![]()
Chapter 1
Introduction
The statement that biocatalysis is of great importance sounds trivial in face of the fact that biocatalysis is the prerequisite for any life at all. Application of biocatalysis has a more than eight thousand year old tradition if the unwitting employment of the underlying processes in ancient times is included, e.g. fermentation in connection with beer brewing or the baking of bread. Nowadays research in biocatalysis increasingly influences all areas of daily life, including medicine, pharmacy, nutritional products, analytics, environmental technology, and others.
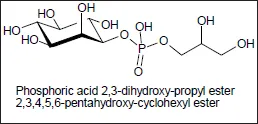
Millions of years of evolution have generated an unimaginable diversity of organisms. Biocatalysts regulate and control all metabolic reactions in microorganisms, plants and animals in an often very selective way and under conditions that are either mild or adapted to the special requirements of the milieu an organism develops its activities in. For instance, microorganisms have been found to live under unusual environmental conditions such as disused chemical plants by developing an enzyme equipment enabling them to grow on the chemicals they discover. Among the so-called extremophiles (Berger et al., 2014, and literature cited therein) barophiles populate deep sea habitats where they grow at temperatures just above 0°C, and at a pressure of more than 1000 bar but perish when temperature increases and the pressure decreases; psychrophiles metabolize at even lower temperatures, e.g. under the conditions of the Siberian permafrost. Of even more biotechnological relevance are thermophilic (moderate-, extreme-, and hyperthermophilic) microorganisms existing in so-called ecological niches, as found, e.g. in the hot springs of the Yellow Stone National Park about 60 years ago. They produce enzymes that function at temperatures up to 130°C and often under extreme pH-conditions (alkalophiles or acidophiles). Such robust enzymes are of great interest for various biotechnological processes. Interestingly, not all enzymes isolated from (hyper)thermophiles are particularly heat stable. These organisms developed alternative strategies to survive under such conditions as, e.g. the synthesis of low-molecular mass metabolites that exert in vivo a stabilizing effect resulting from strengthening intramolecular interactions within the protein molecule with a stabilizing effect on protein unfolding as shown by Roychoudhury et al. (2013) by means of atomic force microscopy. To these so-called compatible solutes belong the disaccharide trehalose, α- and β-glutamate (accumulated for osmoadaption), di-myo-inositol phosphate and 1-glyceryl-1-myo inositol phosphate (opposite figure), discovered in the hyperthermophilic bacterium Aquifex pyrophilus in response to both osmotic and heat stresses (Lamosa et al., 2006). These solutes are of interest for a variety of biotechnological applications. Altogether, there should exist innumerable different enzymes (most of them not detected so far) to catalyze nearly all types of chemical reactions known from Organic Chemistry lessons. The application potential of biocatalysts for chemical synthesis has been recently reviewed by Clouthier and Pelletier (2012), and is discussed here in a variety of chapters, particularly in connection with the topic Industrial Biocatalysis.
1.1 Advantages and Disadvantages of Biocatalysts
Nature has created excellent catalysts by evolution over millions of years. They are mostly proteins (enzymes or catalytic antibodies) but also nucleic acids with catalytic properties similar to those of enzymes detected in the early 80s. Up to now, of these naturally occurring catalysts only enzymes are used in applied biocatalysis.
Enzymes catalyze chemical reactions (and energetic transformations) in a single cell or in a whole organism, essential for survival and reproduction. A biocatalyst may either be the complete cell itself, employed in a viable, non-viable, growing or non-growing state, or an individual enzyme. As other catalyst, they increase the rate at which equilibrium is attained without affecting the equilibrium constant by providing an alternative reaction path with lower activation energy than the one of the corresponding un-catalyzed reaction (Chapter 5). What is spectacular is the degree of rate acceleration.
Table 1.1 The main advantages and disadvantages of biocatalysts with regard to their possible application in biotransformations on laboratory or industrial scale.
Advantages | Disadvantages |
Very efficient catalysis of most known chemical reactions High regio- and stereoselectivity Mild reaction conditions and thus low energy consumption Amount of byproducts is low Preparation on large scale is possible through fermentation (microbial enzymes) Reuse is possible (immobilization) They can be designed to a certain extent They are non-toxic if correctly applied | Protein molecules are rather instable in aqueous media Many enzymes are cofactor- dependent Allergic reactions are possible Enzymes may be inactivated by higher salt concentrations Inactivation may further occur
through inhibition by |
An impressive example is given by the enzyme catalase that catalyzes the decomposition of hydrogen peroxide. From the activation energies for the uncatalyzed and the catalyzed reaction, i.e. 75 kJ/mol and 8 kJ/mol, respectively, results a factor of rate enhancement of about 1015. This individual value lies at the upper limit; usually such factors for enzyme-catalyzed reactions are between 108 and 1012.
The enormous rate acceleration allows reactions to proceed under physiological conditions in a split second that would take ages to reach equilibrium without a catalyst. Enzyme-catalyzed reactions are often highly substrate-specific without forming byproducts, a consequence of their regiospecificity. Furthermore, due to the fact that enzymes are asymmetric molecules, they may precisely differentiate between stereoisomers, resulting in the formation of chiral products whereas chemical synthesis often leads to racemic mixtures. All these properties make enzymes interesting candidates as catalysts for industrial processes. However, enzymes also have disadvantages (Table 1.1) limiting their use.
1.2 Strategies to Improve the Performance of Biocatalysts
Not long ago enzymes had to be employed as provided by Nature. However, with the advent of new biological and molecular tools it became possible to influence the properties of biocatalysts with respect to catalytic activity, selectivity, and stability. High stability of a biocatalyst under process conditions is a prerequisite for its economic application in the industrial production of high-value fine chemicals as well as bulk compounds, in order to create a competitive alternative to traditional chemical procedures. This does not necessarily mean that an existing chemical process will always be entirely substituted by an application of biotechnological methods, but it is to be expected that they will be combined to an increasing extent with conventional chemical technologies, thus contributing to a reduced use of hazardous substances, to minimize energy consumption, and to a reduction in waste generation. Another important aspect is to integrate, wherever possible, renewable raw materials in production processes; for reviews see e.g. Zechendorf, 1999, Busch et al., 2006; Soetaert and Vandamme, 2006.
A variety of biotechnological tools have been developed in the last two decades to make enzyme- or whole-cell-catalyzed reactions more efficient under given process conditions (Bornscheuer et al., 2012; see also Chapters 15, 16). The metabolic engineering of production strains such as the often employed Escherichia coli by insertion of a foreign gene into its genome or by transformation with a plasmid containing the desired gene has benefited from recent developments in what is known as systems biology enabling amongst others a holistic view on biocatalysis. This approach is based on one of the famous statements made by the Greek philosopher Aristotle (384 to 322 BCE; a pupil of Plato and a teacher of Alexander the Great): “The whole is more than the sum of its parts” (shorten...