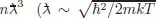![]()
Chapter 1
Phase States of Matter, Their Classification
As a substance advances along the scale of pressures and temperatures, its composition, structure, properties, etc. undergo radical changes from the ideal state of noninteracting neutral particles described by the classical statistical Boltzmann function to the exotic forms of baryon and quark–gluon matter some of whose features now began to show up in recordparameter experiments on relativistic ion collisions [294, 295] and manifest themselves in the interpretation of astronomical observations of so-called “compact” (“neutron” and “strange”) stars [414].
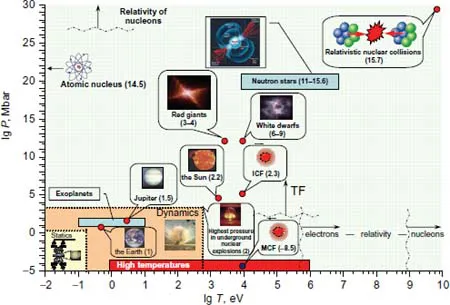
The pressures and temperatures realized in nature are presented schematically in Fig. 1.1.
In the second section of this chapter we shall touch upon ultrahigh compressions when the nuclear properties of matter come out to the foreground.
1.1 Electron component
To begin, we shall consider the states of matter under “moderate” compressions when its properties are determined by electrons resident in atomic and ionic shells and by free electrons. We shall discuss compressed atomic and molecular systems and electron-ion “electromagnetic” plasma [287, 294, 295]. All these interesting and essentially nonlinear phenomena appear in both astrophysical and laboratory plasma, and in spite of giant differences in spatial scales, they have much in common and are a subject of “laboratory astrophysics” [221, 871].
In
Chapter 2 we consider the range of low (
T J, J is ionization potential) temperatures and low (
ρ ρc is density at the critical point)
densities, where the interparticle interaction is small and can be taken into account within perturbation theory. With increasing density the interaction strengthening in the system requires the use of purely empirical descriptions and in supercritical conditions — the models of liquids (
Sec. 2.3) based on either integral equations or the parameter-free methods of molecular dynamics or Monte Carlo method.
Fig. 1.1 Extreme states [545] in nature and laboratory. The numbers in brackets indicate the density logarithm in g/cm3. The region “statics” corresponds to the static and “dynamics” to the dynamic methods of obtaining high pressures, the “high temperatures” domain corresponds to experiments at high temperatures.
The liquid state region (Sec. 2.3) has traditionally been thought of as the most difficult and poorly investigated since the strong interaction and disorder make theoretical predictions of the properties of real liquids extremely ambiguous [656]. It however appeared possible to formulate here realistic zero-approximation models based on the use of exceedingly simplified hard- and soft-sphere potentials permitting the use of the numerical methods of molecular dynamics and the Monte Carlo method. Thus, the main qualitative features of experimentally observed structural factors were reproduced and the nature of melting and crystallization was analyzed (Sec. 7.3). The equations of state (EOS) of real liquids are found from machine calculations applying the variational method of perturbation theory, the parameters of interparticle pair potential being found from comparison of the calculations with experiment.
The absence of reliable experimental data at high pressures and temperatures very largely obstructs the development of realistic models of liquids valid up to the boiling point converting liquid into vapor or (metals) directly into the plasma phase.
As the temperature rises, dissociation and ionization of first external (T ~ Z4/3Ry) and then internal (T ~ Z2Ry) electron shells of atoms proceed successively, and we find ourselves in the plasma region (Sec. 3.3) which is the most widespread state of matter in nature [287, 294, 295]. After all, plasma state is inherent in about 95 to 98% of the baryon matter in nature with the exception of dark matter and dark energy.
The thermodynamic properties of plasma are described by the ionization equilibrium equations involving the experimental and computational data on the spectra of atoms and ions and the computational values of ionization potentials. The applicability limits of such an approach for low densities are determined by violation of the condition of local thermodynamic equilibrium [287].
In the region of higher compressions, the strong interparticle interaction affects not only free electrons but also those bound in atoms and ions thus causing deformation of the discrete energy spectrum of plasma; along with thermal, pressure-induced or “cold” ionization of matter becomes dominant. The condition of this effect is the closeness of atomic and ionic sizes to the average interparticle distance, a0 ~ n1/3. This phenomenon is described by the “restricted” atom models that take into consideration the final actual ion size and the influence of plasma surrounding upon its discrete spectrum [287].
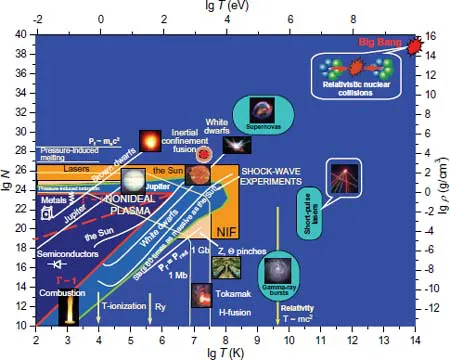
The phase diagram of a substance corresponding to high energy densities is presented in Fig. 1.2 [287, 293, 319, 727] where the conditions in astrophysical objects, in technical and laboratory experimental setups are given. One can see that being the most wide-spread state of matter, nature plasma occupies almost the whole region of phase diagram.
The greatest difficulty in the physical description of such a medium lies in the region of nonideal plasma where the interparticle Coulomb interaction energy e2n1/3 is comparable with or exceeds the kinetic energy Ek of particle motion. In this region, Γ = e2n1/3/Ek > 1, the plasma nonideality effects cannot be described by perturbation theory [287, 545], and application of machine parameter-free Monte Carlo or molecular dynamics methods [287, 545] runs into the difficulties of the choice of adequate pseudopotentials and correct allowance for quantum effects.
Fig. 1.2 Phase diagram of the states of matter [293, 727].
The electron relativism effects in the equation of state and in the transport properties of plasma, when mec2 ~ kT, correspond to T ≈ 0.5 MeV ≈ 6 · 106K. Above this temperature the substance becomes unstable under spontaneous electron–positron pair production.
The quantum effects are determined by the degeneracy parameter
is the thermal de Brog...