Specifically geared to personnel in the pharmaceutical and biotechnology industries, this book describes the basics and challenges of oral bioavailability – one of the most significant hurdles in drug discovery and development.
• Describes approaches to assess pharmacokinetics and how drug efflux and uptake transporters impact oral bioavailability
• Helps readers reduce the failure rate of drug candidates when transitioning from the bench to the clinic during development
• Explains how preclinical animal models – used in preclinical testing – and in vitro tools translate to humans, which is an underappreciated and complicated area of drug development
• Includes chapters about pharmacokinetic modelling, the Biopharmaceutics Drug Disposition Classification System (BDDCS), and the Extended Clearance Classification System (ECCS)
• Has tutorials for applying strategies to medicinal chemistry practices of drug discovery/development

eBook - ePub
Oral Bioavailability Assessment
Basics and Strategies for Drug Discovery and Development
- English
- ePUB (mobile friendly)
- Available on iOS & Android
eBook - ePub
Oral Bioavailability Assessment
Basics and Strategies for Drug Discovery and Development
About this book
Trusted by 375,005 students
Access to over 1.5 million titles for a fair monthly price.
Study more efficiently using our study tools.
Information
CHAPTER 1
DRUG PHARMACOKINETICS AND TOXICOKINETICS
1.1 INTRODUCTION
Pharmacokinetics (PK) is the science that describes the time-course of drug concentration in the body resulting from administration of a certain drug dose. Similarly, toxicokinetics (TK) is the science that investigates how the body handles toxicants as illustrated by its plasma profile at various time points. In comparison, pharmacodynamics (PD) is the science that describes the relationship of the time-course of drug concentration and its effects in the body [1, 2].
PK is considered a biomarker of drug exposure as well as marker of efficacy and safety. Key determinants of the pharmacokinetics of a drug include absorption, distribution, metabolism, and elimination (ADME) [3]. Discovering novel therapeutic agents is an increasingly time-consuming and costly process. Most estimates indicate that it takes approximately 10–15 years and more than $1.2 billion to discover and develop a successful drug product [4]. It is well established that poor drug PK is one of the leading causes of compounds failure in preclinical and clinical drug development [5]. For example, attrition due to poor pharmacokinetics contributed to 10% of the attrition reported for compounds developed by the pharmaceutical industry in 2001 (Figure 1.1) [6].
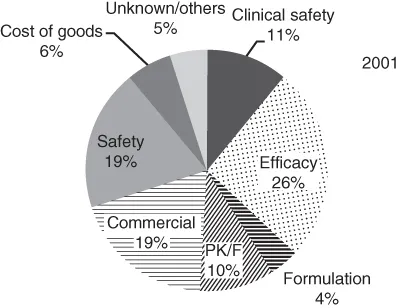
Figure 1.1 The contribution of various factors to the overall attrition of NCEs in year 2001.
Kola and Landis 2004 [6]. Reproduced with permission of Nature Publishing Group.
Compounds with poor PK profile tend to have low oral systemic plasma exposure and high interindividual variability, which limits their therapeutic utility (Figure 1.2) [7]. Therefore, a better understanding of the PK profile early on enables the discovery of compounds with drug-like properties [8]. In drug discovery settings, the main outcomes of PK/TK assessments are to
- select compounds with the maximum potential of reaching the target;
- determine the appropriate route of administration to deliver the drug (typically oral);
- understand how the drug blood levels relate to efficacy or toxicity in order to choose efficacious and safe doses;
- facilitate appropriate dose sections for rodent and/or nonrodent species in toxicology testing and drug safety evaluation;
- decide on the frequency and duration of dosing in order to maintain adequate drug concentration at target for disease modification; and
- accurately predict the PK in humans profile prior to clinical studies.
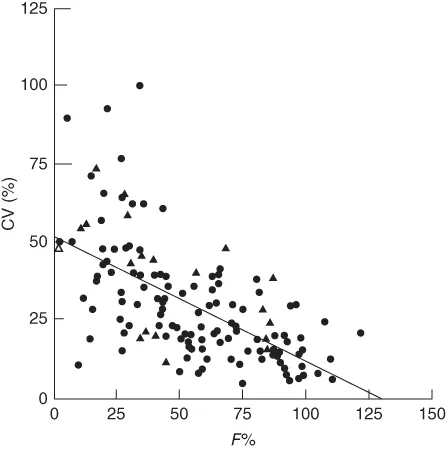
Figure 1.2 The relationship between drug oral bioavailability and interindividual variability reported as coefficient of variation (%).
Hellriegel et al. 1996 [7]. Reproduced with permission of John Wiley & Sons.
A PK/TK study involves dosing animals or humans with NCE and collect blood samples at predefined time points. After sample preparation and quantification, a concentration–time profile is generated (Figure 1.3). In drug discovery, preliminary PK studies are usually conducted in rodents to evaluate the extent of drug exposure in vivo. These rodent studies are commonly followed by studies in nonrodents such as dogs or monkeys to better characterize the PK profile of the compound and to support safety risk assessment studies. Pharmacokinetic scaling, also known as allometry, is a discipline that was extensively used in the past to predict human PK profile using preclinical data and in predicting the drug human half-life, dose, and extent of absorption. This approach is based on empirical observations that various physiological parameters are a function of body size. The allometric methods assume that the same metabolic and disposition processes in the species evaluated are correlated with those observed in humans. However, the cytochrome P450 enzymes in the rat are not the same as those in humans, and thus, may exhibit altered disposition of the compound or even produce different metabolite patterns (see Chapter 2) [9, 10]. Similarly, uptake and efflux transporters in the animal species may differ in substrate specificity or rate, as compared to humans, and thus may confound predictions of human PK [11]. Accurate prediction of human pharmacokinetic profile is imperative to minimize drug failur...
Table of contents
- COVER
- WILEY SERIES ON PHARMACEUTICAL SCIENCE AND BIOTECHNOLOGY: PRACTICES, APPLICATIONS, AND METHODS
- TITLE PAGE
- COPYRIGHT
- TABLE OF CONTENTS
- DEDICATION
- CHAPTER 1: DRUG PHARMACOKINETICS AND TOXICOKINETICS
- CHAPTER 2: GIT ANATOMY AND PHYSIOLOGY AND DRUG ORAL BIOAVAILABILITY: IMPACT OF SPECIES DIFFERENCES
- CHAPTER 3: DRUG ROUTES OF EXCRETION
- CHAPTER 4: PHYSICOCHEMICAL AND BIOPHARMACEUTICAL PROPERTIES THAT AFFECT DRUG ABSORPTION OF COMPOUNDS ABSORBED BY PASSIVE DIFFUSION
- CHAPTER 5: PHYSICOCHEMICAL AND BIOPHARMACEUTICAL FACTORS AFFECTING HEPATIC/INTESTINAL FIRST-PASS EFFECT
- CHAPTER 6: IMPACT OF INTESTINAL EFFLUX TRANSPORTERS ON ORAL ABSORPTION
- CHAPTER 7: IMPACT OF INFLUX TRANSPORTERS ON DRUG ABSORPTION
- CHAPTER 8: EXTENDED CLEARANCE CLASSIFICATION SYSTEM (ECCS) AND ITS UTILITY IN PREDICTING CLEARANCE RATE-DETERMINING STEP IN DRUG DISCOVERY
- CHAPTER 9: IN VITRO AND IN SITU APPROACHES TO MEASURE INTESTINAL PERMEABILITY AND EFFLUX TRANSPORTERS
- CHAPTER 10: IN SILICO APPROACHES TO PREDICT INTESTINAL PERMEABILITY
- CHAPTER 11: IN VIVO PRECLINICAL APPROACHES TO DECONVOLUTE THE CONTRIBUTION OF FIRST-PASS EFFECT FROM ORAL ABSORPTION
- CHAPTER 12: IN VITRO APPROACHES TO ASSESS HEPATIC METABOLISM AND FIRST-PASS EFFECT
- CHAPTER 13: THE UTILITY OF ECCS AS A ROADMAP TO IMPROVE ORAL BIOAVAILABILITY OF NEW MOLECULAR ENTITIES: INDUSTRIAL PERSPECTIVE
- INDEX
- END USER LICENSE AGREEMENT
Frequently asked questions
Yes, you can cancel anytime from the Subscription tab in your account settings on the Perlego website. Your subscription will stay active until the end of your current billing period. Learn how to cancel your subscription
No, books cannot be downloaded as external files, such as PDFs, for use outside of Perlego. However, you can download books within the Perlego app for offline reading on mobile or tablet. Learn how to download books offline
Perlego offers two plans: Essential and Complete
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.5M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
We are an online textbook subscription service, where you can get access to an entire online library for less than the price of a single book per month. With over 1.5 million books across 990+ topics, we’ve got you covered! Learn about our mission
Look out for the read-aloud symbol on your next book to see if you can listen to it. The read-aloud tool reads text aloud for you, highlighting the text as it is being read. You can pause it, speed it up and slow it down. Learn more about Read Aloud
Yes! You can use the Perlego app on both iOS and Android devices to read anytime, anywhere — even offline. Perfect for commutes or when you’re on the go.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app
Yes, you can access Oral Bioavailability Assessment by Ayman F. El-Kattan, Mike S. Lee in PDF and/or ePUB format, as well as other popular books in Medicine & Pharmacology. We have over 1.5 million books available in our catalogue for you to explore.