTER 1.
What Are Genes?
YOU MAY ALREADY KNOW THAT GENES are made of DNA (short for deoxyribonucleic acid). More interesting than knowing this is understanding how we know that DNA is the basis for heredity and understanding the importance of the structure of DNA for inheritance. You will see in this chapter that DNA and its structure are the keys to understanding inheritance.
DNA
DNA has a fascinating history. The Swiss scientist Friedrich Miescher discovered DNA near the end of the nineteenth century. Miescher never knew that the substance he had isolated from sperm and pus (yes, pus!) would turn out to be so critical to the understanding of life. He died several decades before the function of DNA and its famous double-helical structure were uncovered. After Miescher, other scientists tried to identify the chemical composition of sperm, reasoning that sperm must carry the genetic material to the next generation. These scientists also reasoned that sperm cells have very little excess cellular material other than the hereditary material found in the sperm head. In fact, DNA constitutes over 60 percent of the sperm head; the remainder is mostly protein.
For a long time after Miescher’s discovery, DNA was thought to be a simple molecule, consisting of nucleotides strung together like beads on a string. Each nucleotide is composed of a sugar (deoxyribose) chemically linked to phosphorus atoms and one of four different nitrogenous bases (so called because they contain a significant number of nitrogen atoms). The nitrogenous bases are adenine, guanine, cytosine, and thymine. These four bases are abbreviated as A, G, C, and T. Nothing known about the DNA molecule suggested that it could play any role in heredity. The structure of DNA seemed much too simple to account for the many already known hereditary traits. But then scientists found that the building blocks of DNA—the nucleotides—were repeated hundreds of times in the DNA molecule. As techniques to isolate DNA from living cells improved, the number of nucleotides in a DNA molecule was found to be in the thousands, and then in the hundreds of thousands. Scientists had discovered that DNA is a polymer, much like many plastics such as polyethylene and polypropylene, except that DNA is a very long polymer with millions of nucleotides, As, Gs, Cs, and Ts.
Yet nobody knew what living cells did with this polymer, nor did anybody know the structure of the DNA molecule. In fact, some scientists believed that only animals and bacterial cells possessed DNA and that plants were devoid of it. Since plants, as well as animals and bacteria, all had well-defined genetic characteristics (for example, flower color for plants, shape for animals, and pathogenicity for bacteria), DNA could not be the genetic material, so the logic went. We now know that plants do contain DNA, and that the failure to isolate it from them was due to the use of crude techniques. In fact, for geneticists, plants, animals, and bacteria are largely similar in spite of their great diversity. This is because their hereditary properties are all based on the existence of one substance: DNA.
DNA Can Be Specifically Stained and Observed in Cells
An important step in the development of ideas about the chemical nature of the genetic material was the ability to stain DNA. In the 1920s, German biochemist Robert Feulgen developed a way to specifically stain DNA. He then used this method to stain DNA in living tissue. The Feulgen reaction, as it is now called, specifically colors DNA purple. The stained cells can then be viewed under the microscope. Feulgen used this technique on all kinds of tissues from animals, plants, and protozoa. Under the microscope, the purple DNA stain was found in a central compartment of all these cells. The compartment is given the name nucleus, plural nuclei. Feulgen found that the nuclei of all of these cells, including the nuclei of plant cells, became stained. This definitively proved that plants had DNA and that the DNA of cells is located in the nucleus.
With the Feulgen stain, scientists had a tool to measure the amount of DNA present in cells. In 1950, in a paper entitled “Constancy of DNA in Plant Nuclei,” Hewson Swift at the University of Chicago showed that all cells from different parts of a corn plant had a constant amount of DNA. Furthermore, the amount of DNA in pollen was half that found in, for example, the leaf and root cells. He found that rapidly dividing cells in the root tip and other cells prior to cell division had twice as much as DNA. These are what one would expect of the genetic material (see chapter 2). If DNA was the genetic material, its amount should be constant in all the cells of the organism regardless of the size of the cell.
An even more interesting observation was made using the Feulgen stain: DNA changes shape as cells divide. Most of the time, the Feulgen stain showed an amorphous purple sphere in the nucleus, without any substructure. But just before cells divide, the DNA becomes condensed into sausage-looking structures called chromosomes. It was found that the number and shape of these condensed chromosomes was the same in different body cells of the same organism. Furthermore, one could see that in sperm cells the number of chromosomes was halved. We would expect that the amount of hereditary material in the gametes, sperm or pollen and egg or ova, would be half that found in the nonreproductive cells of the organism.
DNA Determines Genetic Properties in Bacteria
That DNA is indeed the genetic material was demonstrated in bacteria in 1944. A team led by the Canadian Oswald Avery at Rockefeller University in New York made this landmark discovery. Their biological material was the bacterium Streptococcus pneumoniae, which, as its name indicates, causes pneumonia. Avery’s laboratory possessed two strains of these bacteria. One strain infects mice with pneumonia (the “virulent” strain), and the other strain does not (the “avirulent” strain). The two strains look different when growing in a petri dish: The virulent strain grows as a smooth, slimy, large collection of cells known as a colony. The avirulent strain produced small, well-defined, rough-looking colonies (figure 1.1). Thus the bacterial strains are distinguishable by two characteristics: physical appearance and the strain’s ability or inability to cause pneumonia. These characteristics are hereditary. Avery knew this because he grew the two bacterial strains for a long time, during which the cells divided many times, but this produced no change in the bacteria’s ability to cause pneumonia or the bacteria’s physical appearance.

Figure 1.1 Virulent and Avirulent Streptococcus Pneumoniae Colonies on an Agar Plate. The large, slimy, smooth colonies on the left are virulent, and the small, distinct colonies on the right are avirulent. Plate and photograph courtesy of Kirstin Malm.
Avery did an experiment that suggested that hereditary properties like virulence and appearance could be exchanged between cells. He knew, as we do, that heating bacterial cells kills them. Indeed, when the smooth, virulent bacteria are heated, they are killed and no longer infect mice with pneumonia. But when one mixes these heat-killed virulent bacteria with live, avirulent rough bacteria, one finds that this mixture can kill mice. Avery reasoned that the genetic material from the heat-killed virulent bacteria changed the properties of the rough strain. In this case, he reasoned, DNA purified from the smooth strain would change the characteristics of the rough strain, but only if the DNA could get into the rough, avirulent bacteria. Avery’s research team purified DNA from the smooth, slimy strain and added the purified DNA to the rough cells. They observed that a few smooth and slimy cells appeared in the culture of rough cells exposed to the DNA! This “transformation,” as they called it, was stable over many cell divisions. Avery and his team interpreted these observations to mean that DNA was the genetic material.
However, to make sure that the DNA did not contain contaminants that could have been the true genetic material, they did further experiments with a variety of enzymes. Suffice it to say that enzymes are proteins that catalyze all sorts of reactions. Biologists commonly give names to these enzymes by putting the suffix “-ase” after the name of the substances they act upon. For example, deoxyribonuclease is an enzyme that destroys DNA by cutting it into its nucleotide building blocks. Similarly, a protease is an enzyme that destroys proteins. When Avery and his coworkers added deoxyribonuclease to their purified DNA and then added this mixture to rough cells, no smooth, slimy cells were recovered. This meant that destroying DNA destroyed the transforming activity. On the other hand, adding proteases to the DNA had no effect on its transforming activity. If contaminating proteins in their DNA samples had been responsible for the transforming activity, the addition of proteases should have destroyed this activity. It did not. This means that proteins were not responsible for the observed transformation. There it was: the genetic material—the genes—of Streptococcus pneumoniae was not made of proteins, but of DNA.
Avery’s work had been done under carefully controlled conditions, and his conclusions were straightforward. Yet nobody at the time believed him! Why was that so? It turns out that as recently as the late 1940s, scientists were convinced that protein, not DNA, was the genetic material. Why? Proteins seemed like a much more likely candidate to be the genetic material. Proteins existed in innumerable varieties that differed enormously among living species. For this reason, proteins were thought to constitute the true genetic material. Another factor is that in 1944 World War II was still raging, and Avery’s discovery must have been seen as of little consequence when people were dying on the battlefields and in bombed-out cities. Finally, some people thought that DNA was possibly the genetic material of some rare bacterial species, but certainly it could not be responsible for the hereditary properties of higher life forms, such as animals and plants. Of course, the skeptics were dead wrong, as we know. Such an important discovery should have earned its authors a Nobel Prize. However, Avery was sixty-seven at the time he made this important discovery, and he died eleven years later. Recognition of important discoveries often takes decades. Since the Nobel Prize is not granted posthumously, Avery was unable to be recognized for his important work.
We know today that DNA is an almost universal genetic material, and that genes present in simple viruses, bacteria, plants, and animals are all made of DNA. Amazingly, some viruses are made of a chemical very similar to DNA, ribonucleic acid (RNA), where the base thymine (T) is replaced by uracil (U) and where the sugar is ribose, not deoxyribose.
DNA Is a Double Helix
By the late 1940s biochemists knew that DNA was a very long polymer made up of millions of nucleotides. Each nucleotide contains one of the four nitrogenous bases (A, T, G, or C) linked to a deoxyribose unit, in turn linked to a chemical group containing a phosphorus atom. DNA is held together by bonds between the phosphate and the deoxyribose units. Therefore, one speaks of the DNA’s “sugar-phosphate backbone” (figure 1.2.A). In those years, it was also known that in all DNA samples isolated from widely different species (human, yeast, and bacteria, for example), the amount of adenine (A) was always equal to the amount of thymine (T). Similarly, guanine (G) was always equal to cytidine (C). Nobody knew how to explain this, but the observation suggested some regularity in the DNA molecule.
A breakthrough occurred when Rosalind Franklin, a researcher at King’s College in London, England, succeeded in crystallizing DNA in the early 1950s. Crystals are formed when identical molecules are packed in a very organized fashion. This is rather simple to do for a small molecule. Perhaps you have made sugar crystals by putting a string into a solution saturated with sugar. This happens because the rough structure of the string initiates crystal formation. Once some sugar molecules attach to the string, other sugar molecules can fit in like bricks in a wall. Because DNA is such a large molecule, it does not form crystals readily. Why was it so important to obtain DNA crystals? One can take advantage of the very regular arrangement of the same molecules in a crystal to determine their structure. There existed at the time a well-established technique used to determine the arrangement of atoms inside a crystal. This technique is called X-ray crystallography, and, as its name indicates, it consists of illuminating a target crystal with X rays. The regular arrangement of atoms in a crystal deflects X rays and forms spots in concentric rings on a photographic film. The more organized the structure, the more spots are formed farther out in the ring. By noting the location and intensity of these spots, one can then determine the relative positions of the atoms in the crystal and determine the three-dimensional structure of the crystallized molecules. Thus, Rosalind Franklin obtained the first high-quality X-ray data for a DNA crystal (figure 1.3).
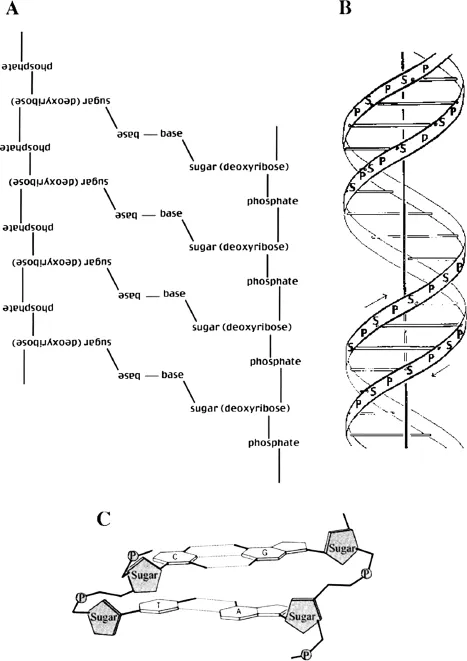
Figure 1.2 Diagrams of DNA. A. A flat diagram shows two strands of DNA each with four bases. Note that each strand is held together by a “sugar-phosphate backbone.” The two strands run in opposite directions, thus the left-hand strand is shown upside down. The strands are held together by weak bonds, called hydrogen bonds, between the bases. B. A diagram of the double-helix structure of DNA. Note the sugar-phosphate backbone in opposite directions shown by the arrows. The rungs of the ladder represent the bases held together by the weak hydrogen bonds. C. A detailed diagram of two bases from two opposing strands of DNA. The phosphates are shown as shaded circles and sugars as shaded pentagons. The dotted lines connecting the bases are weak hydrogen bonds.
At this time, James Watson, a young American postdoctoral scientist, and Francis Crick, an English physicist working on his Ph.D. dissertation, were both at Cambridge University. These two struck up a collaboration to solve the problem of the structure of DNA. Neither of them had done any previous work with DNA. They were thus novices, although Crick knew the theory of X-ray crystallography very well. Indeed, Watson and Crick never did a single experiment to solve the structure of DNA. All the experimental results had been obtained by Rosalind Franklin and later repeated by her boss, Maurice Wilkins. One day, while he was visiting the King’s College researchers, Watson saw a photograph of DNA made by Franklin. The arrangement of the spots radiating out in an X shape immediately suggested to him that DNA must be a helical molecule. Back in Cambridge, Watson convinced Crick of that interpretation, and model building started. After a few days of trial and error, they had a helical molecule that was also consistent with Franklin’s X-ray crystallography data. DNA was a double helix in which the sugar-phosphate backbones are on the outside, while the bases are on the inside of the molecule (figure 1.2.B). This structure was held together by weak bo...
Table of contents
- Cover
- Half title
- Title
- Copyright
- Dedication
- Contents
- Acknowledgments
- List of Contributors
- Preface: Why Is Genetics Important?
- Chapter 1. What Are Genes?
- Chapter 2. Inheritance of Single-Gene Traits
- Chapter 3. Mendelian Traits in Humans
- Chapter 4. From Genes to Phenotype
- Chapter 5. Using Bacteria as Protein Factories
- Chapter 6. Genetically Modified Plants
- Chapter 7. When Things Go Wrong
- Chapter 8. Mutagens, Teratogens, and Human Reproduction
- Chapter 9. Linkage and Mapping: Gene Discovery
- Chapter 10. Genetics of Populations and Genetic Testing
- Chapter 11. Survival of the Fittest?
- Chapter 12. Nature Versus Nurture
- Chapter 13. Genetically Modified Animals and the Applications of Gene Technology for Humans
- Appendix A. Internet Resources
- Appendix B. Glossary of Scientific Names of Organisms
- Appendix C. Glossary of Human Genetic Diseases
- Appendix D. Glossary of Terms
- Index
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Genes and DNA by Charlotte K. Omoto in PDF and/or ePUB format, as well as other popular books in Biowissenschaften & Biologie. We have over 1.5 million books available in our catalogue for you to explore.