Chromatography
Basic Principles, Sample Preparations and Related Methods
- English
- ePUB (mobile friendly)
- Available on iOS & Android
Chromatography
Basic Principles, Sample Preparations and Related Methods
About this book
Finally a book on chromatography which is easy to grasp for undergraduates and technicians; covers the area in sufficient depth while still being concise. The book includes all recent technology advances and has core textbook features further improving the learning experience.
This book is the perfect introduction into a methodology which is the underlying principle of the vast majority of separation methods worldwide. Everyone working in a lab environment must be familiar with the basis of these technologies and Tyge Greibrokk, Elsa Lundanes and Leon Reubsaet succeed in delivering a text which is easy to read for undergraduates and laboratory technicians, and covers the area in sufficient depth while still being concise. The book includes all recent technology advances and has core textbook features further improving the learning experience.
Tools to learn more effectively

Saving Books

Keyword Search

Annotating Text

Listen to it instead
Information
1.1 Introduction
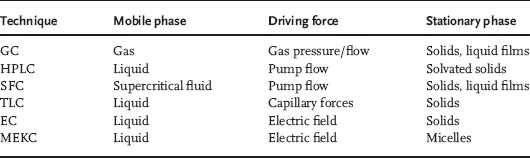
1.2 Migration and Retention
1.2.1 General
1.2.2 Mo...
Table of contents
- Cover
- Related Titles
- Title Page
- Copyright
- Preface
- Chapter 1: General Concepts
- Chapter 2: Gas Chromatography
- Chapter 3: High-Performance Liquid Chromatography (HPLC)
- Chapter 4: Thin Layer Chromatography (TLC)
- Chapter 5: Supercritical Fluid Chromatography
- Chapter 6: Electrophoresis and Potential-Driven Chromatography
- Chapter 7: Chromatography on a Chip
- Chapter 8: Field-Flow Fractionation
- Chapter 9: Sample Preparation
- Chapter 10: Quantitation
- Index
Frequently asked questions
- Essential is ideal for learners and professionals who enjoy exploring a wide range of subjects. Access the Essential Library with 800,000+ trusted titles and best-sellers across business, personal growth, and the humanities. Includes unlimited reading time and Standard Read Aloud voice.
- Complete: Perfect for advanced learners and researchers needing full, unrestricted access. Unlock 1.4M+ books across hundreds of subjects, including academic and specialized titles. The Complete Plan also includes advanced features like Premium Read Aloud and Research Assistant.
Please note we cannot support devices running on iOS 13 and Android 7 or earlier. Learn more about using the app