![]()
Chapter 1
Semiconductor Quantum Dots for Sentinel Lymph Node Mapping through In Vivo Fluorescent Imaging
1.Introduction
Semiconductor QDs, also called semiconductor metalloid-crystal nanostructures, are small nanoparticles that are usually spherical in shape, possess bulk-like crystalline structures and contain around 200–10,000 atoms. The size of QDs ranges from 1 to 10 nm, which is close to or smaller than the dimensions of the exciton Bohr radius. The most commonly used QDs in biomedical research are cadmium (Cd)-based, especially cadmium selenide (CdSe) and cadmium telluride (CdTe). The band gap, which is the energy difference between the top of the valence band and the bottom of the conduction band, of Cd-based QDs is located between 1.7 and 3.5 eV [1]. As a result, they emit bright visible (blue, green, yellow, or red) and near-infrared (NIR) fluorescence from a low energy band. For example, the fluorescent emission spectrum of CdSe QDs can extend to ~700 nm, and that of CdTe QDs can extend to ~800 nm [2, 3]. CdSe and CdTe QDs are easy to synthesize in aqueous solution, and the products obtained are water-soluble. They typically display fluorescence with a narrow full width at half-maximum (FWHM), short lifetime and high quantum yield. These features have facilitated the use of CdSe and CdTe QDs as well as those coated with an inorganic shell of zinc sulfide (ZnS) or silica in both in vitro and in vivo fluorescent imaging for biomedical detection.
However, Cd is a probable carcinogen, and has a long biological half-life in the body [4]. Therefore, Cd-free QDs have also attracted considerable worldwide attention recently. These Cd-free QDs include III(A)–V(A) semiconductors like indium arsenide and indium phosphide, IV(A)–VI(A) semiconductors such as lead selenide (PbSe) and lead sulfide, II(B)–VI(A) semiconductors including mercury telluride and zinc selenide, I(B)–VI(A) semiconductors such as silver sulfide and silver selenide, and I(B)–III(A)–VI(A) semiconductors including copper indium sulfide (CuInS2) and copper indium selenide. The band gaps of most of these Cd-free QDs are narrower than those of the visible QDs mentioned above. For example, the band gaps of PbSe and lead telluride are less than 0.5 eV [5]. Therefore, such Cd-free QDs can be excited by visible and NIR photons and then emit NIR fluorescence from a low energy band [6]. However, these Cd-free QDs are usually synthesized in organic solution and their quantum yield obviously decreases when they are transferred into aqueous solution. In addition, most of these Cd-free QDs exhibit fluorescence with a broad FWHM, long lifetime and low quantum yield compared with that of Cd-based QDs [7].
The fluorescence of both Cd-containing and Cd-free QDs is superior to that of traditional organic dyes, which is the most attractive properties of QDs. Since QDs were first used for cell labeling and fluorescent imaging by Nie et al. [8] and Alivisatos et al. [9] in 1998, these novel fluorescent nanomaterials have attracted great attention in biomedical research for in vivo tissue-targeted imaging. SLN imaging using QDs may be the most easily achieved example of in vivo targeted imaging. This is because QDs passively migrate into lymph channels and then accumulate in SLNs after being injected subcutaneously into a body. In this chapter, the optical properties of QDs and their use in SLN mapping are described.
2.Optical Properties of QDs
Labeling and tracing of molecules or cells are very important techniques in biomedical research. Compared with radioactive tracers, fluorescent probes are less hazardous to animals and humans. Moreover, fluorescent imaging is simple and convenient. Semiconductor QDs can be used as ultra-sensitive fluorescent probes for in vitro and in vivo biomedical imaging because of their unique optical properties. Semiconductor QDs possess several advantages over traditional organic fluorescent dyes, fluorescent proteins, and lanthanide chelates as imaging probes, which are discussed as follows:
(1)QDs exhibit size-dependent fluorescence because of strong quantum confinement effects.
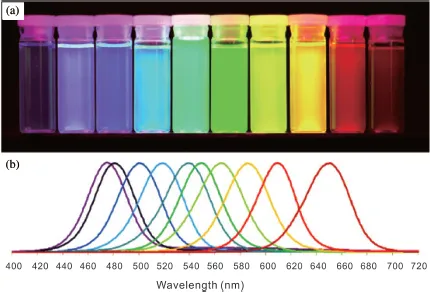
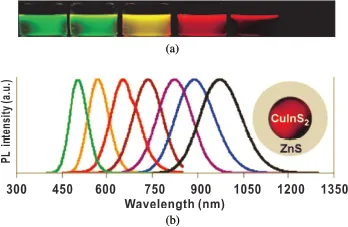
The optical properties of QDs can be tuned by changing their size because the band-gap energy of QDs is controlled by size. The larger the QDs, the lower their band-gap energy, which is termed the quantum confinement effect. As a result, large QDs can be excited by low-energy light, and then emit low-energy fluorescence; that is, at a long wavelength. Therefore, the color of fluorescent from QDs changes from blue to green, yellow, then red (including NIR) as their size increases. As shown in Fig. 1.1, as the diameter of CdSe/ZnS core–shell QDs increased slightly, their fluorescence obviously shifted to a longer wavelength. This was visibly observed as changes in the color of the QD solution [10]. QDs of the same materials that emit multiple colors are therefore easily obtained by tailoring their size, which is attractive for high-throughput biological detection. The color and intensity of QD fluorescence can also be varied by changing the composition or surface coating of the QDs. Figure 1.2 shows the distinct fluorescence of Cd-free CuInS2/ZnS core–shell QDs with different diameters [11]. In addition, CdSe/ZnS QDs of the same diameter as CuInS2/ZnS ones emit fluorescence of different colors.
Fig. 1.1. Size-dependent optical properties of Cd-containing CdSe/ZnS QDs. (a) Fluorescent image of 10 vials of monodisperse QDs of different size. (b) Fluorescence spectra of the same 10 QD solutions. The fluorescent emission maxima are located at 443, 473, 481, 500, 518, 543, 565, 587, 610, and 655 nm (from left to right), respectively. Reprinted with permission from Ref. [10]. Copyright (2009) Elsevier.
Fig. 1.2. Size-dependent optical properties of Cd-free CuInS2/ZnS QDs. (a) Fluorescent images of monodisperse QDs of different size. (b) Fluorescence spectra of the same QD solutions. Reprinted with permission from Ref. [11]. Inset is a diagram of the structure of a single QD. Copyright (2009) American Chemical Society.
(2)QDs have broad continuous excitation spectra and narrow symmetric fluorescent emission spectra.
QDs exhibit broad continuous excitation spectra extending from ultraviolet to the NIR region (at shorter wavelength than that of emission). Therefore, QD fluorescence can be generated by a wide range of excitation wavelengths, and different-colored QDs can be excited simultaneously by a single light source. Figure 1.3a shows the broad absorption spectra of four sizes of CdSe QDs (2.2, 2.9, 4.1 and 7.3 nm) [12]. The absorption band edge of the largest QDs (7.3 nm) extends to longer than 650 nm. This is contrast to organic fluorescent materials, e.g., methylene blue (Fig. 1.3d) [13], which exhibit a sharp absorption spectrum. These four sizes of CdSe QDs display narrow symmetric fluorescence spectra, and their emission peaks are separated from each other (Fig. 1.3b) [12]. In addition, these QD solutions simultaneously emit bright blue, green, orange-red and deep red fluorescence when they are irradiated by a single ultraviolet source (Fig. 1.3c) [12].
In another example, the polymer coated CdSe/ZnS QD microbeads with a diameter of 0.5 μm that exhibited green, yellow and red fluorescence were injected subcutaneously at three adjacent locations in a host mouse. Wavelength-resolved spectral imaging was then carried out using a spectral imaging system (CRI, Inc., Woburn, MA, USA) composed of an optical head containing a liquid crystal tunable filter with a bandwidth of 20 nm and scanning wavelength range of 400–720 nm. Excitation of the QD microbeads caused them to simultaneously emit bright green, yellow, and red fluorescence (Fig. 1.4) [14]. These emission properties of QDs also can be seen in Figs. 1.1 and 1.2 above. Multiple-colored QD beads suitable for gene, protein and small-molecule library encoding [10, 15] have been developed because of the narrow emission and broad absorption spectra o...