The definitive textbook on the chemical analysis of pharmaceutical drugs – fully revised and updated
Introduction to Pharmaceutical Analytical Chemistry enables students to gain fundamental knowledge of the vital concepts, techniques and applications of the chemical analysis of pharmaceutical ingredients, final pharmaceutical products and drug substances in biological fluids. A unique emphasis on pharmaceutical laboratory practices, such as sample preparation and separation techniques, provides an efficient and practical educational framework for undergraduate studies in areas such as pharmaceutical sciences, analytical chemistry and forensic analysis. Suitable for foundational courses, this essential undergraduate text introduces the common analytical methods used in quantitative and qualitative chemical analysis of pharmaceuticals.
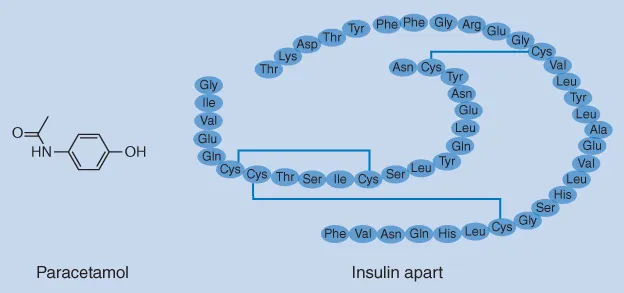
This extensively revised second edition includes a new chapter on chemical analysis of biopharmaceuticals, which includes discussions on identification, purity testing and assay of peptide and protein-based formulations. Also new to this edition are improved colour illustrations and tables, a streamlined chapter structure and text revised for increased clarity and comprehension.
- Introduces the fundamental concepts of pharmaceutical analytical chemistry and statistics
- Presents a systematic investigation of pharmaceutical applications absent from other textbooks on the subject
- Examines various analytical techniques commonly used in pharmaceutical laboratories
- Provides practice problems, up-to-date practical examples and detailed illustrations
- Includes updated content aligned with the current European and United States Pharmacopeia regulations and guidelines
Covering the analytical techniques and concepts necessary for pharmaceutical analytical chemistry, Introduction to Pharmaceutical Analytical Chemistry is ideally suited for students of chemical and pharmaceutical sciences as well as analytical chemists transitioning into the field of pharmaceutical analytical chemistry.