
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
About this book
How can I use my HPLC/UHPLC equipment in an optimal way, where are the limitations of the technique? These questions are discussed in detail in the sequel of the successful "HPLC Expert" in twelve chapters written by experts in the respective fields. The topics encompass - complementary to the first volume - typical HPLC users' problems and questions such as gradient optimization and hyphenated techniques (LC-MS). An important key aspect of the book is UHPLC: For which analytical problem is it essential, what should be considered? Besides presentation of latest developments directly from the main manufacturers, also UHPLC users and independent service engineers impart their knowledge. Consistent with the target groups, the level is advanced, but the emphasis is on practical applications.
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
Chapter 1
When Should I Use My UHPLC as a UHPLC?
Stavros Kromidas
1.1 Introduction
Modern analytical LC systems are designed without exception as ultrahigh-performance liquid chromatography (UHPLC) systems. However, outside of pure research laboratories, a maximum of about 20–30% of the separations are performed under UHPLC conditions. By this, pressures above approximately 800 bar are meant. In which cases does it make sense, or is even necessary, to use the existing UHPLC system under truly UHPLC conditions? On the other hand, when should the UHPLC system perhaps be used as a fast, but “classical,” high-performance liquid chromatography (HPLC)? This chapter deals with exactly this point. To this end, the answers to two questions can help, both of which we will deal with. The first is “What do I really need?” Here it is necessary to define which characteristics of an HPLC method in exactly this situation are in the foreground, among others, for example, short retention times, a robust method, maximum resolution/peak capacity, and low detection limit. The second question is much simpler: “Why is the UHPLC more capable than the HPLC?” Afterward, we will discuss the key question: “How do I reasonably combine my requirements on the method and the potential of UHPLC – taking into consideration the real laboratory situation?”
Note
Familiarity with the theoretical background is assumed, and the principles of HPLC optimization are therefore only mentioned but not derived. For this, reference is made to the relevant literature (for example, [1–5]).
1.2 What Do I Want to Achieve and What Is a UHPLC Capable of?
1.2.1 What Do I Want to Achieve?
One often wants more than only one attribute from an HPLC method, for example, “good” and “fast” separation. However, before deciding on the method design – which indeed includes the question of the necessity of UHPLC conditions – two points urgently have to be clarified. Firstly, what are the peculiarities of the method, and how is its environment? Here we are interested in, among other things, the following crucial features of the proposed analysis: matrix, time required for sample preparation and manual reintegration, experience of the user, changing or constant chromatographic conditions, research or routine laboratory, and so on. Secondly, what is the primary requirement for this method in the specific case? The main objective should be clearly identified, a second (or third?) regarded merely as a wish, for example: “In this case we need, for this reason the maximum possible sensitivity – if the method is also precise, that would be good too … ” Four typical requirements for an HPLC method, which we will subsequently consider in more detail, are listed as follows:
- Good separation: this can mean, firstly, sufficient resolution – separation between two critical peaks or possibly between 2 and 3 relevant peak pairs. Or, secondly, sufficient peak capacity – separation of many (or all?) – possibly chemically similar components, see Section 1.3.1
- Fast separation: short retention times; this often goes hand in hand with a low solvent consumption, see Section 1.3.2
- Sensitive measurement: decrease in the detection limit, which means an improvement in the relative mass sensitivity, see Section 1.3.3
- Robust conditions: reliable methods, which lead to the avoidance of repeat measurements and minimization of equipment downtime, see Section 1.3.4.
1.2.2 What Is a UHPLC Capable of?
Put simply, a UHPLC system is an instrument that, first of all, compared to an HPLC system, has about 10 times lower dead volume (dispersion volume or “Extracolumn Volume”: the volume from the autosampler to the detector without a column) and also dwell/delay volume (the volume from the mixing valve/mixing chamber to the head of the column).The dead volume of a modern UHPLC system is nowadays about ≤7–10 µl, with the aid of special kits even about ≤4 µl, the dwell volumes are about 100–200 µl with low-pressure gradient (LPG), and about 25–35 µl with high-pressure gradient (HPG) systems.
Note
Nowadays, we talk less of “Extracolumn Volume” but rather of “Extracolumn Dispersion.” This takes into account the fact that the geometry of, for example, connections and mixing valves, and thus the flow profile, has more influence on the peak broadening than the absolute dead volume, see also Chapter 3. Secondly, a modern UHPLC system allows working pressures up to around 1500 bar.
1.3 What Is Required from an HPLC Method?
1.3.1 Separate Well
First of all, we show briefly how the separation in HPLC can be improved in principle, and then we will have a closer look at the contribution UHPLC can make toward a better separation.
In chromatography, we distinguish with respect to the quality of a separation between two cases:
- 1. I am really only interested in one or a few components. It is therefore a question of– according to my individual criteria – sufficient separation between the component of interest and an “interfering” component – in other words, ultimately on the separation of two peaks. The focus can be on the critical pair (e.g., main and secondary components), possibly on two to three more peak pairs. The criterion here is the resolution, and when simplified, it describes the distance between the peaks at the baseline. 1.1
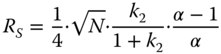
- where R = resolution, N = plate number (fundamentally defined for isocratic conditions), α = separation factor (formerly selectivity factor), and k = retention factor (formerly capacity factor k′).
- 2. I want to or have to separate “all” existing peaks sufficiently well, that is, when possible with baseline separation. In this case, the peak capacity comes into play. This is the total number of peaks that I can separate in a certain time with a sufficiently good resolution (commonly R = 1). The sum of all resolutions is often stated as a measure of the peak capacity. In the literature, one finds several formulas for the peak capacity, we consider here the two simplest: 1.2a
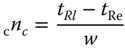
- or 1.2b
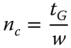
- where nc = peak capacity, tRl = retention time of the last peak, tRe = retention time of the first peak, w = peak width, and tG = gradient duration.
Note
About 70–80% of separations, nowadays, are gradient separations. Consequently, today's HPLC/UHPLC system is a high- or low-pressure gradient with DAD and/or MS/MS, and furthermore, aerosol detectors are becoming more common. The thoughts presented here apply in principle for both isocratic and gradient separations, but for the aforementioned reason, I will lay the focus a little more on gradient separations.
Let us look first at the resolution.
Equation 1.1 shows that the resolution can be improved by increasing efficiency, selectivity, and retention. The requirement for the retention term is strong interactions, the optimum value lies around k ≈ 3–5, and this means that the peaks of interest should elute by or after approximately three to five times the dead or mobile time. From Equation 1.1, it can be seen that the term for selectivity, and thus the separation factor α, is by far the most sensitive function of the resolution: α − 1/α! On the other hand, the plate number is under the root, a doubling of N improves the resolution by a factor of “only” 1.4. Two numerical examples illustrate this; for a detailed discussion, see [6]:
- 1. Assume that two peaks elute with an α-value of 1.01. To achieve baseline separation of these two peaks, one would need about 160 000 plates. If the α-value could be increased from 1.01 to 1.10, for the same resolution, just less than 2000 plates would be required. Even a seemingly small improvement in the α-value from 1.01 to 1.05 means that instead of 160 000 plates, only about 6000 plates are necessary.
- 2. Further assume that we have a separation with the following values: k = 2, α = 1.05, and N = 9000. This results in a resolution of R = 0.76. This is not enough, and the resolution should be improved. To start with, the interactions can be increased, for example, through a more hydrophobic stationary phase or more water in the mobile phase. Assuming that the stronger interactions affect the two components equally, then the selectivity remains constant. The k-value increases from k = 2 to, for example, k = 6, and the resolution increases to R = 0.97. Alternatively, one could use a column with 15 000 plates, and the resolution improves to R = 0.98. Both measures are therefore correct; however, they are not particularly effective when it comes to significantly improving the resolution. If the α-value could be increased from 1.05 to 1.10, this would result in a resolution of R = 1.45. Let us finish the second example with the following observation: when two peaks ...
Table of contents
- Cover
- Title Page
- Copyright
- Table of Contents
- List of Contributors
- Foreword
- The Structure of “The HPLC-Expert 2”
- Chapter 1: When Should I Use My UHPLC as a UHPLC?
- Part I: Hardware and Software, Separation Modes, Materials
- Part II: Experience Reports, Trends
- About the Authors
- Index
- End User License Agreement
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access The HPLC Expert II by Stavros Kromidas in PDF and/or ePUB format, as well as other popular books in Physical Sciences & Analytic Chemistry. We have over 1.5 million books available in our catalogue for you to explore.